Marcus theory
•Download as PPTX, PDF•
8 likes•14,024 views
An overview of the use of the Marcus Theory to calculate the energies of transition states. Contributed by: Elizabeth Greenhalgh, Amanda Bischoff, and Matthew Sigman, University of Utah, 2015
Report
Share
Report
Share
Recommended
Electron Spin Resonance Spectroscopy 

ESR Spectra- Principle-Hyperfine Splitting and structure- g-factor - Factors affecting g-value - Anisotropic system- Triplet state - Kramer's degeneracy - Zero Field Splitting
Lanthanide shift reagents in nmr

Lanthanide Shift Reagents in Nuclear Magnetic Resonance Spectroscopy
REDOX REACTION : inner & outer sphere Complimentary & non-complimentary reaction

Introduction
Definition of Redox Reaction
Outer sphere mechanism
Inner sphere mechanism
Requirements of ISM
Complimentary reactions
Non-complimentary reaction
Evidence of Metal Ligand Bonding.pptx

This Presentation describes about the evidence of metal ligand bonding in a molecule. In this presentation various evidences are explained. Learn and grow.
NUCLEAR QUADRUPOLE RESONANCE SPECTROSCOPY 

NQR - DEFINITION - ELECTRIC FIELD GRADIENT - NUCLEAR QUADRUPOLE MOMENT - NUCLEAR QUADRUPOLE COUPLING CONSTANT - PRINCIPLE OF NQR - ENERGY OF INTERACTION - SELECTION RULE - FREQUENCY OF TRANSITION - APPLICATIONS
CONTACT SHIFT and SHIFT REAGENTS

It contains what are the shift reagents, and how they will use in NMR spectroscopy. It includes lanthanide shift reagents and their effect using NMR spectroscopy. It has mostly used shift reagents like Europium and their importance. paramagnetic species that affect the NMR spectra are also explained in detail. What are contact shift and pseudo-contact shift also explained. It contains what are the chiral shift reagent, and the advantages, and disadvantages of lanthanide shift reagents. Reference books are also included.
Recommended
Electron Spin Resonance Spectroscopy 

ESR Spectra- Principle-Hyperfine Splitting and structure- g-factor - Factors affecting g-value - Anisotropic system- Triplet state - Kramer's degeneracy - Zero Field Splitting
Lanthanide shift reagents in nmr

Lanthanide Shift Reagents in Nuclear Magnetic Resonance Spectroscopy
REDOX REACTION : inner & outer sphere Complimentary & non-complimentary reaction

Introduction
Definition of Redox Reaction
Outer sphere mechanism
Inner sphere mechanism
Requirements of ISM
Complimentary reactions
Non-complimentary reaction
Evidence of Metal Ligand Bonding.pptx

This Presentation describes about the evidence of metal ligand bonding in a molecule. In this presentation various evidences are explained. Learn and grow.
NUCLEAR QUADRUPOLE RESONANCE SPECTROSCOPY 

NQR - DEFINITION - ELECTRIC FIELD GRADIENT - NUCLEAR QUADRUPOLE MOMENT - NUCLEAR QUADRUPOLE COUPLING CONSTANT - PRINCIPLE OF NQR - ENERGY OF INTERACTION - SELECTION RULE - FREQUENCY OF TRANSITION - APPLICATIONS
CONTACT SHIFT and SHIFT REAGENTS

It contains what are the shift reagents, and how they will use in NMR spectroscopy. It includes lanthanide shift reagents and their effect using NMR spectroscopy. It has mostly used shift reagents like Europium and their importance. paramagnetic species that affect the NMR spectra are also explained in detail. What are contact shift and pseudo-contact shift also explained. It contains what are the chiral shift reagent, and the advantages, and disadvantages of lanthanide shift reagents. Reference books are also included.
Alpha axial haloketone rule and octant rule

This presentation discusses the use of alpha axial halo ketone rule and Octant rule for determination of absolute configuration
WALSH DIAGRAM- SEMINAR TOPIC- ABSTRACT

CONTENTS
INTRODUCTION
CONCEPTS OF WALSH DIAGRAM
APPLICATION IN TRIATOMIC MOLECULES
[IN AH₂ TYPE OF MOLECULES(BeH₂,BH₂,H₂O)]
INTRODUCTION
Arthur Donald Walsh FRS The introducer of walsh diagram (8 August 1916-23 April 1977) was a British chemist, professor of chemistry at the University of Dundee . He was elected FRS in 1964. He was educated at Loughborough Grammar School.
Walsh diagrams were first introduced in a series of ten papers in one issue of the Journal of the Chemical Society . Here, he aimed to rationalize the shapes adopted by polyatomic molecules in the ground state as well as in excited states, by applying theoretical contributions made by Mulliken .
Mossbauer spectroscopy - Principles and applications

Mossbauer spectroscopy an effective tool to study Mossbauer active nuclei.
Esr spectroscopy

For UG students of All Engineering Branches (Mechanical Engg., Chemical Engg., Instrumentation Engg., Food Technology) and PG students of Chemistry, Physics, Biochemistry, Pharmacy
The link of the video lecture at YouTube is
https://www.youtube.com/watch?v=t3QDG8ZIX-8
WALSH DIAGRAM- SEMINAR TOPIC

