Hyperconjugation
•Download as PPTX, PDF•
15 likes•15,006 views
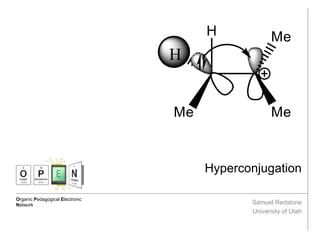
Hyperconjugation is the donation of a sigma bond into an adjacent empty or partially filled p orbital, which results in an increased stability of the molecule. Contributed by: Samuel Redstone (Undergraduate), University of Utah, 2016
Report
Share
Report
Share
Recommended
Hyperconjugation - organic chemistry

This presentation describes the concept of Hyperconjugation in simple words, gives definition of hyperconjugation, explains why it is called as 'No bond Resonance' and gives the effects of hyperconjugation on the chemical properties of compounds: alkyl cations and their relative stability, alkyl radicals and their relative stability, alkenes and their relative stability, bond length, anomeric effect and Baker - Nathan effect.
Hyperconjugation effect

This document discusses the concept of hyperconjugation, which involves the delocalization of sigma electrons from an adjacent C-H bond into an empty p-orbital of an unsaturated system like an alkene or benzene ring. This effect increases the stability of alkenes and carbocations with more alkyl substituents by allowing for additional no bond resonance structures. The stability of alkenes and carbocations increases with the number of alkyl groups due to greater hyperconjugative stabilization from more C-H bonds. Hyperconjugation is an important effect that helps explain the observed stability and reactivity patterns of unsaturated organic compounds.
Hyperconjugation

Dr. Neelam from the Department of Chemistry presented on the topic of hyperconjugation. Hyperconjugation is the delocalization of σ-electrons from a C-H bond into an adjacent unsaturated system. It can occur in alkenes, alkynes, carbocations, and carbon radicals. The number of possible hyperconjugative structures equals the number of alpha hydrogens on sp3 hybridized carbon atoms. Hyperconjugation explains trends in stability and heats of hydrogenation between different alkenes. It is a permanent effect that does not change hybridization and is distance independent.
Inductive & mesomeric effect s k katoch

This document discusses inductive effect and mesomeric (or resonance) effect, including their definitions, types, and applications. The key points are:
1. Inductive effect is the polarization of a sigma bond due to electron donating or withdrawing groups, transmitted through sigma bonds. It influences chemical and physical properties.
2. Mesomeric effect is polarity produced between pi bonds or a pi bond and lone pair, leading to electron delocalization. Groups like -NO2 show negative mesomeric effect while -OH shows positive effect.
3. Applications include reactivity patterns of aromatic compounds affected by activating or deactivating groups, stability of carbocations/carbanions, and acid/base
Conjugation

Conjugation involves the overlap of p-orbitals across intervening sigma bonds, creating a system of delocalized electrons within alternating single and multiple bonds. This conjugated system can be cyclic, linear, or mixed and increases stability compared to non-conjugated systems. Hyperconjugation also stabilizes compounds through the interaction of sigma bonds with adjacent empty or partially filled p-orbitals, without actual bond resonance. Both conjugation and hyperconjugation extend molecular orbitals to increase stability.
Organic reactions and mechanisms

The document discusses organic reactions and reaction mechanisms. It defines nucleophiles and electrophiles, and provides examples of each. It then summarizes several common types of organic reactions including addition reactions, substitution reactions, elimination reactions, and aromatic substitutions. The mechanisms and examples of nucleophilic addition, electrophilic addition, nucleophilic substitution, and electrophilic aromatic substitutions like nitration, sulfonation, and halogenation are described in detail.
Inductive effect

This is a presentation file on Inductive Effect, Bond Length, Bond Energy, Bond Angle for the course Organic Pharmacy I, course code is PHAR-1105 specially for the pharmacy students. Also it can be used for the Biochemistry students and other like as HSC level in Bangladesh or another country. We are the students of Mawlana Bhashani Science and Technology University, department of pharmacy which is located in Tangail.
Thank you for your patient.
Aromaticity

This document discusses aromaticity, including its introduction, criteria for aromatic compounds, Hückel's rule, examples of aromatic and anti-aromatic compounds, and non-aromatic compounds. Aromatic compounds are cyclic, planar, and have delocalized pi electrons that follow Hückel's rule of 4n+2 pi electrons. Benzene is used to originally define aromaticity. Resonance contributes greatly to aromatic stability. Anti-aromatic compounds have 4n pi electrons and are destabilized by cyclic pi electron delocalization. Cyclooctatetraene is provided as an example of a non-aromatic compound for not being planar.
Recommended
Hyperconjugation - organic chemistry

This presentation describes the concept of Hyperconjugation in simple words, gives definition of hyperconjugation, explains why it is called as 'No bond Resonance' and gives the effects of hyperconjugation on the chemical properties of compounds: alkyl cations and their relative stability, alkyl radicals and their relative stability, alkenes and their relative stability, bond length, anomeric effect and Baker - Nathan effect.
Hyperconjugation effect

This document discusses the concept of hyperconjugation, which involves the delocalization of sigma electrons from an adjacent C-H bond into an empty p-orbital of an unsaturated system like an alkene or benzene ring. This effect increases the stability of alkenes and carbocations with more alkyl substituents by allowing for additional no bond resonance structures. The stability of alkenes and carbocations increases with the number of alkyl groups due to greater hyperconjugative stabilization from more C-H bonds. Hyperconjugation is an important effect that helps explain the observed stability and reactivity patterns of unsaturated organic compounds.
Hyperconjugation

Dr. Neelam from the Department of Chemistry presented on the topic of hyperconjugation. Hyperconjugation is the delocalization of σ-electrons from a C-H bond into an adjacent unsaturated system. It can occur in alkenes, alkynes, carbocations, and carbon radicals. The number of possible hyperconjugative structures equals the number of alpha hydrogens on sp3 hybridized carbon atoms. Hyperconjugation explains trends in stability and heats of hydrogenation between different alkenes. It is a permanent effect that does not change hybridization and is distance independent.
Inductive & mesomeric effect s k katoch

This document discusses inductive effect and mesomeric (or resonance) effect, including their definitions, types, and applications. The key points are:
1. Inductive effect is the polarization of a sigma bond due to electron donating or withdrawing groups, transmitted through sigma bonds. It influences chemical and physical properties.
2. Mesomeric effect is polarity produced between pi bonds or a pi bond and lone pair, leading to electron delocalization. Groups like -NO2 show negative mesomeric effect while -OH shows positive effect.
3. Applications include reactivity patterns of aromatic compounds affected by activating or deactivating groups, stability of carbocations/carbanions, and acid/base
Conjugation

Conjugation involves the overlap of p-orbitals across intervening sigma bonds, creating a system of delocalized electrons within alternating single and multiple bonds. This conjugated system can be cyclic, linear, or mixed and increases stability compared to non-conjugated systems. Hyperconjugation also stabilizes compounds through the interaction of sigma bonds with adjacent empty or partially filled p-orbitals, without actual bond resonance. Both conjugation and hyperconjugation extend molecular orbitals to increase stability.
Organic reactions and mechanisms

The document discusses organic reactions and reaction mechanisms. It defines nucleophiles and electrophiles, and provides examples of each. It then summarizes several common types of organic reactions including addition reactions, substitution reactions, elimination reactions, and aromatic substitutions. The mechanisms and examples of nucleophilic addition, electrophilic addition, nucleophilic substitution, and electrophilic aromatic substitutions like nitration, sulfonation, and halogenation are described in detail.
Inductive effect

This is a presentation file on Inductive Effect, Bond Length, Bond Energy, Bond Angle for the course Organic Pharmacy I, course code is PHAR-1105 specially for the pharmacy students. Also it can be used for the Biochemistry students and other like as HSC level in Bangladesh or another country. We are the students of Mawlana Bhashani Science and Technology University, department of pharmacy which is located in Tangail.
Thank you for your patient.
Aromaticity

This document discusses aromaticity, including its introduction, criteria for aromatic compounds, Hückel's rule, examples of aromatic and anti-aromatic compounds, and non-aromatic compounds. Aromatic compounds are cyclic, planar, and have delocalized pi electrons that follow Hückel's rule of 4n+2 pi electrons. Benzene is used to originally define aromaticity. Resonance contributes greatly to aromatic stability. Anti-aromatic compounds have 4n pi electrons and are destabilized by cyclic pi electron delocalization. Cyclooctatetraene is provided as an example of a non-aromatic compound for not being planar.
Electrochemistry 

1. Electrochemistry deals with the transformation of electrical energy to chemical energy and vice versa. It involves the chemical applications of electricity.
2. An electrolytic cell converts electrical energy to chemical energy, while an electrochemical cell converts chemical energy to electrical energy.
3. Arrhenius' theory of electrolytic dissociation states that when an electrolyte dissolves in water, it breaks up into ions. There is a dynamic equilibrium between the ionized and non-ionized molecules. The degree of ionization depends on factors like the ionization constant.
Aromaticity

The document summarizes aromaticity and related topics for chemistry students. It discusses:
- Benzenoid and non-benzenoid aromatic compounds, including their properties and reactions.
- Resonance structures of benzene and how it follows Huckel's rule for aromaticity.
- Classification of compounds based on aromaticity and examples of antiaromatic compounds.
- Aromatic ions and heterocyclic aromatic compounds like pyrrole, furan and pyridine.
Carbenes .......

