Inductive effects
•Download as PPTX, PDF•
6 likes•5,582 views
This document discusses inductive effects in organic chemistry. It begins by defining inductive effects as the withdrawal of electrons from a σ bond to the more electronegative atom or group, creating a dipole. This effect can alter the reactivity of other groups on the molecule. More electronegative substituents increase the acidity of carboxylic acids by withdrawing electron density from the acidic proton. The document also examines how inductive effects influence aromatic systems' affinity for carbon dioxide, with methyl groups increasing and halogen groups decreasing this affinity through their respective positive and negative inductive effects.
Report
Share
Report
Share
Recommended
1.5 elimination reaction

This power point presentation summarizes elimination reactions, specifically 1,2 elimination reactions. It defines elimination reactions as reactions where two atoms or groups are removed from a reactant to form an unsaturated product. 1,2 elimination reactions eliminate atoms or groups from the 1 and 2 positions on a molecule. Three possible mechanisms are discussed: E2, E1, and E1cb. Evidence for the E2 mechanism includes kinetic isotope effects, the element effect showing dependence on leaving group ability, and the lack of hydrogen exchange. The Saytzeff rule and factors influencing its application, like stability and transition state crowding, are also covered.
Inductive effect and mesomeric effect

The document discusses inductive effect and resonance effect. Inductive effect refers to polarization of a sigma bond due to electron withdrawing or donating groups. Electron withdrawing groups have a negative inductive effect (-I) while electron donating groups have a positive inductive effect (+I). Resonance effect refers to delocalization of pi electrons or a lone pair. Electron withdrawing groups have a negative resonance effect (-R) while electron donating groups have a positive resonance effect (+R). In most cases, resonance effect is stronger than inductive effect. The document provides examples of how these effects influence acidity, reactivity, and stability.
Hyperconjugation effect

This document discusses the concept of hyperconjugation, which involves the delocalization of sigma electrons from an adjacent C-H bond into an empty p-orbital of an unsaturated system like an alkene or benzene ring. This effect increases the stability of alkenes and carbocations with more alkyl substituents by allowing for additional no bond resonance structures. The stability of alkenes and carbocations increases with the number of alkyl groups due to greater hyperconjugative stabilization from more C-H bonds. Hyperconjugation is an important effect that helps explain the observed stability and reactivity patterns of unsaturated organic compounds.
Effect of conformation on reactivity

This document discusses the effect of conformation on the reactivity of acyclic and cyclic compounds. It provides examples of how the conformation of a compound, whether the substituents are equatorial or axial, can impact the rate and outcome of various chemical reactions including E2 eliminations, SN1 and SN2 substitutions, esterification, hydrolysis, elimination reactions, molecular rearrangements, neighboring group participation, and oxidation reactions. Specifically, it notes that equatorial substituents are often more reactive than axial substituents due to differences in steric hindrance and stereoelectronic effects. Reaction rates and products can differ significantly between conformational isomers of the same compound.
Metal carbonyls

Metal carbonyls are coordination complexes of transition metals with carbon monoxide ligands. They were first synthesized in 1868 by passing carbon monoxide over platinum. Metal carbonyls typically obey the 18 electron rule and are often diamagnetic. They have applications as catalysts in organic synthesis and in producing pure metals like nickel. Precautions must be taken when using metal carbonyls due to their toxicity.
Basic effects in Organic chemistry

Basic effects in Organic chemistrySri Ramakrishna Mission Vidyalaya College of Arts and Science,Coimbatore-20.
This presentaion describes about the basic principle effects in organic chemistry like inductive,mesomeric,electromeric, resonance and hyperconjugation. this presentation contains some JAM competitive questions.Carbene

This document discusses carbenes, which are neutral carbon molecules with two unshared valence electrons. It describes the different types of carbenes, including singlet and triplet carbenes, and their electronic structures and bonding properties. Methods of forming carbenes are presented, such as alpha elimination reactions and decomposition of diazo compounds. The major reactions of carbenes are also summarized, including insertion, addition, and rearrangement reactions. Carbene reactivity depends on whether they are in singlet or triplet states.
Inductive effect