CONTENTS
INTRODUCTION
CONCEPTS OF WALSH DIAGRAM
APPLICATION IN TRIATOMIC MOLECULES
[IN AH₂ TYPE OF MOLECULES(BeH₂,BH₂,H₂O)]
INTRODUCTION
Arthur Donald Walsh FRS The introducer of walsh diagram (8 August 1916-23 April 1977) was a British chemist, professor of chemistry at the University of Dundee . He was elected FRS in 1964. He was educated at Loughborough Grammar School.
Walsh diagrams were first introduced in a series of ten papers in one issue of the Journal of the Chemical Society . Here, he aimed to rationalize the shapes adopted by polyatomic molecules in the ground state as well as in excited states, by applying theoretical contributions made by Mulliken .
Mossbauer spectroscopy

A complete Detail on Mossbauer spectroscopy which gives a broad idea about the topic
Lect. 23 rotational vibrational raman spectroscopy

rotational Raman spectrum vibrational Raman spectrum,
Electron Spin Resonance (ESR) Spectroscopy

Electron Spin Resonance Spectroscopy
Also called EPR Spectroscopy
Electron Paramagnetic Resonance Spectroscopy
Non-destructive technique
Applications
Extensively used in transition metal complexes
Deviated geometries in crystals
NMR Shift reagents

These are chemical shift reagents and solvent induced shifts have their application in resolving the NMR Spectra of complex structures by inducing shift with respect to reference compound. Thus useful in interpretation of structures of complex organic compounds.
Coexistence of Superconductivity and Itinerant Ferromagnetism in Ucoge

The coexistence of BCS superconductivity and itinerant ferromagnetism in uranium based intermetallic systems is analyzed using a Hubbard Hamiltonian. To obtain the superconducting transition
temperatureTC and Curie temperatureTFM , we used the Green’s function method. The order parameter of superconductivity ( ∆ ) and ferromagnetism ( m or I) are obtained in the mean field approximation. It is found that there generally exist coexistent solutions to coupled equations of the order parameter in the temperature range ( ) T TC TFM 0 < < min , . In our model, ferromagnetism is itinerant and therefore carried by the conduction electrons. This arises from a splitting of the spin-up and spin- down band. A consequence is that the ferromagnetism and superconductivity is carried by same electrons. Expressions for specific heat, energy spectra and density of states are derived. The specific heat has linear temperature dependence as opposed to that of the exponential decrease in the BCS theory. The density of states for a finite magnetic order parameter increases as opposed to that of a ferromagnetic metal. The theory is
applied to explain the observations in uranium based intermetallic compoundUCoGe . The agreement between theory and experiments is quite encouraging.
Coexistence of Superconductivity and Itinerant Ferromagnetism in Ucoge 

The coexistence of BCS superconductivity and itinerant ferromagnetism in uranium based intermetallic
systems is analyzed using a Hubbard Hamiltonian. To obtain the superconducting transition
temperatureTC
and Curie temperatureTFM , we used the Green’s function method. The order parameter of
superconductivity ( ∆ ) and ferromagnetism ( m or I) are obtained in the mean field approximation. It is
found that there generally exist coexistent solutions to coupled equations of the order parameter in the
temperature range ( ) T TC TFM 0 < < min , . In our model, ferromagnetism is itinerant and therefore
carried by the conduction electrons. This arises from a splitting of the spin-up and spin- down band. A
consequence is that the ferromagnetism and superconductivity is carried by same electrons. Expressions
for specific heat, energy spectra and density of states are derived. The specific heat has linear temperature
dependence as opposed to that of the exponential decrease in the BCS theory. The density of states for a
finite magnetic order parameter increases as opposed to that of a ferromagnetic metal. The theory is
applied to explain the observations in uranium based intermetallic compoundUCoGe . The agreement
between theory and experiments is quite encouraging.
More Related Content
What's hot
Alpha axial haloketone rule and octant rule

This presentation discusses the use of alpha axial halo ketone rule and Octant rule for determination of absolute configuration
WALSH DIAGRAM- SEMINAR TOPIC- ABSTRACT

CONTENTS
INTRODUCTION
CONCEPTS OF WALSH DIAGRAM
APPLICATION IN TRIATOMIC MOLECULES
[IN AH₂ TYPE OF MOLECULES(BeH₂,BH₂,H₂O)]
INTRODUCTION
Arthur Donald Walsh FRS The introducer of walsh diagram (8 August 1916-23 April 1977) was a British chemist, professor of chemistry at the University of Dundee . He was elected FRS in 1964. He was educated at Loughborough Grammar School.
Walsh diagrams were first introduced in a series of ten papers in one issue of the Journal of the Chemical Society . Here, he aimed to rationalize the shapes adopted by polyatomic molecules in the ground state as well as in excited states, by applying theoretical contributions made by Mulliken .
Mossbauer spectroscopy - Principles and applications

Mossbauer spectroscopy an effective tool to study Mossbauer active nuclei.
Esr spectroscopy

For UG students of All Engineering Branches (Mechanical Engg., Chemical Engg., Instrumentation Engg., Food Technology) and PG students of Chemistry, Physics, Biochemistry, Pharmacy
The link of the video lecture at YouTube is
https://www.youtube.com/watch?v=t3QDG8ZIX-8
WALSH DIAGRAM- SEMINAR TOPIC