1. Carbenes are neutral molecules containing a divalent carbon atom with two unshared valence electrons. They exist in both singlet and triplet states depending on the electronic spin.
2. Carbenes undergo insertion reactions into X-H and C-C bonds. They also add across double bonds, with singlet carbenes preserving alkene stereochemistry and triplet carbenes losing it.
3. Carbenes are generated by reactions such as α-elimination of halogenated compounds with base or decomposition of diazo compounds. They can rearrange through migrations such as the Wolff or Arndt-Eistert reactions.
Unsaturated Carbonyl Compound

1) α,β-Unsaturated carbonyl compounds contain a carbonyl group and a conjugated carbon-carbon double bond separated by one carbon-carbon single bond.
2) These compounds undergo both electrophilic and nucleophilic addition reactions due to the conjugation between the carbonyl and double bond.
3) Common reactions include the Michael addition, in which a carbanion adds to the β-carbon, and the Diels-Alder reaction, where a conjugated diene adds to form a six-membered ring.
Aromaticity and Anti-Aromaticity 

1) The document summarizes aromaticity and anti-aromaticity, discussing the key criteria for aromatic compounds including being cyclic, conjugated, planar, and having 4n+2 π electrons.
2) It provides examples of aromatic compounds like benzene and non-aromatic compounds that do not meet the criteria. Anti-aromatic compounds are also discussed.
3) NMR spectroscopy is discussed as a way to distinguish aromatic protons from other types of protons based on chemical shift values.
Reaction mechanisms

This document discusses organic reactions and mechanisms. It defines key terms like substrate, reagent, products, and mechanism. It describes how factors like inductive and mesomeric effects can influence reactions by altering electron density. It also discusses different types of reaction intermediates that can form, such as carbonium ions, carbanions, free radicals, and carbenes. The document classifies reagents as electrophiles or nucleophiles and describes their behaviors. It explains concepts like activation energy and the transition state that systems must go through for a reaction to occur.
Hybridization- sp, sp2 and sp3

Hybridization is the idea that atomic orbitals fuse to form newly hybridized orbitals, which in turn, influences molecular geometry and bonding properties. Hybridization is also an expansion of the valence bond theory.
Carbocation ppt

An organic species which has a carbon atom bearing only six electrons in its outermost shell and has a positive charge is called carbocation.
The positively charged carbon of carbocation is sp2 hybridized.
The unhybridized p-orbital remains vacant.
They are highly reactive and act as reaction intermediate.
They are also called carbonium ion.
Inductive Effect

Inductive Effect is the important topic in organic chemistry that gives us idea about electron withdrawing and accepting module of the specific group that determines the reactivity of the molecule or compound.
Electronic effects inductive,mesomeric,electromeric

This document discusses different electronic effects in organic chemistry. It describes inductive effect as a permanent polarization of electron density between two unlike atoms in a bond. Electron-withdrawing and electron-releasing groups are discussed. Mesomeric effect allows for resonance stabilization through delocalization of charge in conjugated systems. Electromeric effect involves the temporary shift of a pi bond electron pair to one atom upon reaction with an electrophile. The order of effects from strongest to weakest is given as mesomeric, electromeric, and inductive. Applications to stability of ions and carbocations are outlined.
Reaction intermediates

Reaction intermediates such as carbocation, carbanion, free radicals, carbene, nitrene and ylides were discussed.
Carbanion organic ppt

This document discusses carbanions, which are negatively charged organic species where carbon carries three bond pairs and one lone pair. Carbanions are stabilized through conjugation, resonance effects, field effects, and aromaticity. They are generated through heterolytic bond cleavage or addition of a negative ion to a carbon-carbon multiple bond. As nucleophiles, carbanions undergo reactions such as alpha-halogenation of ketones, additions to carbonyls, nucleophilic acyl substitutions, substitutions with alkyl halides, and Michael additions.
Resonance ,inductive effect -----(Organic )

Resonance occurs when a molecule can be represented by multiple Lewis structures and the actual structure is a hybrid of all the contributing structures. A resonance hybrid is the average structure between all the canonical or contributing Lewis structures. Delocalization of electrons through resonance leads to decreased potential energy and increased stability compared to any single contributing structure. Electron donating groups cause positive resonance or inductive effects while electron withdrawing groups cause negative resonance or inductive effects. Resonance and inductive effects influence acidity, basicity, and reactivity.
Basic effects in Organic chemistry

Basic effects in Organic chemistrySri Ramakrishna Mission Vidyalaya College of Arts and Science,Coimbatore-20.
This presentaion describes about the basic principle effects in organic chemistry like inductive,mesomeric,electromeric, resonance and hyperconjugation. this presentation contains some JAM competitive questions.Mesomeric Effect

Mesomeric (or resonance) effect refers to the polarity produced in a molecule through delocalization of pi electrons between bonds or a bond and lone pair. This effect can increase or decrease electron density in different parts of a molecule. Negative mesomeric effects are shown by groups like nitro (-NO2) that withdraw electron density, while positive effects are shown by groups like hydroxyl (-OH) that release electron density. Understanding mesomeric effects helps explain a molecule's reactivity toward electrophiles and nucleophiles by determining where electron density is increased or decreased.
Inductive effect pptx

This document discusses electronic displacement in organic compounds. It describes two types of electronic displacement: permanent displacement including inductive, resonance, and mesomeric effects; and temporary displacement through electromeric effects. Inductive effects are further broken down into +I effects where groups donate electron density and -I effects where groups withdraw electron density. Examples of inductive effects include their impact on acid/base strength, stability of carbocations/carbanions, and dipole moments.
Conformational analysis of cyclohexane

Cyclohexane exists in different conformations viz chair, boat, twist boat and half chair. These conformations possess different energies. Therefore they differ in energy.
Topicity

Homomorphic ligands / groups/ atoms, Homotopic, heterotopic, enantiotopic, prochirality and diastereotopic ligands and faces with examples
Resonance----(Oganic)

Resonance structures represent different arrangements of electrons in a molecule that have the same positions of nuclei but different bonding patterns. Resonance contributes to the stability of molecules like benzene by delocalizing electrons across multiple equivalent structures. The actual structure of a molecule represented by resonance is a hybrid of the contributing structures, with bond lengths intermediate between single and double bonds. Delocalization of electrons is depicted using curved arrows between resonance structures.
Resonance

Resonance effect occurs when a compound can be represented by two or more Lewis structures with the same arrangement of atoms. These contributing structures are called resonance structures.
The resonance structures are hypothetical and do not represent real molecules individually. Together they contribute to a resonance hybrid structure that is more stable than any individual resonance structure. The most stable contributing structure contributes the most to the resonance hybrid.
Conditions for resonance include sp or sp2 hybridized atoms involved, overlapping parallel p-orbitals to form a conjugated system. The sigma bond framework remains the same between structures, atomic positions do not change, and electron counts are equal.
Inductive effect

This document discusses inductive effect, which is the permanent displacement of a carbon chain's shared electron pair towards the more electronegative atom or group. It describes the negative inductive effect caused by electron-withdrawing groups and the positive inductive effect of electron-donating groups. Finally, it explains how inductive effect influences the strength of organic acids and bases, with acid strength decreasing as positive inductive effect or negative inductive effect decreases.
IR spectroscopy

Infrared spectroscopy involves using infrared radiation to analyze materials. Molecules absorb specific infrared frequencies that are characteristic of their structure, such as bond vibrations and stretches. There are two main methods for infrared spectroscopy - scanning monochromator which analyzes one wavelength at a time, and Fourier transform infrared spectroscopy which uses interferometry to measure all infrared wavelengths simultaneously. Fourier transform then converts this raw interferogram data into the infrared spectrum. Infrared spectroscopy can be used to identify functional groups and molecular structures in compounds like 1-Hexene, Toluene, and Cyclohexanol based on their characteristic absorption peaks.
More Related Content
What's hot
Electrochemistry 

1. Electrochemistry deals with the transformation of electrical energy to chemical energy and vice versa. It involves the chemical applications of electricity.
2. An electrolytic cell converts electrical energy to chemical energy, while an electrochemical cell converts chemical energy to electrical energy.
3. Arrhenius' theory of electrolytic dissociation states that when an electrolyte dissolves in water, it breaks up into ions. There is a dynamic equilibrium between the ionized and non-ionized molecules. The degree of ionization depends on factors like the ionization constant.
Aromaticity

The document summarizes aromaticity and related topics for chemistry students. It discusses:
- Benzenoid and non-benzenoid aromatic compounds, including their properties and reactions.
- Resonance structures of benzene and how it follows Huckel's rule for aromaticity.
- Classification of compounds based on aromaticity and examples of antiaromatic compounds.
- Aromatic ions and heterocyclic aromatic compounds like pyrrole, furan and pyridine.
Carbenes .......

