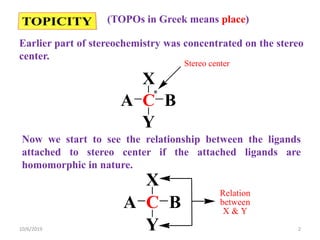
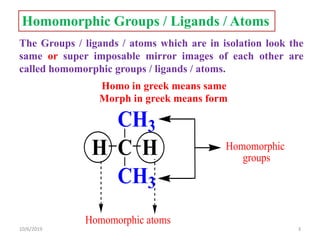
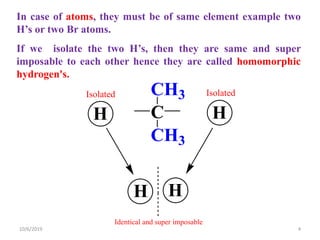
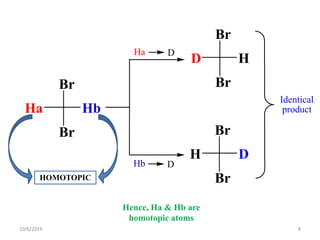
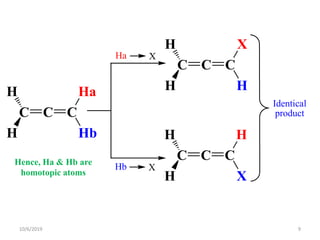
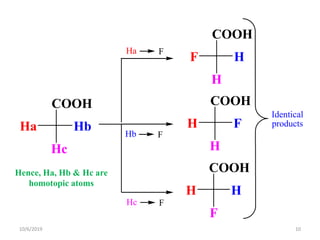
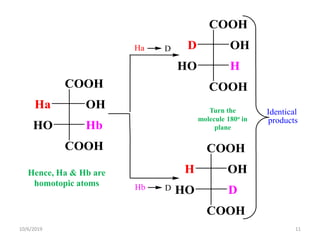
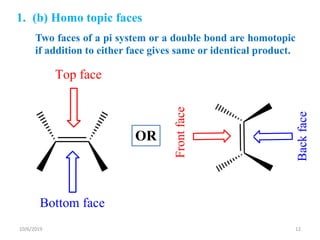
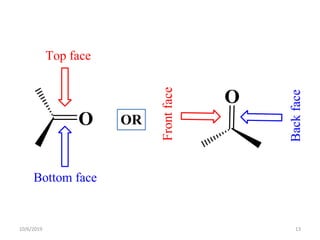
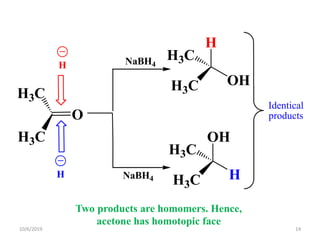
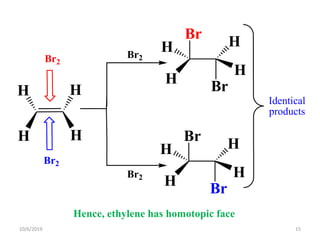
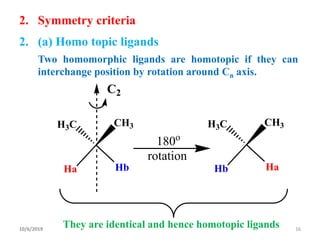
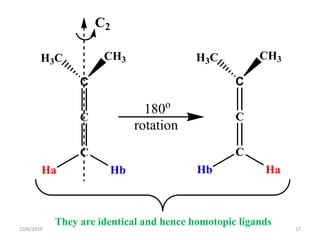
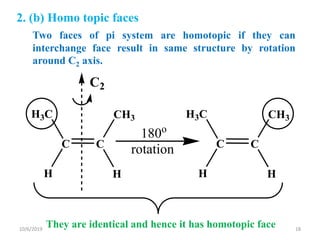
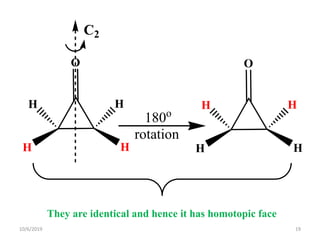
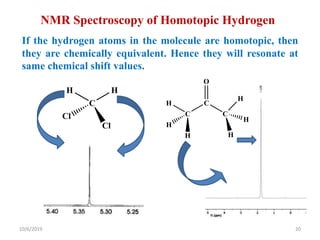
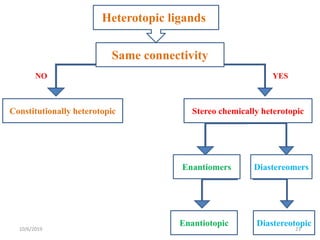
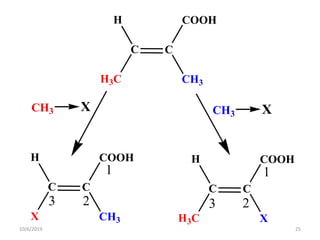
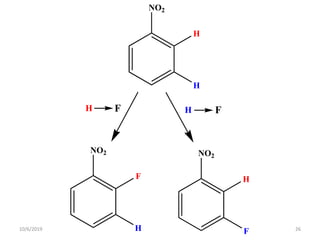
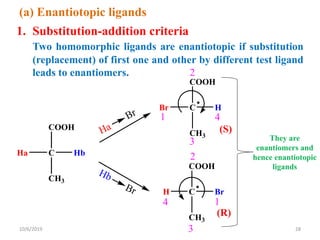