CONTENTS
INTRODUCTION
CONCEPTS OF WALSH DIAGRAM
APPLICATION IN TRIATOMIC MOLECULES
[IN AH₂ TYPE OF MOLECULES(BeH₂,BH₂,H₂O)]
INTRODUCTION
Arthur Donald Walsh FRS The introducer of walsh diagram (8 August 1916-23 April 1977) was a British chemist, professor of chemistry at the University of Dundee . He was elected FRS in 1964. He was educated at Loughborough Grammar School.
Walsh diagrams were first introduced in a series of ten papers in one issue of the Journal of the Chemical Society . Here, he aimed to rationalize the shapes adopted by polyatomic molecules in the ground state as well as in excited states, by applying theoretical contributions made by Mulliken .
Mossbauer spectroscopy

A complete Detail on Mossbauer spectroscopy which gives a broad idea about the topic
Lect. 23 rotational vibrational raman spectroscopy

rotational Raman spectrum vibrational Raman spectrum,
Electron Spin Resonance (ESR) Spectroscopy

Electron Spin Resonance Spectroscopy
Also called EPR Spectroscopy
Electron Paramagnetic Resonance Spectroscopy
Non-destructive technique
Applications
Extensively used in transition metal complexes
Deviated geometries in crystals
NMR Shift reagents

These are chemical shift reagents and solvent induced shifts have their application in resolving the NMR Spectra of complex structures by inducing shift with respect to reference compound. Thus useful in interpretation of structures of complex organic compounds.
What's hot (20)
Mossbauer spectroscopy - Principles and applications

Mossbauer spectroscopy - Principles and applications
Lect. 23 rotational vibrational raman spectroscopy

Lect. 23 rotational vibrational raman spectroscopy
Similar to Marcus theory
Coexistence of Superconductivity and Itinerant Ferromagnetism in Ucoge

The coexistence of BCS superconductivity and itinerant ferromagnetism in uranium based intermetallic systems is analyzed using a Hubbard Hamiltonian. To obtain the superconducting transition
temperatureTC and Curie temperatureTFM , we used the Green’s function method. The order parameter of superconductivity ( ∆ ) and ferromagnetism ( m or I) are obtained in the mean field approximation. It is found that there generally exist coexistent solutions to coupled equations of the order parameter in the temperature range ( ) T TC TFM 0 < < min , . In our model, ferromagnetism is itinerant and therefore carried by the conduction electrons. This arises from a splitting of the spin-up and spin- down band. A consequence is that the ferromagnetism and superconductivity is carried by same electrons. Expressions for specific heat, energy spectra and density of states are derived. The specific heat has linear temperature dependence as opposed to that of the exponential decrease in the BCS theory. The density of states for a finite magnetic order parameter increases as opposed to that of a ferromagnetic metal. The theory is
applied to explain the observations in uranium based intermetallic compoundUCoGe . The agreement between theory and experiments is quite encouraging.
Coexistence of Superconductivity and Itinerant Ferromagnetism in Ucoge 

The coexistence of BCS superconductivity and itinerant ferromagnetism in uranium based intermetallic
systems is analyzed using a Hubbard Hamiltonian. To obtain the superconducting transition
temperatureTC
and Curie temperatureTFM , we used the Green’s function method. The order parameter of
superconductivity ( ∆ ) and ferromagnetism ( m or I) are obtained in the mean field approximation. It is
found that there generally exist coexistent solutions to coupled equations of the order parameter in the
temperature range ( ) T TC TFM 0 < < min , . In our model, ferromagnetism is itinerant and therefore
carried by the conduction electrons. This arises from a splitting of the spin-up and spin- down band. A
consequence is that the ferromagnetism and superconductivity is carried by same electrons. Expressions
for specific heat, energy spectra and density of states are derived. The specific heat has linear temperature
dependence as opposed to that of the exponential decrease in the BCS theory. The density of states for a
finite magnetic order parameter increases as opposed to that of a ferromagnetic metal. The theory is
applied to explain the observations in uranium based intermetallic compoundUCoGe . The agreement
between theory and experiments is quite encouraging.
ELECTRONIC STRUCTURE OF STRONGLY CORRELATED SYSTEMS

ELECTRONIC STRUCTURE OF
STRONGLY CORRELATED SYSTEMS
Oe3424742482

International Journal of Engineering Research and Applications (IJERA) is an open access online peer reviewed international journal that publishes research and review articles in the fields of Computer Science, Neural Networks, Electrical Engineering, Software Engineering, Information Technology, Mechanical Engineering, Chemical Engineering, Plastic Engineering, Food Technology, Textile Engineering, Nano Technology & science, Power Electronics, Electronics & Communication Engineering, Computational mathematics, Image processing, Civil Engineering, Structural Engineering, Environmental Engineering, VLSI Testing & Low Power VLSI Design etc.
Dielectric Dilemma 1901.10805 v2 feb 4 2019

A dielectric dilemma faces scientists because Maxwell's equations are poor approximations as usually written, with a single dielectric constant. Maxwell's equations are then not accurate enough to be useful in many applications. The dilemma can be partially resolved by a rederivation of conservation of current, where current is defined now to include the epolarization of the vacuumf ..0 .......... Conserveration of current becomes Kirchoff's current law with this definition, in the one dimensional circuits of our electronic technology. With this definition, Kirchoff's laws are valid whenever Maxwell's equations are valid, explaining why those laws reliably describe circuits that switch in nanoseconds.
Magnetic Materials Assignment Help