1. Carbenes are neutral molecules containing a divalent carbon atom with two unshared valence electrons. They exist in both singlet and triplet states depending on the electronic spin.
2. Carbenes undergo insertion reactions into X-H and C-C bonds. They also add across double bonds, with singlet carbenes preserving alkene stereochemistry and triplet carbenes losing it.
3. Carbenes are generated by reactions such as α-elimination of halogenated compounds with base or decomposition of diazo compounds. They can rearrange through migrations such as the Wolff or Arndt-Eistert reactions.
Unsaturated Carbonyl Compound

1) α,β-Unsaturated carbonyl compounds contain a carbonyl group and a conjugated carbon-carbon double bond separated by one carbon-carbon single bond.
2) These compounds undergo both electrophilic and nucleophilic addition reactions due to the conjugation between the carbonyl and double bond.
3) Common reactions include the Michael addition, in which a carbanion adds to the β-carbon, and the Diels-Alder reaction, where a conjugated diene adds to form a six-membered ring.
Aromaticity and Anti-Aromaticity 

1) The document summarizes aromaticity and anti-aromaticity, discussing the key criteria for aromatic compounds including being cyclic, conjugated, planar, and having 4n+2 π electrons.
2) It provides examples of aromatic compounds like benzene and non-aromatic compounds that do not meet the criteria. Anti-aromatic compounds are also discussed.
3) NMR spectroscopy is discussed as a way to distinguish aromatic protons from other types of protons based on chemical shift values.
Reaction mechanisms

This document discusses organic reactions and mechanisms. It defines key terms like substrate, reagent, products, and mechanism. It describes how factors like inductive and mesomeric effects can influence reactions by altering electron density. It also discusses different types of reaction intermediates that can form, such as carbonium ions, carbanions, free radicals, and carbenes. The document classifies reagents as electrophiles or nucleophiles and describes their behaviors. It explains concepts like activation energy and the transition state that systems must go through for a reaction to occur.
Hybridization- sp, sp2 and sp3

Hybridization is the idea that atomic orbitals fuse to form newly hybridized orbitals, which in turn, influences molecular geometry and bonding properties. Hybridization is also an expansion of the valence bond theory.
Carbocation ppt

An organic species which has a carbon atom bearing only six electrons in its outermost shell and has a positive charge is called carbocation.
The positively charged carbon of carbocation is sp2 hybridized.
The unhybridized p-orbital remains vacant.
They are highly reactive and act as reaction intermediate.
They are also called carbonium ion.
Inductive Effect

Inductive Effect is the important topic in organic chemistry that gives us idea about electron withdrawing and accepting module of the specific group that determines the reactivity of the molecule or compound.
Electronic effects inductive,mesomeric,electromeric

This document discusses different electronic effects in organic chemistry. It describes inductive effect as a permanent polarization of electron density between two unlike atoms in a bond. Electron-withdrawing and electron-releasing groups are discussed. Mesomeric effect allows for resonance stabilization through delocalization of charge in conjugated systems. Electromeric effect involves the temporary shift of a pi bond electron pair to one atom upon reaction with an electrophile. The order of effects from strongest to weakest is given as mesomeric, electromeric, and inductive. Applications to stability of ions and carbocations are outlined.
Reaction intermediates

Reaction intermediates such as carbocation, carbanion, free radicals, carbene, nitrene and ylides were discussed.
Carbanion organic ppt

This document discusses carbanions, which are negatively charged organic species where carbon carries three bond pairs and one lone pair. Carbanions are stabilized through conjugation, resonance effects, field effects, and aromaticity. They are generated through heterolytic bond cleavage or addition of a negative ion to a carbon-carbon multiple bond. As nucleophiles, carbanions undergo reactions such as alpha-halogenation of ketones, additions to carbonyls, nucleophilic acyl substitutions, substitutions with alkyl halides, and Michael additions.
Resonance ,inductive effect -----(Organic )

Resonance occurs when a molecule can be represented by multiple Lewis structures and the actual structure is a hybrid of all the contributing structures. A resonance hybrid is the average structure between all the canonical or contributing Lewis structures. Delocalization of electrons through resonance leads to decreased potential energy and increased stability compared to any single contributing structure. Electron donating groups cause positive resonance or inductive effects while electron withdrawing groups cause negative resonance or inductive effects. Resonance and inductive effects influence acidity, basicity, and reactivity.
Basic effects in Organic chemistry

Basic effects in Organic chemistrySri Ramakrishna Mission Vidyalaya College of Arts and Science,Coimbatore-20.
This presentaion describes about the basic principle effects in organic chemistry like inductive,mesomeric,electromeric, resonance and hyperconjugation. this presentation contains some JAM competitive questions.Mesomeric Effect

Mesomeric (or resonance) effect refers to the polarity produced in a molecule through delocalization of pi electrons between bonds or a bond and lone pair. This effect can increase or decrease electron density in different parts of a molecule. Negative mesomeric effects are shown by groups like nitro (-NO2) that withdraw electron density, while positive effects are shown by groups like hydroxyl (-OH) that release electron density. Understanding mesomeric effects helps explain a molecule's reactivity toward electrophiles and nucleophiles by determining where electron density is increased or decreased.
Inductive effect pptx

This document discusses electronic displacement in organic compounds. It describes two types of electronic displacement: permanent displacement including inductive, resonance, and mesomeric effects; and temporary displacement through electromeric effects. Inductive effects are further broken down into +I effects where groups donate electron density and -I effects where groups withdraw electron density. Examples of inductive effects include their impact on acid/base strength, stability of carbocations/carbanions, and dipole moments.
Conformational analysis of cyclohexane

Cyclohexane exists in different conformations viz chair, boat, twist boat and half chair. These conformations possess different energies. Therefore they differ in energy.
Topicity

Homomorphic ligands / groups/ atoms, Homotopic, heterotopic, enantiotopic, prochirality and diastereotopic ligands and faces with examples
Resonance----(Oganic)

Resonance structures represent different arrangements of electrons in a molecule that have the same positions of nuclei but different bonding patterns. Resonance contributes to the stability of molecules like benzene by delocalizing electrons across multiple equivalent structures. The actual structure of a molecule represented by resonance is a hybrid of the contributing structures, with bond lengths intermediate between single and double bonds. Delocalization of electrons is depicted using curved arrows between resonance structures.
Resonance

Resonance effect occurs when a compound can be represented by two or more Lewis structures with the same arrangement of atoms. These contributing structures are called resonance structures.
The resonance structures are hypothetical and do not represent real molecules individually. Together they contribute to a resonance hybrid structure that is more stable than any individual resonance structure. The most stable contributing structure contributes the most to the resonance hybrid.
Conditions for resonance include sp or sp2 hybridized atoms involved, overlapping parallel p-orbitals to form a conjugated system. The sigma bond framework remains the same between structures, atomic positions do not change, and electron counts are equal.
What's hot (20)
Electronic effects inductive,mesomeric,electromeric

Electronic effects inductive,mesomeric,electromeric
Viewers also liked
Inductive effect

This document discusses inductive effect, which is the permanent displacement of a carbon chain's shared electron pair towards the more electronegative atom or group. It describes the negative inductive effect caused by electron-withdrawing groups and the positive inductive effect of electron-donating groups. Finally, it explains how inductive effect influences the strength of organic acids and bases, with acid strength decreasing as positive inductive effect or negative inductive effect decreases.
IR spectroscopy

Infrared spectroscopy involves using infrared radiation to analyze materials. Molecules absorb specific infrared frequencies that are characteristic of their structure, such as bond vibrations and stretches. There are two main methods for infrared spectroscopy - scanning monochromator which analyzes one wavelength at a time, and Fourier transform infrared spectroscopy which uses interferometry to measure all infrared wavelengths simultaneously. Fourier transform then converts this raw interferogram data into the infrared spectrum. Infrared spectroscopy can be used to identify functional groups and molecular structures in compounds like 1-Hexene, Toluene, and Cyclohexanol based on their characteristic absorption peaks.
Anomeric effect

The anomeric effect was discovered in 1955 with the work of J.T. Edward, N.-J. Chu, and R.U. Lemieux.
Contributed by: Cody F. Bender, Charles E. Price (Undergraduates), University of Utah, 2016
Investigation of one compound effect on main neuronal properties in a native ...