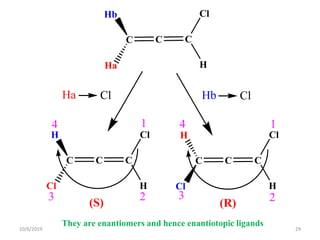
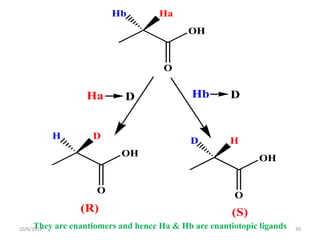
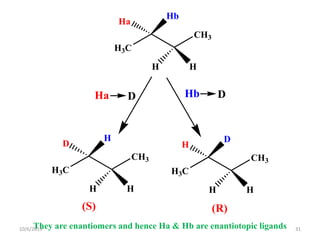
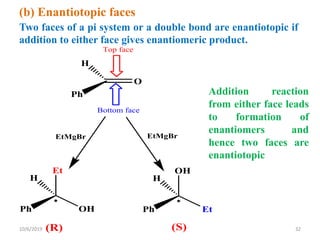
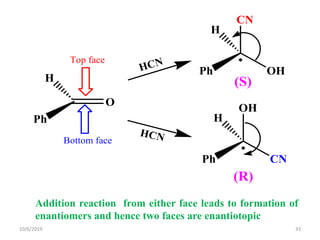
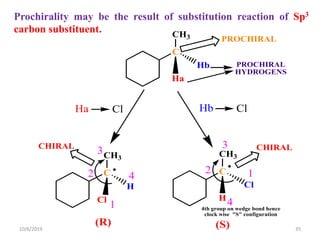
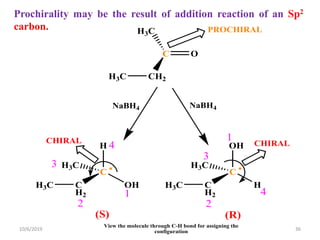
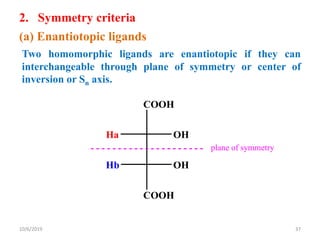
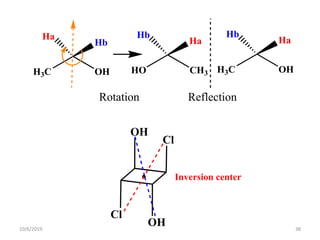
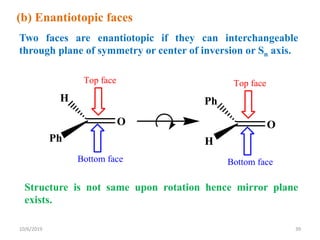
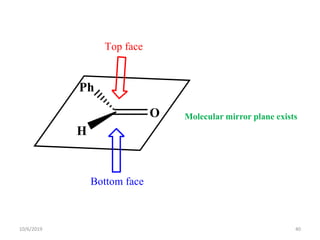
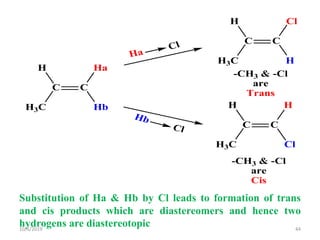
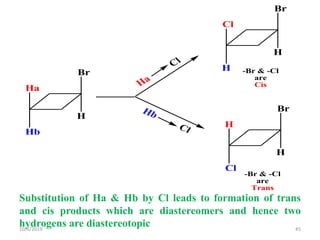
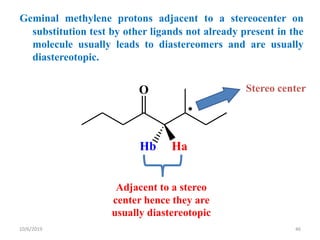
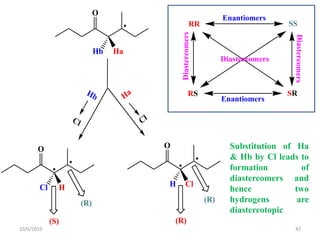
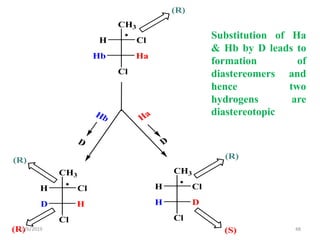
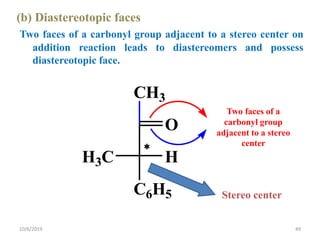
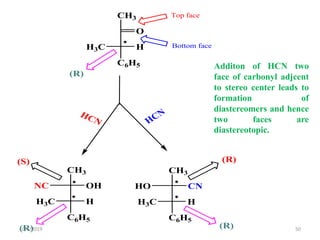
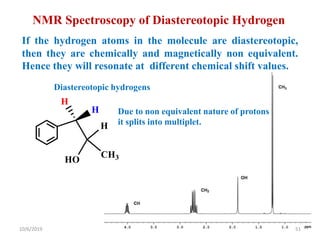