I am Baddie K. I am a Magnetic Materials Assignment Expert at eduassignmenthelp.com. I hold a Masters's Degree in Electro-Magnetics, from The University of Malaya, Malaysia. I have been helping students with their assignments for the past 12 years. I solve assignments related to Magnetic Materials.
Visit eduassignmenthelp.com or email info@eduassignmenthelp.com. You can also call on +1 678 648 4277 for any assistance with Magnetic Materials Assignments.
Superconductivity

Presentation on superconductivity by Phelim Bradley for Lab module in University College Cork
Dynamics of Twointeracting Electronsinthree-Dimensional Lattice

The physical property of strongly correlated electrons on a three-dimensional (3D) 3 x 3 x 3 cluster of the simple cubic lattice is here presented.In the work we developed the unit step Hamiltonian as a solution to the single band Hubbard Hamiltonian for the case of two electrons interaction in a finite three dimensional lattice. The approximation to the Hubbard Hamiltonian study is actually necessary because of the strong limitation and difficulty pose by the Hubbard Hamiltonian as we move away from finite - size lattices to larger N - dimensional lattices. Thus this work has provided a means of overcoming the finite - size lattice defects as we pass on to a higher dimension. We have shown in this study, that the repulsive Coulomb interaction which in part leads to the strong electronic correlations, would indicate that the two electron system prefer not to condense into s-wave superconducting singlet state (s = 0), at high positive values of the interaction strength. This study reveals that when the Coulomb interaction is zero, that is, for free electron system (non-interacting), thevariational parameters which describe the probability distribution of lattice electron system is the same. The spectra intensity for on-site electrons is zero for all values of the interaction strength
Similar to Marcus theory (20)
Coexistence of Superconductivity and Itinerant Ferromagnetism in Ucoge

Coexistence of Superconductivity and Itinerant Ferromagnetism in Ucoge
Coexistence of Superconductivity and Itinerant Ferromagnetism in Ucoge 

Coexistence of Superconductivity and Itinerant Ferromagnetism in Ucoge
ELECTRONIC STRUCTURE OF STRONGLY CORRELATED SYSTEMS

ELECTRONIC STRUCTURE OF STRONGLY CORRELATED SYSTEMS
UV-Vis molecular absorption spectroscopy- BSc-Lect 5.pdf

UV-Vis molecular absorption spectroscopy- BSc-Lect 5.pdf
Dynamics of Twointeracting Electronsinthree-Dimensional Lattice

Dynamics of Twointeracting Electronsinthree-Dimensional Lattice
More from Daniel Morton
CH Functionalizations on Electron-Deficient Aromatics in the synthesis of Pi-...

This module provides an overview of the techniques being developed to functionalize CH bonds on electron deficient aromatics for the preparation of Pi-conjugated materials.
Contributed by the Marder Group, 2014
C–H bond hydroxylation at non heme carboxylate-bridged diiron centers

This unit provides an overview of how Nature has inspired the development of novel diiron bridged complexes for use in C–H Hydroxylation.
Contributed by Omar Villanueva and Cora MacBeth, Emory University, 2014
Collagen triple helix stability

Collagen is made up of the repeating pattern Glycine-X-Y, where X and Y are commonly L-proline (Pro) and 4(R)-hydroxy-l-proline (Hyp), respectively. By substituting X and Y with a fluorine probe, stereoelectronic effects can be observed and compared to the effects of hydrogen bonding which has been predicted to be the main contributor to the collagen triple helix strength.
Contributed by: Alexandra Zudova, Samuel Broadbent (Undergraduates), University of Utah, 2013
Bio inspired metal-oxo catalysts for c–h bond functionalization

Metal-oxo complexes are important species in the activation of strong C–H bonds in biological systems. The high reactivity of metal-oxo complexes results from the way their valence electrons are arranged, and this arrangement depends strongly on the geometry around the metal center.
Contributed by: A.S. Borovik and Sarah Cook, University of California-Irvine, 2014
Fischer and schrock carbenes

A carbene is any neutral carbon species which contains a non-bonding valance pair of electrons.
Contributed by Alison Brown & Nathan Buehler, Undergraduates, University of Utah
Hammett parameters

The Hammett Plot is a type of Linear Free-Energy Relationship (LFER) analysis designed to model the electronic effect of substituents on aromatic systems (in the para and meta positions only). Information gathered can be used to probe the mechanism of the reaction and can be applied in the optimization of reaction conditions.
Contributed by: Eric Amerling & Christine Nervig (Undergraduates), University of Utah, 2013
Aryl fluorination

Aryl fluorines are present in two of the top 10 grossing drugs (Lipitor and Reserdal). The C-F bond is uniquely strong and prevents facile metabolism, giving these compounds longer T1/2 in vivo. The generalized mechanism for electrophilic aryl fluorination proceeds through a high valent metal species.
Contributed by: Zach Niemeyer, Justin Salvant (Undergraduates), University of Utah, 2013
Allylic strain

The allylic position is the atom bound to a double bonded atom. The substituents on the allylic carbon and the doubly bonded atoms can result in allylic strain.
Contributed by: Sophia Robinson, (Undergraduate), Physical Organic Chemistry I, CHEM 7240 (Sigman), University of Utah, 2015
1 3-dipolar-cycloadditions

A 1,3-dipolar cycloaddition is a fascinating and diverse reaction that can be used in stepwise syntheses of large molecules, such as masked aldol reactions, and has potential biological applications. While it is an incredibly useful reactions, is also has a simple mechanism.
Contributed by: Created by Alexandra Kent and Judy Zhu (Undergraduates)Edited by Margaret Hilton
Honors Organic Chemistry
University of Utah
Heck redox relay