The document reports on an investigation of the effects of Compound A on neuronal properties in rat hippocampal slices. The study evaluated the effects of Compound A on input/output properties, paired-pulse facilitation, basal synaptic transmission, long-term potentiation, long-term depression, and paired-pulse inhibition. The results showed that Compound A slightly decreased basal synaptic transmission but did not modify other properties like paired-pulse facilitation or long-term potentiation and depression. The conclusion is that Compound A does not significantly affect the main neuronal properties evaluated in the study.
Steric Effects on the Configuration of the Nitrogen In Piperidine

The document discusses steric effects on the configuration of nitrogen in piperidine. Specifically, it summarizes that:
1) The nitrogen proton in piperidine prefers the axial conformation due to an attractive interaction between the NH and CH groups, rather than solely repulsive steric interactions.
2) Piperidine exists as two conformational isomers in equilibrium.
3) NMR spectroscopy can be used to measure the conformation of the nitrogen proton, with axial protons giving larger coupling constants.
Optically active compounds and their effect on and importance in biological s...

The document discusses the benefits of exercise for mental health. Regular physical activity can help reduce anxiety and depression and improve mood and cognitive function. Exercise causes chemical changes in the brain that may help protect against mental illness and improve symptoms.
B.tech. ii engineering chemistry unit 4 B organic chemistry

Organic reactions and their mechanisms are described. Key topics covered include nucleophiles and electrophiles, reaction types (addition, elimination, substitution), and organic intermediates. Electron displacement effects such as inductive, mesomeric, electromeric and inductometric effects are also discussed. Common organic reactions like nitration, halogenation and nucleophilic aromatic substitution are summarized.
Resoning

This document contains 60 questions related to organic chemistry concepts like alcohols, phenols, ethers, aldehydes, ketones, carboxylic acids, amines, etc. The questions cover topics like relative acidities, reactivities, mechanisms of reactions, effect of substituents, physical properties and their relationship to molecular structure. Answers provided explain the concepts in brief.
Hydrogen bonding 

Hydrogen bonding occurs between polar molecules where a hydrogen atom is bonded to a highly electronegative atom like oxygen or nitrogen. This partial positive charge on the hydrogen allows it to form an attractive force with another partially negatively charged atom. Water exhibits strong hydrogen bonding between its molecules, giving rise to properties like its high boiling point. Ethanol also exhibits hydrogen bonding between its -OH groups, allowing it to have a higher boiling point than similar sized molecules without this bonding, like methoxymethane. This hydrogen bonding also allows ethanol to be soluble in water whereas non-polar molecules cannot disrupt the water's hydrogen bonding network.
Aromaticity

This document discusses aromatic compounds and Hückel's rule for aromaticity. It defines aromatic compounds as cyclic, planar and fully conjugated compounds that have 4n + 2 π electrons according to Hückel's rule. These compounds are highly stable due to delocalization of π electrons over the whole ring. They undergo substitution rather than addition reactions and have intermediate bond lengths and diatropic NMR properties. Anti-aromatic compounds have 4n π electrons and show the opposite NMR characteristics. Molecular orbital theory is used to explain the stability and properties of aromatic compounds.
Adaptive Resonance Theory

This presentation gives brief overview of adaptive resonance theory, it's structure, working and uses.
Resonance

This document discusses the concept of resonance in organic chemistry. Resonance describes when the bonding in a molecule cannot be accurately represented by a single Lewis structure, and instead requires multiple structures that depict the delocalized electrons. Ozone is provided as an example where two resonance structures are needed to represent its true structure with equal bond lengths and partial charges on the outer oxygens. Benzene is also described as having two resonance structures, and its hexagonal structure with a circle inside represents the delocalized pi electrons in the ring.
Isomerism and its types

Organic compounds can exist as isomers - compounds with the same molecular formula but different structural formulas or spatial arrangements. There are two main types of isomerism: structural isomerism and stereoisomerism. Structural isomerism includes six sub-types based on differences in carbon chain structure, functional groups, or ring formations. Stereoisomerism involves two sub-types, geometric isomers which differ in spatial arrangements around double bonds, and optical isomers which are non-superimposable mirror images called enantiomers.
ORGANIC CHEMISTRY REASONING QUESTIONS

This document discusses various concepts in organic chemistry related to alcohols, phenols, ethers, aldehydes, ketones, carboxylic acids, amines and aromatic compounds. Some key points covered include:
1. The bond angle in alcohols is less than tetrahedral due to repulsion between oxygen's lone pairs. Branched alcohols have lower boiling points than straight-chain alcohols due to decreased intermolecular forces.
2. Phenol is a stronger acid than alcohols due to resonance stabilization of the phenoxide ion. Ethers have lower boiling points than alcohols due to the absence of hydrogen bonding.
3.
Organic reaction mechanism

The document discusses different types of substitution reactions including nucleophilic substitution, electrophilic substitution, and free radical substitution. It provides details on the mechanisms, kinetics, stereochemistry and factors affecting the rate of nucleophilic substitution reactions SN1 and SN2. SN1 follows a unimolecular mechanism involving a carbocation intermediate while SN2 follows a bimolecular mechanism with a single concerted transition state. The document also discusses electrophilic aromatic substitution reactions and addition and elimination reactions of alkenes and alkynes.
Resonance presentation

The document discusses the concept of resonance through examples such as bridges and mechanical systems. It explains that resonance occurs when an object vibrates at greater amplitudes due to another source emitting its natural frequency. Specifically, it discusses how the Tacoma Narrows Bridge collapsed in 1940 due to wind-induced oscillations resonating with the structure of the bridge. It also summarizes the collapse of the Nimitz Freeway in 1989, which was caused by an earthquake matching the bridge's resonant frequency. The document provides background on concepts like kinetic energy, potential energy, amplitude, and frequency to explain the physics of resonance.
Hydrogen bonding

1. Hydrogen bonding occurs between hydrogen atoms attached to electronegative atoms like oxygen, fluorine, and nitrogen of one molecule and an electronegative atom of another molecule.
2. Water is able to form extensive hydrogen bonding networks between molecules due to each water molecule having two hydrogen atoms and two lone pairs of electrons on the oxygen atom.
3. The hydrogen bonding network in liquid water is responsible for its unique properties, while hydrogen bonding in ice forms its crystalline lattice structure.
Viewers also liked (18)
Investigation of one compound effect on main neuronal properties in a native ...

Investigation of one compound effect on main neuronal properties in a native ...
Steric Effects on the Configuration of the Nitrogen In Piperidine

Steric Effects on the Configuration of the Nitrogen In Piperidine
Optically active compounds and their effect on and importance in biological s...