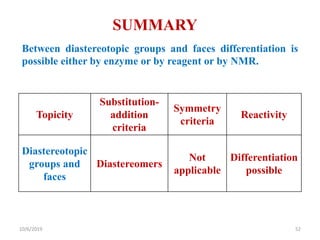
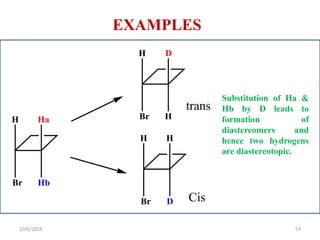
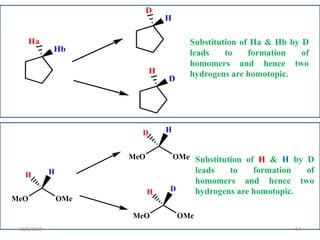
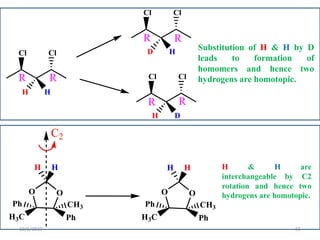
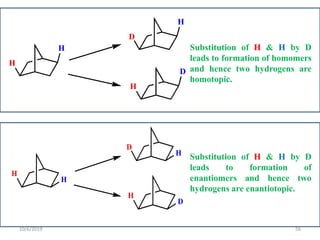
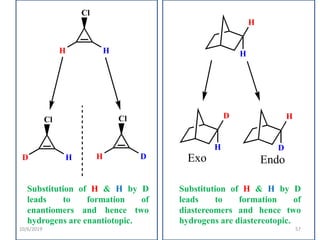
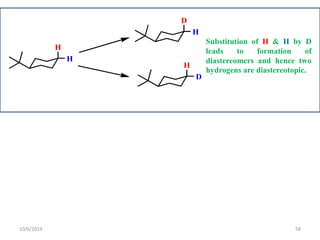
The document discusses the concepts of homotopic and heterotopic ligands within the context of stereochemistry, specifically focusing on the criteria for determining the equivalence of ligands based on substitution and symmetry. It explains how homotopic ligands result in identical products upon substitution, while heterotopic ligands yield structurally different products. Additionally, it elaborates on the significance of NMR spectroscopy in identifying homotopic and diastereotopic ligands based on their chemical equivalence.