Since Heck’s seminal publication in 1968, the Heck reaction has been a powerful synthetic tool for the functionalization of olefins with aryl groups.
Contributed by Margaret Hilton, Sigman Lab, University of Utah, 2014
Stability and reactions of n heterocyclic carbenes

Carbenes are two-coordinate carbon compounds with a sextet of electrons on the carbon. Most of them the indicate instability and high reactivity. Stable carbenes lead to new possibilities and fields. This overview introduces N-heterocyclic carbenes, a stable class of these unusual compounds.
Contributed by:
Özlem Urcan, F. Richard Kurth, Jonas Renner (Undergraduates), University of Utah, 2016
Pyridine CH functionalization

This unit provides an overview of the different strategies that have been develop to selectively functionalize the 2,3 and 4 positions of pyridine units.
Contributed by the Sarpong Group, UC-Berkeley, 2013
Brønsted catalysis

The Brønsted catalysis relationship is a Linear Free Energy Relationship (LFER) that relates ionization of an acid or base which catalyzes a reaction and the rate of the reaction.
Contributed by: Quincy Davis, Jonathan Greenhalgh, Joshua Visser (Undergraduates), University of Utah, 2016
Quantum Tunneling in Organic Chemistry

Tunneling is a phenomenon of Quantum Mechanics in which particles, with a small amount of probability, are able to “tunnel” or travel through a large, finite potential energy (PE) barrier instead of traveling over the barrier as Classic Mechanics dictates should occur.
Contributed by: Mark O’Dair, Undergraduate, University of Utah, 2014
The Prins-Pinacol Reaction

The Prins-Pinacol reaction is a two step process. It begins with the Prins reaction, which is the attack by a nucleophilic alkene on a Lewis acid-activated aldehyde. This forms a cationic intermediate. The pinacol rearrangement is a methyl shift which pushes the cation on to an oxygen, which is then deprotonated.
Contributed by: Andy Clevenger (Undergraduate), University of Utah, 2016
Solvation Effects on pKa Values

When substrates are put in solution, the solvent molecules can organize themselves around a charged species to stabilize it. Solvents can stabilize a charge most effectively when the charge on the substrate is easy to get to.
Contributed by: Jamie Allen, Jacqueline Pasek-Allen, Sarah Lefave (Undergraduates), University of Utah, 2016
Solvation effects on reactions

Solvation can be defined as any stabilizing interaction of a solute (or solute moiety) and the solvent. These interactions can be weak, purely electrostatic, as is the case with non-polar solutes and solvents, or more significant, involving the interactions between dipole moments or between dipoles and formal charges.
Contributed by: Anton S. Klimenko (Undergraduate), Department of Chemistry, The University of Utah, 2016
Hyperconjugation

Hyperconjugation is the donation of a sigma bond into an adjacent empty or partially filled p orbital, which results in an increased stability of the molecule.
Contributed by: Samuel Redstone (Undergraduate), University of Utah, 2016
Hammett Plots in the World of Enzymes

A substituent effect is the change in a molecule’s reactivity when a substituent on the molecule is changed. In 1935, Louis Hammett designed a scale to measure influence of various substituents (X) at the meta- or para- positions on the acidity of benzoic acid.
Contributed by: Erika Aoyama and Megan Browning, University of Utah, 2016
Anomeric effect

The anomeric effect was discovered in 1955 with the work of J.T. Edward, N.-J. Chu, and R.U. Lemieux.
Contributed by: Cody F. Bender, Charles E. Price (Undergraduates), University of Utah, 2016
More from Daniel Morton (20)
CH Functionalizations on Electron-Deficient Aromatics in the synthesis of Pi-...

CH Functionalizations on Electron-Deficient Aromatics in the synthesis of Pi-...
C–H bond hydroxylation at non heme carboxylate-bridged diiron centers

C–H bond hydroxylation at non heme carboxylate-bridged diiron centers
Bio inspired metal-oxo catalysts for c–h bond functionalization

Bio inspired metal-oxo catalysts for c–h bond functionalization
Stability and reactions of n heterocyclic carbenes

Stability and reactions of n heterocyclic carbenes
Recently uploaded
Cancer cell metabolism: special Reference to Lactate Pathway