Optically active compounds and their effect on and importance in biological s...
B.tech. ii engineering chemistry unit 4 B organic chemistry

B.tech. ii engineering chemistry unit 4 B organic chemistry
Similar to Hyperconjugation
Conjugation & hyperconjugation (1).pptx

This document discusses conjugation, hyperconjugation, and cross-conjugation. Conjugation involves the overlap of p-orbitals across sigma bonds, resulting in alternating single and multiple bonds that increase stability. Hyperconjugation stabilizes molecules through interactions between sigma bonds and empty or partially filled orbitals. It increases stability in carbocations, radicals, and alkenes. Cross-conjugation involves the exclusion of one pi bond from conjugative interactions. Hyperconjugation disperses charge and stabilizes molecules. More substituted systems experience greater hyperconjugation and stability.
Electron Displacement Effect

This document summarizes different types of electron displacement effects including permanent polarization effects like inductive effect, mesomeric effect, and hyperconjugation, as well as temporary polarization effects like electromeric effect. It describes the inductive effect as the displacement of electron density in a covalent bond towards the more electronegative atom. The mesomeric effect involves resonance stabilization of molecules through pi electron delocalization. Hyperconjugation refers to sigma electron delocalization between C-H bonds and adjacent pi systems. The electromeric effect temporarily polarizes multiple bonds during reaction with electrophiles.
Electron displacement effect

This document discusses various types of electron displacement effects that can occur in covalent bonds, including permanent polarization effects like inductive effect, mesomeric effect, and hyperconjugation, as well as temporary polarization like electromeric effect. It provides examples and explanations of each type of effect, such as how inductive effect involves the displacement of electrons towards the more electronegative atom in a bond. It also discusses applications of these effects, such as explaining relative stabilities of carbocations and carbanions.
organic chemistry leacture 01 uol.pptx

This document discusses various types of organic reaction intermediates. It explains that reaction intermediates are transient chemical species that are formed in one step of a reaction mechanism and consumed in a subsequent step. Common types of intermediates discussed include radicals, carbocations, and carbanions. The document compares the stability of primary, secondary, and tertiary carbocations and carbanions based on factors like inductive effects, hybridization, and resonance. It also provides examples and structures of different organic reaction intermediates.
isotope effect and fragmantation

Isotopes are two atoms of the same element that have the same number of protons but different numbers of neutrons. Isotopes are specified by the mass number.
Unit -III Chemical bonding.pptx

Chemical Bonding
Ionic bonds form when electrons are transferred from a metal to a nonmetal, creating oppositely charged ions. Covalent bonds form when atoms share electrons. Ionic bonds are generally stronger than covalent bonds. The strength of an ionic bond depends on the charge and size of the ions - higher charges and smaller sizes result in stronger bonds. Ionic solids form crystalline structures with repeating patterns of ions. Lattice energy is required to separate ions in an ionic solid into gaseous ions.
ORGANIC Reaction mechanism ncert class 11

1. Organic reaction mechanisms involve the reaction of a substrate with a reagent, forming intermediates and ultimately products.
2. Bond cleavage can occur through either a heterolytic or homolytic process. Heterolytic cleavage leads to the formation of ions while homolytic cleavage leads to free radicals.
3. Electrophiles are electron seeking species that attack nucleophilic centers, while nucleophiles are electron pairing species that attack electrophilic centers. Common electrophiles include carbocations and carbonyl groups, while common nucleophiles include carbanions.
Reaction.pptx

This document discusses structural theory in organic chemistry. It covers topics like bond fission and its types (homolysis and heterolysis), organic reagents (electrophiles, nucleophiles, free radicals), types of organic reactions (substitution, elimination, addition, rearrangement), inductive effect and its applications, and mesomeric effect and its applications. Examples are provided to illustrate key concepts. Essay and short answer questions are also included at the end to test understanding of topics covered.
Introduction to Foundation of Chemistry 1

This document provides an introduction to foundational concepts in organic chemistry. It begins with learning outcomes focusing on orbitals, bonding structures, and the periodic table. It then reviews electron configuration, atomic structure including shells and subshells. The document discusses hybridization and molecular shapes for sp, sp2, and sp3 including examples. It introduces ionic and covalent bonding, and how atoms bond to attain stable electron configurations. Key concepts are defined such as line angle formulas, Hund's rule, and octet rule. Exercises are provided to identify bonding types and draw Lewis structures.
chapter-alkene.ppt

1) Alkenes are hydrocarbons containing a carbon-carbon double bond. They include many naturally occurring compounds like flavors and fragrances.
2) This chapter focuses on the general reaction of alkenes, which is electrophilic addition. It examines the consequences of alkene stereoisomerism and how double bonds are present in most organic molecules.
3) Electrophilic addition of alkenes involves the attack of an electrophile like HBr on the pi bond, forming a carbocation intermediate that then reacts with the bromide ion. This two-step process allows preparations using HCl or HI as well.
ch1_Basic_concepts_Organic_sem1.ppt

This document discusses various concepts related to organic chemistry including carbocations, carbocation stability, inductive effects, mesomeric effects, hyperconjugation, conjugation, and resonance. It explains that carbocations are carbon-containing species with a positive formal charge that are classified based on the number of carbons bonded to the charged carbon. Tertiary carbocations are the most stable due to effects such as hyperconjugation and inductive/mesomeric effects of alkyl groups that stabilize the positive charge. Inductive and mesomeric effects involve polarization of bonds that can stabilize or destabilize carbocations. Hyperconjugation and conjugation further contribute to stability through resonance structures.
Bonding by rawat sir jfc

1. The document discusses various types of chemical bonds including ionic bonds, covalent bonds, and hydrogen bonds. It describes how these bonds are formed and the factors that influence their formation.
2. Covalent bonds can be single, double, or triple depending on how many electron pairs are shared between atoms. Hybridization of atomic orbitals also influences molecular geometry and structure.
3. The Valence Shell Electron Pair Repulsion (VSEPR) theory is described as predicting the shapes of molecules based on hybridization and the repulsions between bonding pairs and lone pairs in the valence shell. Molecular shapes can deviate from the predicted geometry due to lone pair - bonding pair repulsions.
Presentation1 ADVANCED ORGANIC CHEMISTRY_.pptx

This document discusses several key concepts in chemical bonding and molecular structure including:
1) Hybridization which involves combining atomic orbitals to form new hybrid orbitals to explain bonding in molecules containing second-row elements like carbon, nitrogen, and oxygen.
2) Electronegativity and polarity which describes an atom's ability to attract electrons in a bond, with more electronegative elements taking on partial negative charges.
3) Hard-soft acid-base theory which predicts that hard acids prefer hard bases and soft acids prefer soft bases based on factors like size, charge, and polarizability.
Molecular orbital theory (conjugated molecules)

Bonding and Antibonding interactions; Idea about σ, σ*, π, π *, n – MOs; HOMO, LUMO and SOMO; Energy levels of π MOs of different conjugated acyclic and cyclic systems; Hückel’s rules for aromaticity; Frost diagram
molecularorbital theory pptx

1) The document discusses molecular orbital theory and how it applies to conjugated systems, including acyclic and cyclic systems. It outlines the molecular orbitals formed from p orbitals and how their energies depend on factors like conjugation and aromaticity.
2) Benzene is discussed as an example of an aromatic system, where the pi molecular orbitals are degenerate and fully occupied, conferring stability. Cyclobutadiene is an antiaromatic example where the highest occupied orbitals are non-bonding.
3) The 4n+2 Hückel rule for aromaticity is described, and examples of aromatic and antiaromatic compounds are given based on their pi electron counts. Ann
UV-Vis molecular absorption spectroscopy- BSc-Lect 5.pdf

1. UV-Vis spectroscopy detects electronic transitions in molecules when photons are absorbed, promoting electrons to higher energy states. Transitions involve π or n electrons and occur in the 200-700nm region.
2. Absorption depends on functional groups called chromophores as well as conjugation, solvent effects, and molecular structure. Selection rules govern allowed transitions.
3. Spectra appear as bands representing many overlapping transitions between vibrational/rotational energy levels of ground and excited electronic states. Band features provide structural information.
ELECTRONEGATIVITY

Pauling was the first to propose a scale of electronegativity in 1932 based on the difference in the measured energy of an AB bond and the expected energy of a purely covalent AB bond. Mulliken suggested an approach to electronegativity in 1934 based on ionization enthalpy and electron affinity, defining electronegativity as the arithmetic mean of ionization energy and electron affinity. Electronegativity is influenced by factors such as charge on the atom, hybridization state, ionization energy, electron affinity, and effective nuclear charge. It is used to determine bond polarity, percent ionic character, and enthalpy of formation.
Therotical Organic Chemistry.pptx

This document discusses structural theory in organic chemistry. It covers topics like bond fission and its types (homolysis and heterolysis), organic reagents (electrophiles, nucleophiles, free radicals), types of organic reactions (substitution, elimination, addition, rearrangement), inductive effect and its applications, and mesomeric effect and its applications. Bond fission can occur through homolysis, where the electron pair is split equally forming neutral radicals, or heterolysis where one atom takes both electrons forming charged species like carbocations or carbanions. The document defines different organic reagents and reactions. It also explains inductive and mesomeric effects which influence molecular stability and reactivity in organic compounds.
Introduction to electrochemistry 2 by t. hara

This document provides an overview of electrochemistry concepts including:
1. Electrochemistry involves redox reactions where electrons are gained or lost. Reactions occur at electrode interfaces in electrolytes.
2. Thermodynamics and kinetics govern electrochemical reactions. Thermodynamics is based on potential differences and kinetics considers charge/mass transfer limitations.
3. Experimental methods like measuring cell potentials at different concentrations are used to study electrochemical principles and reactions.
Introduction to electrochemistry 2 by t. hara

This document provides an overview of electrochemistry concepts including:
1. Electrochemistry involves redox reactions where electrons are gained or lost at electrode interfaces.
2. Thermodynamics and kinetics control redox reactions based on potential differences and charge/mass transfer limitations.
3. The electric double layer forms at electrode interfaces and can be modeled by the Helmholtz and Stern models.
Similar to Hyperconjugation (20)
UV-Vis molecular absorption spectroscopy- BSc-Lect 5.pdf

UV-Vis molecular absorption spectroscopy- BSc-Lect 5.pdf
More from Daniel Morton
CH Functionalizations on Electron-Deficient Aromatics in the synthesis of Pi-...