Normal Cell Metabolism:
Cellular respiration describes the series of steps that cells use to break down sugar and other chemicals to get the energy we need to function.
Energy is stored in the bonds of glucose and when glucose is broken down, much of that energy is released.
Cell utilize energy in the form of ATP.
The first step of respiration is called glycolysis. In a series of steps, glycolysis breaks glucose into two smaller molecules - a chemical called pyruvate. A small amount of ATP is formed during this process.
Most healthy cells continue the breakdown in a second process, called the Kreb's cycle. The Kreb's cycle allows cells to “burn” the pyruvates made in glycolysis to get more ATP.
The last step in the breakdown of glucose is called oxidative phosphorylation (Ox-Phos).
It takes place in specialized cell structures called mitochondria. This process produces a large amount of ATP. Importantly, cells need oxygen to complete oxidative phosphorylation.
If a cell completes only glycolysis, only 2 molecules of ATP are made per glucose. However, if the cell completes the entire respiration process (glycolysis - Kreb's - oxidative phosphorylation), about 36 molecules of ATP are created, giving it much more energy to use.
IN CANCER CELL:
Unlike healthy cells that "burn" the entire molecule of sugar to capture a large amount of energy as ATP, cancer cells are wasteful.
Cancer cells only partially break down sugar molecules. They overuse the first step of respiration, glycolysis. They frequently do not complete the second step, oxidative phosphorylation.
This results in only 2 molecules of ATP per each glucose molecule instead of the 36 or so ATPs healthy cells gain. As a result, cancer cells need to use a lot more sugar molecules to get enough energy to survive.
Unlike healthy cells that "burn" the entire molecule of sugar to capture a large amount of energy as ATP, cancer cells are wasteful.
Cancer cells only partially break down sugar molecules. They overuse the first step of respiration, glycolysis. They frequently do not complete the second step, oxidative phosphorylation.
This results in only 2 molecules of ATP per each glucose molecule instead of the 36 or so ATPs healthy cells gain. As a result, cancer cells need to use a lot more sugar molecules to get enough energy to survive.
introduction to WARBERG PHENOMENA:
WARBURG EFFECT Usually, cancer cells are highly glycolytic (glucose addiction) and take up more glucose than do normal cells from outside.
Otto Heinrich Warburg (; 8 October 1883 – 1 August 1970) In 1931 was awarded the Nobel Prize in Physiology for his "discovery of the nature and mode of action of the respiratory enzyme.
WARNBURG EFFECT : cancer cells under aerobic (well-oxygenated) conditions to metabolize glucose to lactate (aerobic glycolysis) is known as the Warburg effect. Warburg made the observation that tumor slices consume glucose and secrete lactate at a higher rate than normal tissues.
Observation of Io’s Resurfacing via Plume Deposition Using Ground-based Adapt...

Since volcanic activity was first discovered on Io from Voyager images in 1979, changes
on Io’s surface have been monitored from both spacecraft and ground-based telescopes.
Here, we present the highest spatial resolution images of Io ever obtained from a groundbased telescope. These images, acquired by the SHARK-VIS instrument on the Large
Binocular Telescope, show evidence of a major resurfacing event on Io’s trailing hemisphere. When compared to the most recent spacecraft images, the SHARK-VIS images
show that a plume deposit from a powerful eruption at Pillan Patera has covered part
of the long-lived Pele plume deposit. Although this type of resurfacing event may be common on Io, few have been detected due to the rarity of spacecraft visits and the previously low spatial resolution available from Earth-based telescopes. The SHARK-VIS instrument ushers in a new era of high resolution imaging of Io’s surface using adaptive
optics at visible wavelengths.
Deep Behavioral Phenotyping in Systems Neuroscience for Functional Atlasing a...

Functional Magnetic Resonance Imaging (fMRI) provides means to characterize brain activations in response to behavior. However, cognitive neuroscience has been limited to group-level effects referring to the performance of specific tasks. To obtain the functional profile of elementary cognitive mechanisms, the combination of brain responses to many tasks is required. Yet, to date, both structural atlases and parcellation-based activations do not fully account for cognitive function and still present several limitations. Further, they do not adapt overall to individual characteristics. In this talk, I will give an account of deep-behavioral phenotyping strategies, namely data-driven methods in large task-fMRI datasets, to optimize functional brain-data collection and improve inference of effects-of-interest related to mental processes. Key to this approach is the employment of fast multi-functional paradigms rich on features that can be well parametrized and, consequently, facilitate the creation of psycho-physiological constructs to be modelled with imaging data. Particular emphasis will be given to music stimuli when studying high-order cognitive mechanisms, due to their ecological nature and quality to enable complex behavior compounded by discrete entities. I will also discuss how deep-behavioral phenotyping and individualized models applied to neuroimaging data can better account for the subject-specific organization of domain-general cognitive systems in the human brain. Finally, the accumulation of functional brain signatures brings the possibility to clarify relationships among tasks and create a univocal link between brain systems and mental functions through: (1) the development of ontologies proposing an organization of cognitive processes; and (2) brain-network taxonomies describing functional specialization. To this end, tools to improve commensurability in cognitive science are necessary, such as public repositories, ontology-based platforms and automated meta-analysis tools. I will thus discuss some brain-atlasing resources currently under development, and their applicability in cognitive as well as clinical neuroscience.
Structures and textures of metamorphic rocks

It is useful for the Under Graduating students for easy understanding and it's useful for the exam preparations.
Nucleic Acid-its structural and functional complexity.

This presentation explores a brief idea about the structural and functional attributes of nucleotides, the structure and function of genetic materials along with the impact of UV rays and pH upon them.
Structural Classification Of Protein (SCOP)

A brief information about the SCOP protein database used in bioinformatics.
The Structural Classification of Proteins (SCOP) database is a comprehensive and authoritative resource for the structural and evolutionary relationships of proteins. It provides a detailed and curated classification of protein structures, grouping them into families, superfamilies, and folds based on their structural and sequence similarities.
What is greenhouse gasses and how many gasses are there to affect the Earth.

What are greenhouse gasses how they affect the earth and its environment what is the future of the environment and earth how the weather and the climate effects.
RNA INTERFERENCE: UNRAVELING GENETIC SILENCING

Introduction:
RNA interference (RNAi) or Post-Transcriptional Gene Silencing (PTGS) is an important biological process for modulating eukaryotic gene expression.
It is highly conserved process of posttranscriptional gene silencing by which double stranded RNA (dsRNA) causes sequence-specific degradation of mRNA sequences.
dsRNA-induced gene silencing (RNAi) is reported in a wide range of eukaryotes ranging from worms, insects, mammals and plants.
This process mediates resistance to both endogenous parasitic and exogenous pathogenic nucleic acids, and regulates the expression of protein-coding genes.
What are small ncRNAs?
micro RNA (miRNA)
short interfering RNA (siRNA)
Properties of small non-coding RNA:
Involved in silencing mRNA transcripts.
Called “small” because they are usually only about 21-24 nucleotides long.
Synthesized by first cutting up longer precursor sequences (like the 61nt one that Lee discovered).
Silence an mRNA by base pairing with some sequence on the mRNA.
Discovery of siRNA?
The first small RNA:
In 1993 Rosalind Lee (Victor Ambros lab) was studying a non- coding gene in C. elegans, lin-4, that was involved in silencing of another gene, lin-14, at the appropriate time in the
development of the worm C. elegans.
Two small transcripts of lin-4 (22nt and 61nt) were found to be complementary to a sequence in the 3' UTR of lin-14.
Because lin-4 encoded no protein, she deduced that it must be these transcripts that are causing the silencing by RNA-RNA interactions.
Types of RNAi ( non coding RNA)
MiRNA
Length (23-25 nt)
Trans acting
Binds with target MRNA in mismatch
Translation inhibition
Si RNA
Length 21 nt.
Cis acting
Bind with target Mrna in perfect complementary sequence
Piwi-RNA
Length ; 25 to 36 nt.
Expressed in Germ Cells
Regulates trnasposomes activity
MECHANISM OF RNAI:
First the double-stranded RNA teams up with a protein complex named Dicer, which cuts the long RNA into short pieces.
Then another protein complex called RISC (RNA-induced silencing complex) discards one of the two RNA strands.
The RISC-docked, single-stranded RNA then pairs with the homologous mRNA and destroys it.
THE RISC COMPLEX:
RISC is large(>500kD) RNA multi- protein Binding complex which triggers MRNA degradation in response to MRNA
Unwinding of double stranded Si RNA by ATP independent Helicase
Active component of RISC is Ago proteins( ENDONUCLEASE) which cleave target MRNA.
DICER: endonuclease (RNase Family III)
Argonaute: Central Component of the RNA-Induced Silencing Complex (RISC)
One strand of the dsRNA produced by Dicer is retained in the RISC complex in association with Argonaute
ARGONAUTE PROTEIN :
1.PAZ(PIWI/Argonaute/ Zwille)- Recognition of target MRNA
2.PIWI (p-element induced wimpy Testis)- breaks Phosphodiester bond of mRNA.)RNAse H activity.
MiRNA:
The Double-stranded RNAs are naturally produced in eukaryotic cells during development, and they have a key role in regulating gene expression .
Orion Air Quality Monitoring Systems - CWS

Professional air quality monitoring systems provide immediate, on-site data for analysis, compliance, and decision-making.
Monitor common gases, weather parameters, particulates.
Richard's entangled aventures in wonderland

Since the loophole-free Bell experiments of 2020 and the Nobel prizes in physics of 2022, critics of Bell's work have retreated to the fortress of super-determinism. Now, super-determinism is a derogatory word - it just means "determinism". Palmer, Hance and Hossenfelder argue that quantum mechanics and determinism are not incompatible, using a sophisticated mathematical construction based on a subtle thinning of allowed states and measurements in quantum mechanics, such that what is left appears to make Bell's argument fail, without altering the empirical predictions of quantum mechanics. I think however that it is a smoke screen, and the slogan "lost in math" comes to my mind. I will discuss some other recent disproofs of Bell's theorem using the language of causality based on causal graphs. Causal thinking is also central to law and justice. I will mention surprising connections to my work on serial killer nurse cases, in particular the Dutch case of Lucia de Berk and the current UK case of Lucy Letby.
filosofia boliviana introducción jsjdjd.pptx

La filosofía boliviana y la búsqueda por construir pensamientos propios
Citrus Greening Disease and its Management

Citrus Greening was one of the major causes of decline in the citrus production. So, effective management cultural practices should be incorporated
Recently uploaded (20)
Cancer cell metabolism: special Reference to Lactate Pathway

Cancer cell metabolism: special Reference to Lactate Pathway
Observation of Io’s Resurfacing via Plume Deposition Using Ground-based Adapt...

Observation of Io’s Resurfacing via Plume Deposition Using Ground-based Adapt...
Deep Behavioral Phenotyping in Systems Neuroscience for Functional Atlasing a...

Deep Behavioral Phenotyping in Systems Neuroscience for Functional Atlasing a...
Nucleic Acid-its structural and functional complexity.

Nucleic Acid-its structural and functional complexity.
What is greenhouse gasses and how many gasses are there to affect the Earth.

What is greenhouse gasses and how many gasses are there to affect the Earth.
Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...

Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...
platelets- lifespan -Clot retraction-disorders.pptx

platelets- lifespan -Clot retraction-disorders.pptx
In silico drugs analogue design: novobiocin analogues.pptx

In silico drugs analogue design: novobiocin analogues.pptx
Marcus theory
- 1. Organic Pedagogical Electronic Network Marcus Theory Elizabeth Greenhalgh, Amanda Bischoff, and Matthew Sigman University of Utah
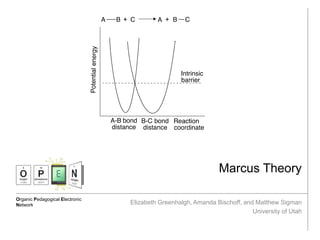
- 2. Describing Electron Transfer Reactions Marcus, R. A. The Nobel Prize in Chemistry 1992 1992, 69-92 Anslyn, E. V.; Dougherty, D.A (2006) Modern Physical Organic Chemistry. University Science Books Marcus, R. A. The Journal of Chemical Physics 1956, 24(5), 966-978 Transition State Theory (TST) Overview: • Developed by Henry Eyring in the 1930s • Describes reaction rates focusing on geometry of the transition state at the top of the energy barrier • Effective for describing bond breakage/bond formation The Problem: Reactions that do not involve bond breakage/formation, i.e. electron transfer reactions, involve little nuclear movement in the transition state (rate of electron transfer is faster than rate of molecular vibrations); thus, TST fails and this necessitates a different model. Background: Morse potentials Morse potentials describe potential energy as a function of bond distance. Marcus theory focuses on a parabolic approximation of the boxed portion. Frank-Condon principle In an electronic transition between two reactants, the solvent molecules do not have time to rearrange; thus, the atomic configuration and total energy of the system remain the same; only the electronic state changes. Quantum Tunneling Some small particles (electrons, hydrogen atoms, etc.) can tunnel through energy barriers rather than going over them. This complicates free energy calculations.
- 3. Marcus Theory and Implications Marcus, R. A. The Nobel Prize in Chemistry 1992 1992, 69-92 Anslyn, E. V.; Dougherty, D.A (2006) Modern Physical Organic Chemistry. University Science Books Why does Marcus theory work when TST doesn’t? TST focuses on orientation of reactants, while Marcus theory accounts for the many configurations of the solvent surrounding the inner coordination sphere; Marcus theory finds the potential curves for different orientations of the solvent molecules, and the point of intersection is the point at which electron transfer occurs. Marcus Theory Overview of Marcus Theory: • Developed by Rudolph A. Marcus in the 1950s • Uses Morse potentials to describe reaction coordinates • This theory won the Nobel Prize in chemistry in 1992 Marcus Theory and Exergonicity After aligning the Morse potentials along the intrinsic barrier, the product potential is adjusted to compensate for the free energy change of the reaction. The intersection of the curves defines the energy of the transition state. A tunneling term is also included. As the reaction becomes more exergonic, the energy of the transition state lowers; but, as it becomes still more exergonic, the energy of the transition state rises! The region beyond the zero point is called the “Marcus inverted region.”
- 4. Marcus Theory to Predict Hydrogen Atom Transfer Mayer, J. Understanding Hydrogen Atom Transfer: From Bond Strengths to Marcus Theory, Acc. Chem. Res. 2011 44 (1), 36-46. A H + A A + H A kAH/A (3) The simplest form of the Marcus equation for electron transfer (eq 1) predicts the reaction barrier (∆G‡) from the reaction driving force (∆G°) and intrinsic barrier (λ) which is the energy required to reorganize the reactants and surrounding solvent without electron transfer. This equation, with a few assumptions, can be rearranged to the Marcus cross relation (eq 2). The kinetic information is primarily in the rate constants for the respective hydrogen-atom self exchange reactions (such as eq 3). Tests of the Marcus cross relation for HAT: log/log plot of observed versus calculated HAT rate constant for a number of metal complexes reacting with various substrates. The diagonal line illustrates kobs = kcalc. The estimated errors on kcalc are typically ±1 log unit; they are larger for MeCN reactions of Ru(O)bpy2py2+ because the BDFE is only available in H2O, and smaller in three cases where KAH/B was measured directly.
- 5. Hydrogen Atom Transfer Continued Schematic free energy surface for the reaction shown above through H bonding intermediates. HAT from FeII(H2bip) to the stable nitroxyl radical TEMPO is very unusual in that it becomes faster at lower temperatures, with ∆H‡ = -2.7 ± 0.4 kcal mol-1. The figure below shows the cross relation quantitatively predicts the cross rate constants and the negative temperature dependence. The most temperature dependent parameter is keq, as the reaction is more favorable at lower temperatures causing the Marcus cross relation to have a temperature dependence as well. Mayer, J. Understanding Hydrogen Atom Transfer: From Bond Strengths to Marcus Theory, Acc. Chem. Res. 2011 44 (1), 36-46.
- 6. Problems 1. What does Marcus theory describe that transition state theory doesn’t? a. Quantum tunneling b. Electron transfer reactions c. Transition state energies d. All of the above 2. Which pair of potential curves would you expect to result in the reaction with the fastest rate?
- 7. Problems 3. Does the box on the adjacent diagram represent the intrinsic barrier, the transition state, or both? What is the difference between the two? 4. Marcus cross relation relates what values? a. Resonance b. Induction c. Reaction rates d. All of the above 5. Marcus theory is based on the quantum mechanical principle called_______. 6. True or False: Marcus cross relation can tell us information about temperature.
- 8. This work is licensed under a Creative Commons Attribution- ShareAlike 4.0 International License. Contributed by: Elizabeth Greenhalgh, Amanda Bischoff, and Matthew Sigman University of Utah, 2015