This document discusses the use of C-H functionalization catalysis to incorporate electron-deficient moieties into conjugated materials without needing to first install reactive halides or stannanes. Electron-deficient moieties are important for electronic materials as they can tune optical properties and transport characteristics. Direct C-H functionalization avoids harsh halogenation steps and bypasses instability issues of stannanes. Examples are given of one-pot syntheses using this approach to obtain up to 96% yields of differentially substituted electron-accepting building blocks. Challenges include controlling reaction selectivity when multiple C-H bonds on a moiety could potentially react.
C–H bond hydroxylation at non heme carboxylate-bridged diiron centers

This unit provides an overview of how Nature has inspired the development of novel diiron bridged complexes for use in C–H Hydroxylation.
Contributed by Omar Villanueva and Cora MacBeth, Emory University, 2014
Collagen triple helix stability

Collagen is made up of the repeating pattern Glycine-X-Y, where X and Y are commonly L-proline (Pro) and 4(R)-hydroxy-l-proline (Hyp), respectively. By substituting X and Y with a fluorine probe, stereoelectronic effects can be observed and compared to the effects of hydrogen bonding which has been predicted to be the main contributor to the collagen triple helix strength.
Contributed by: Alexandra Zudova, Samuel Broadbent (Undergraduates), University of Utah, 2013
Bio inspired metal-oxo catalysts for c–h bond functionalization

Metal-oxo complexes are important species in the activation of strong C–H bonds in biological systems. The high reactivity of metal-oxo complexes results from the way their valence electrons are arranged, and this arrangement depends strongly on the geometry around the metal center.
Contributed by: A.S. Borovik and Sarah Cook, University of California-Irvine, 2014
Fischer and schrock carbenes

A carbene is any neutral carbon species which contains a non-bonding valance pair of electrons.
Contributed by Alison Brown & Nathan Buehler, Undergraduates, University of Utah
Hammett parameters

The document discusses the Hammett plot, which is a linear free-energy relationship analysis used to model the electronic effects of substituents on aromatic systems. It describes how σ values are derived from ionization reactions to indicate whether a substituent is electron-donating or electron-withdrawing. These σ values can then be used to analyze reaction mechanisms and optimize reaction conditions for similar processes. Examples are given of how split Hammett plots reveal changing mechanisms depending on the electronic nature of the substituent. Problems involving interpreting ρ values in Hammett plots to determine reaction pathways are also presented.
Aryl fluorination

Organic Pedagogical Electronic Network
Aryl Fluorination
This document summarizes aryl fluorination, an important reaction for introducing fluorine groups onto aromatic rings. It notes that two of the top 10 grossing drugs, Lipitor and Reserdal, contain aryl fluorines. The mechanism proceeds through oxidation of an aryl group to a high valent metal species, followed by transmetalation and reductive elimination to introduce the fluorine. Examples are given of stoichiometric and catalytic aryl fluorination reactions using Selectfluor and silver oxide catalysts.
Allylic strain

The allylic position is the atom bound to a double bonded atom. The substituents on the allylic carbon and the doubly bonded atoms can result in allylic strain.
Contributed by: Sophia Robinson, (Undergraduate), Physical Organic Chemistry I, CHEM 7240 (Sigman), University of Utah, 2015
1 3-dipolar-cycloadditions

A 1,3-dipolar cycloaddition is a fascinating and diverse reaction that can be used in stepwise syntheses of large molecules, such as masked aldol reactions, and has potential biological applications. While it is an incredibly useful reactions, is also has a simple mechanism.
Contributed by: Created by Alexandra Kent and Judy Zhu (Undergraduates)Edited by Margaret Hilton
Honors Organic Chemistry
University of Utah
Heck redox relay

This document summarizes work on the Heck reaction and redox-relay Heck reaction. It provides background on the Heck reaction and its applications. It then discusses chain-walking in the Heck reaction where the palladium catalyst migrates down an alkyl chain, producing alkene isomers. It introduces Sigman's work developing a redox-relay Heck reaction where chain-walking of palladium is controlled by an alcohol thermodynamic sink on the substrate, transferring unsaturation to form aldehydes or ketones. It is authored by Margaret Hilton from the Sigman Lab at the University of Utah in 2014.
Stability and reactions of n heterocyclic carbenes

1) N-heterocyclic carbenes (NHCs) are stable two-coordinate carbon compounds that were first synthesized in 1991. Their stability is due to mesomeric and inductive effects from the nitrogen atoms that make the carbenes strong π-donors and weak σ-acceptors.
2) The thermodynamics of dissociation were studied for a bis(benzimidazol-2-ylidene), finding an enthalpy of 13.7 kcal/mol and entropy of 30.4 cal/mol*K. NHCs were also ranked based on their acidity.
3) NHCs form stable complexes with transition metals and have applications in organomet
Pyridine CH functionalization

This unit provides an overview of the different strategies that have been develop to selectively functionalize the 2,3 and 4 positions of pyridine units.
Contributed by the Sarpong Group, UC-Berkeley, 2013
Brønsted catalysis

The Brønsted catalysis relationship is a Linear Free Energy Relationship (LFER) that relates ionization of an acid or base which catalyzes a reaction and the rate of the reaction.
Contributed by: Quincy Davis, Jonathan Greenhalgh, Joshua Visser (Undergraduates), University of Utah, 2016
Quantum Tunneling in Organic Chemistry

Tunneling is a phenomenon of Quantum Mechanics in which particles, with a small amount of probability, are able to “tunnel” or travel through a large, finite potential energy (PE) barrier instead of traveling over the barrier as Classic Mechanics dictates should occur.
Contributed by: Mark O’Dair, Undergraduate, University of Utah, 2014
The Prins-Pinacol Reaction

The Prins-Pinacol reaction is a two-step process that begins with the Prins reaction, where an alkene attacks an aldehyde activated by a Lewis acid to form a cationic intermediate. This is followed by a pinacol rearrangement, where a methyl shift pushes the cation onto an oxygen. The reaction forms complex natural product backbones and allows stereoselective tetrahydropyran synthesis. Driving forces include increased stability of the carbocation intermediate and relief of ring strain. The Lewis acid activates the aldehyde for attack.
Solvation Effects on pKa Values

When substrates are put in solution, the solvent molecules can organize themselves around a charged species to stabilize it. Solvents can stabilize a charge most effectively when the charge on the substrate is easy to get to.
Contributed by: Jamie Allen, Jacqueline Pasek-Allen, Sarah Lefave (Undergraduates), University of Utah, 2016
Solvation effects on reactions

Solvation can be defined as any stabilizing interaction of a solute (or solute moiety) and the solvent. These interactions can be weak, purely electrostatic, as is the case with non-polar solutes and solvents, or more significant, involving the interactions between dipole moments or between dipoles and formal charges.
Contributed by: Anton S. Klimenko (Undergraduate), Department of Chemistry, The University of Utah, 2016
Hammett Plots in the World of Enzymes

A substituent effect is the change in a molecule’s reactivity when a substituent on the molecule is changed. In 1935, Louis Hammett designed a scale to measure influence of various substituents (X) at the meta- or para- positions on the acidity of benzoic acid.
Contributed by: Erika Aoyama and Megan Browning, University of Utah, 2016
Marcus theory

An overview of the use of the Marcus Theory to calculate the energies of transition states.
Contributed by: Elizabeth Greenhalgh, Amanda Bischoff, and Matthew Sigman, University of Utah, 2015
Inductive effects

This document discusses inductive effects in organic chemistry. It begins by defining inductive effects as the withdrawal of electrons from a σ bond to the more electronegative atom or group, creating a dipole. This effect can alter the reactivity of other groups on the molecule. More electronegative substituents increase the acidity of carboxylic acids by withdrawing electron density from the acidic proton. The document also examines how inductive effects influence aromatic systems' affinity for carbon dioxide, with methyl groups increasing and halogen groups decreasing this affinity through their respective positive and negative inductive effects.
More from Daniel Morton (20)
CH Functionalizations on Electron-Deficient Aromatics in the synthesis of Pi-...

CH Functionalizations on Electron-Deficient Aromatics in the synthesis of Pi-...
C–H bond hydroxylation at non heme carboxylate-bridged diiron centers

C–H bond hydroxylation at non heme carboxylate-bridged diiron centers
Bio inspired metal-oxo catalysts for c–h bond functionalization

Bio inspired metal-oxo catalysts for c–h bond functionalization
Stability and reactions of n heterocyclic carbenes

Stability and reactions of n heterocyclic carbenes
Recently uploaded
Direct Seeded Rice - Climate Smart Agriculture

Direct Seeded Rice - Climate Smart AgricultureInternational Food Policy Research Institute- South Asia Office
PPT on Direct Seeded Rice presented at the three-day 'Training and Validation Workshop on Modules of Climate Smart Agriculture (CSA) Technologies in South Asia' workshop on April 22, 2024.
Randomised Optimisation Algorithms in DAPHNE

Slides from talk:
Aleš Zamuda: Randomised Optimisation Algorithms in DAPHNE .
Austrian-Slovenian HPC Meeting 2024 – ASHPC24, Seeblickhotel Grundlsee in Austria, 10–13 June 2024
https://ashpc.eu/
Phenomics assisted breeding in crop improvement

As the population is increasing and will reach about 9 billion upto 2050. Also due to climate change, it is difficult to meet the food requirement of such a large population. Facing the challenges presented by resource shortages, climate
change, and increasing global population, crop yield and quality need to be improved in a sustainable way over the coming decades. Genetic improvement by breeding is the best way to increase crop productivity. With the rapid progression of functional
genomics, an increasing number of crop genomes have been sequenced and dozens of genes influencing key agronomic traits have been identified. However, current genome sequence information has not been adequately exploited for understanding
the complex characteristics of multiple gene, owing to a lack of crop phenotypic data. Efficient, automatic, and accurate technologies and platforms that can capture phenotypic data that can
be linked to genomics information for crop improvement at all growth stages have become as important as genotyping. Thus,
high-throughput phenotyping has become the major bottleneck restricting crop breeding. Plant phenomics has been defined as the high-throughput, accurate acquisition and analysis of multi-dimensional phenotypes
during crop growing stages at the organism level, including the cell, tissue, organ, individual plant, plot, and field levels. With the rapid development of novel sensors, imaging technology,
and analysis methods, numerous infrastructure platforms have been developed for phenotyping.
Compexometric titration/Chelatorphy titration/chelating titration

Classification
Metal ion ion indicators
Masking and demasking reagents
Estimation of Magnisium sulphate
Calcium gluconate
Complexometric Titration/ chelatometry titration/chelating titration, introduction, Types-
1.Direct Titration
2.Back Titration
3.Replacement Titration
4.Indirect Titration
Masking agent, Demasking agents
formation of complex
comparition between masking and demasking agents,
Indicators/Metal ion indicators/ Metallochromic indicators/pM indicators,
Visual Technique,PM indicators (metallochromic), Indicators of pH, Redox Indicators
Instrumental Techniques-Photometry
Potentiometry
Miscellaneous methods.
Complex titration with EDTA.
THEMATIC APPERCEPTION TEST(TAT) cognitive abilities, creativity, and critic...

THEMATIC APPERCEPTION TEST(TAT) cognitive abilities, creativity, and critic...Abdul Wali Khan University Mardan,kP,Pakistan
hematic appreciation test is a psychological assessment tool used to measure an individual's appreciation and understanding of specific themes or topics. This test helps to evaluate an individual's ability to connect different ideas and concepts within a given theme, as well as their overall comprehension and interpretation skills. The results of the test can provide valuable insights into an individual's cognitive abilities, creativity, and critical thinking skillsThe binding of cosmological structures by massless topological defects

Assuming spherical symmetry and weak field, it is shown that if one solves the Poisson equation or the Einstein field
equations sourced by a topological defect, i.e. a singularity of a very specific form, the result is a localized gravitational
field capable of driving flat rotation (i.e. Keplerian circular orbits at a constant speed for all radii) of test masses on a thin
spherical shell without any underlying mass. Moreover, a large-scale structure which exploits this solution by assembling
concentrically a number of such topological defects can establish a flat stellar or galactic rotation curve, and can also deflect
light in the same manner as an equipotential (isothermal) sphere. Thus, the need for dark matter or modified gravity theory is
mitigated, at least in part.
SAR of Medicinal Chemistry 1st by dk.pdf

In this presentation include the prototype drug SAR on thus or with their examples .
Syllabus of Second Year B. Pharmacy
2019 PATTERN.
Authoring a personal GPT for your research and practice: How we created the Q...

Thematic analysis in qualitative research is a time-consuming and systematic task, typically done using teams. Team members must ground their activities on common understandings of the major concepts underlying the thematic analysis, and define criteria for its development. However, conceptual misunderstandings, equivocations, and lack of adherence to criteria are challenges to the quality and speed of this process. Given the distributed and uncertain nature of this process, we wondered if the tasks in thematic analysis could be supported by readily available artificial intelligence chatbots. Our early efforts point to potential benefits: not just saving time in the coding process but better adherence to criteria and grounding, by increasing triangulation between humans and artificial intelligence. This tutorial will provide a description and demonstration of the process we followed, as two academic researchers, to develop a custom ChatGPT to assist with qualitative coding in the thematic data analysis process of immersive learning accounts in a survey of the academic literature: QUAL-E Immersive Learning Thematic Analysis Helper. In the hands-on time, participants will try out QUAL-E and develop their ideas for their own qualitative coding ChatGPT. Participants that have the paid ChatGPT Plus subscription can create a draft of their assistants. The organizers will provide course materials and slide deck that participants will be able to utilize to continue development of their custom GPT. The paid subscription to ChatGPT Plus is not required to participate in this workshop, just for trying out personal GPTs during it.
Micronuclei test.M.sc.zoology.fisheries.

Current Ms word generated power point presentation covers major details about the micronuclei test. It's significance and assays to conduct it. It is used to detect the micronuclei formation inside the cells of nearly every multicellular organism. It's formation takes place during chromosomal sepration at metaphase.
Immersive Learning That Works: Research Grounding and Paths Forward

We will metaverse into the essence of immersive learning, into its three dimensions and conceptual models. This approach encompasses elements from teaching methodologies to social involvement, through organizational concerns and technologies. Challenging the perception of learning as knowledge transfer, we introduce a 'Uses, Practices & Strategies' model operationalized by the 'Immersive Learning Brain' and ‘Immersion Cube’ frameworks. This approach offers a comprehensive guide through the intricacies of immersive educational experiences and spotlighting research frontiers, along the immersion dimensions of system, narrative, and agency. Our discourse extends to stakeholders beyond the academic sphere, addressing the interests of technologists, instructional designers, and policymakers. We span various contexts, from formal education to organizational transformation to the new horizon of an AI-pervasive society. This keynote aims to unite the iLRN community in a collaborative journey towards a future where immersive learning research and practice coalesce, paving the way for innovative educational research and practice landscapes.
Thornton ESPP slides UK WW Network 4_6_24.pdf

ESPP presentation to EU Waste Water Network, 4th June 2024 “EU policies driving nutrient removal and recycling
and the revised UWWTD (Urban Waste Water Treatment Directive)”
Describing and Interpreting an Immersive Learning Case with the Immersion Cub...

Current descriptions of immersive learning cases are often difficult or impossible to compare. This is due to a myriad of different options on what details to include, which aspects are relevant, and on the descriptive approaches employed. Also, these aspects often combine very specific details with more general guidelines or indicate intents and rationales without clarifying their implementation. In this paper we provide a method to describe immersive learning cases that is structured to enable comparisons, yet flexible enough to allow researchers and practitioners to decide which aspects to include. This method leverages a taxonomy that classifies educational aspects at three levels (uses, practices, and strategies) and then utilizes two frameworks, the Immersive Learning Brain and the Immersion Cube, to enable a structured description and interpretation of immersive learning cases. The method is then demonstrated on a published immersive learning case on training for wind turbine maintenance using virtual reality. Applying the method results in a structured artifact, the Immersive Learning Case Sheet, that tags the case with its proximal uses, practices, and strategies, and refines the free text case description to ensure that matching details are included. This contribution is thus a case description method in support of future comparative research of immersive learning cases. We then discuss how the resulting description and interpretation can be leveraged to change immersion learning cases, by enriching them (considering low-effort changes or additions) or innovating (exploring more challenging avenues of transformation). The method holds significant promise to support better-grounded research in immersive learning.
Bob Reedy - Nitrate in Texas Groundwater.pdf

Presented at June 6-7 Texas Alliance of Groundwater Districts Business Meeting
Unlocking the mysteries of reproduction: Exploring fecundity and gonadosomati...

The pygmy halfbeak Dermogenys colletei, is known for its viviparous nature, this presents an intriguing case of relatively low fecundity, raising questions about potential compensatory reproductive strategies employed by this species. Our study delves into the examination of fecundity and the Gonadosomatic Index (GSI) in the Pygmy Halfbeak, D. colletei (Meisner, 2001), an intriguing viviparous fish indigenous to Sarawak, Borneo. We hypothesize that the Pygmy halfbeak, D. colletei, may exhibit unique reproductive adaptations to offset its low fecundity, thus enhancing its survival and fitness. To address this, we conducted a comprehensive study utilizing 28 mature female specimens of D. colletei, carefully measuring fecundity and GSI to shed light on the reproductive adaptations of this species. Our findings reveal that D. colletei indeed exhibits low fecundity, with a mean of 16.76 ± 2.01, and a mean GSI of 12.83 ± 1.27, providing crucial insights into the reproductive mechanisms at play in this species. These results underscore the existence of unique reproductive strategies in D. colletei, enabling its adaptation and persistence in Borneo's diverse aquatic ecosystems, and call for further ecological research to elucidate these mechanisms. This study lends to a better understanding of viviparous fish in Borneo and contributes to the broader field of aquatic ecology, enhancing our knowledge of species adaptations to unique ecological challenges.
Applied Science: Thermodynamics, Laws & Methodology.pdf

When I was asked to give a companion lecture in support of ‘The Philosophy of Science’ (https://shorturl.at/4pUXz) I decided not to walk through the detail of the many methodologies in order of use. Instead, I chose to employ a long standing, and ongoing, scientific development as an exemplar. And so, I chose the ever evolving story of Thermodynamics as a scientific investigation at its best.
Conducted over a period of >200 years, Thermodynamics R&D, and application, benefitted from the highest levels of professionalism, collaboration, and technical thoroughness. New layers of application, methodology, and practice were made possible by the progressive advance of technology. In turn, this has seen measurement and modelling accuracy continually improved at a micro and macro level.
Perhaps most importantly, Thermodynamics rapidly became a primary tool in the advance of applied science/engineering/technology, spanning micro-tech, to aerospace and cosmology. I can think of no better a story to illustrate the breadth of scientific methodologies and applications at their best.
Recently uploaded (20)
Compexometric titration/Chelatorphy titration/chelating titration

Compexometric titration/Chelatorphy titration/chelating titration
THEMATIC APPERCEPTION TEST(TAT) cognitive abilities, creativity, and critic...

THEMATIC APPERCEPTION TEST(TAT) cognitive abilities, creativity, and critic...
The binding of cosmological structures by massless topological defects

The binding of cosmological structures by massless topological defects
Authoring a personal GPT for your research and practice: How we created the Q...

Authoring a personal GPT for your research and practice: How we created the Q...
Immersive Learning That Works: Research Grounding and Paths Forward

Immersive Learning That Works: Research Grounding and Paths Forward
Describing and Interpreting an Immersive Learning Case with the Immersion Cub...

Describing and Interpreting an Immersive Learning Case with the Immersion Cub...
Unlocking the mysteries of reproduction: Exploring fecundity and gonadosomati...

Unlocking the mysteries of reproduction: Exploring fecundity and gonadosomati...
Applied Science: Thermodynamics, Laws & Methodology.pdf

Applied Science: Thermodynamics, Laws & Methodology.pdf
Hyperconjugation
- 1. Organic Pedagogical Electronic Network Hyperconjugation Samuel Redstone University of Utah
- 2. Hyperconjugation: Definitions 1. J. Chem. Soc., 1935, 1844-1847. Hyperconjugation is the donation of a sigma bond into an adjacent empty or partially filled p orbital, which results in an increased stability of the molecule. Hyperconjugation contributes to the resonance stabilization of this tertiary carbocation, where electrons from the C-H sigma bonding orbital are donated to the empty p orbital of the cation. Hyperconjugation was first described by Baker and Nathan in 1935 to describe the “abnormal behavior” of alkyl-substituted compounds.1
- 3. Trends in Hyperconjugation 1. Less electronegative atoms make for better sigma bond donors. This is because more electronegative atoms have lower lying HOMOs, which have poor overlap with the LUMO of the electron acceptor. > In other words, less electronegative atoms are more willing to share there electrons with the carbocation. Therefore, the 2-methyl butane is more stabilized through hyperconjugation than the 3-fluoro 2-methyl butane.
- 4. Trends in Hyperconjugation 3. The greater the number of substituents on a carbocation, the more stabilized it will be through hyperconjugation. 2. Lower lying LUMOs make for better electron acceptors, as there is better orbital overlap with the HOMO of the electron donor. > > >Atomic orbital sp π* sp2 π* sp3 π* > > Therefore, tertiary carbocations are more stable than secondary or primary.
- 5. Other types of Hyperconjugation Occasionally, electron density will be donated from a filled 𝜋 or p orbital into an adjacent σ* orbital. This is known as negative hyperconjugation. Other times, electron density will be donated from a p orbital into an adjacent 𝜋* orbital; the result is an overall neutral charge on the molecule. This is known as neutral hyperconjugation.
- 6. Consequences of Hyperconjugation 2. Chem. Phys. Lett., 1981, 84, 267. 1. Conformational Preferences of Esters (E) Conformer (Z) Conformer ΔG=+4.8 kcal/mol Esters exist in two conformers, E and Z. (E) Conformer (Z) Conformer Because the Z conformation allows for the overlap of the lone pair with the C—O σ* it is favored over the E conformation, where the lone pair is aligned with the C—R σ*.2 No resonance, poor orbital overlap!
- 7. Consequences of Hyperconjugation J. Chem. Phys., 1951, 19, 342. 2. The Secondary Kinetic Isotope Effect The kinetic isotope effect (KIE) is the change in rate of a reaction when an atom is replaced with a heavier isotope. A secondary KIE is when the isotope substitution occurs at an atom where a bond is not being broken or formed. Because heavier atoms form stronger bonds, they are less willing to contribute to hyperconjugation. Therefore, reactions that require the elimination of a leaving group proceed more slowly. For example: *Note: TsO, or tosylate, is a leaving group. 1. 2. Due to the weaker C—H bond, Reaction 1 proceeds faster than Reaction 2. This is because the C—H σ bond is more willing to push electrons into the empty orbital of the carbocation and contribute to hyperconjugation. KH/KD is the ratio of the rates of the individual reactions. A KH/KD >1 suggests that the reaction occurs more slowly with the isotopic substitution. KH/KD = 1.15, as found by Streitwieser, et al.3
- 8. Problems 1. Rank the ability of the following bonds to contribute to hyperconjugation: C—F, C—O, C—N, C—H. A. C—F > C—O > C—N > C—H B. C—H > C—N > C—O > C—F C. C—H > C—O > C—N > C—F D. C—F > C—H > C—N > C—O 2. The following molecule will not experience hyperconjugation. Why is that? A. The hybridization of the molecule does not permit proper orbital overlap. B. The deuterium does not contribute hyperconjugation very well. C. There is poor overlap between the LUMO of the N—Me bond and HOMO of the N—D bond. D. Hyperconjugation does not occur on heteroatoms such as nitrogen. 3. Which of the following most readily ionizes? A. B. C. D. 4. Identify whether the following molecules have positive, negative, or neutral hyperconjugation. i. A. Positive B. Negative C. Neutral ii. A. Positive B. Negative C. Neutral A. Positive B. Negative C. Neutral iii.
- 9. Solutions 1. B 2. C 3. A 4. i. B ii. C iii. A
- 10. This work is licensed under a Creative Commons Attribution- ShareAlike 4.0 International License. Contributed by: Samuel Redstone (Undergraduate) University of Utah, 2016