This is a presentation file on Inductive Effect, Bond Length, Bond Energy, Bond Angle for the course Organic Pharmacy I, course code is PHAR-1105 specially for the pharmacy students. Also it can be used for the Biochemistry students and other like as HSC level in Bangladesh or another country. We are the students of Mawlana Bhashani Science and Technology University, department of pharmacy which is located in Tangail.
Thank you for your patient.
Recommended
1.5 elimination reaction

This power point presentation summarizes elimination reactions, specifically 1,2 elimination reactions. It defines elimination reactions as reactions where two atoms or groups are removed from a reactant to form an unsaturated product. 1,2 elimination reactions eliminate atoms or groups from the 1 and 2 positions on a molecule. Three possible mechanisms are discussed: E2, E1, and E1cb. Evidence for the E2 mechanism includes kinetic isotope effects, the element effect showing dependence on leaving group ability, and the lack of hydrogen exchange. The Saytzeff rule and factors influencing its application, like stability and transition state crowding, are also covered.
Inductive effect and mesomeric effect

The document discusses inductive effect and resonance effect. Inductive effect refers to polarization of a sigma bond due to electron withdrawing or donating groups. Electron withdrawing groups have a negative inductive effect (-I) while electron donating groups have a positive inductive effect (+I). Resonance effect refers to delocalization of pi electrons or a lone pair. Electron withdrawing groups have a negative resonance effect (-R) while electron donating groups have a positive resonance effect (+R). In most cases, resonance effect is stronger than inductive effect. The document provides examples of how these effects influence acidity, reactivity, and stability.
Hyperconjugation effect

This document discusses the concept of hyperconjugation, which involves the delocalization of sigma electrons from an adjacent C-H bond into an empty p-orbital of an unsaturated system like an alkene or benzene ring. This effect increases the stability of alkenes and carbocations with more alkyl substituents by allowing for additional no bond resonance structures. The stability of alkenes and carbocations increases with the number of alkyl groups due to greater hyperconjugative stabilization from more C-H bonds. Hyperconjugation is an important effect that helps explain the observed stability and reactivity patterns of unsaturated organic compounds.
Effect of conformation on reactivity

This document discusses the effect of conformation on the reactivity of acyclic and cyclic compounds. It provides examples of how the conformation of a compound, whether the substituents are equatorial or axial, can impact the rate and outcome of various chemical reactions including E2 eliminations, SN1 and SN2 substitutions, esterification, hydrolysis, elimination reactions, molecular rearrangements, neighboring group participation, and oxidation reactions. Specifically, it notes that equatorial substituents are often more reactive than axial substituents due to differences in steric hindrance and stereoelectronic effects. Reaction rates and products can differ significantly between conformational isomers of the same compound.
Metal carbonyls

Metal carbonyls are coordination complexes of transition metals with carbon monoxide ligands. They were first synthesized in 1868 by passing carbon monoxide over platinum. Metal carbonyls typically obey the 18 electron rule and are often diamagnetic. They have applications as catalysts in organic synthesis and in producing pure metals like nickel. Precautions must be taken when using metal carbonyls due to their toxicity.
Basic effects in Organic chemistry

Basic effects in Organic chemistrySri Ramakrishna Mission Vidyalaya College of Arts and Science,Coimbatore-20.
This presentaion describes about the basic principle effects in organic chemistry like inductive,mesomeric,electromeric, resonance and hyperconjugation. this presentation contains some JAM competitive questions.Carbene

This document discusses carbenes, which are neutral carbon molecules with two unshared valence electrons. It describes the different types of carbenes, including singlet and triplet carbenes, and their electronic structures and bonding properties. Methods of forming carbenes are presented, such as alpha elimination reactions and decomposition of diazo compounds. The major reactions of carbenes are also summarized, including insertion, addition, and rearrangement reactions. Carbene reactivity depends on whether they are in singlet or triplet states.
Inductive effect

This is a presentation file on Inductive Effect, Bond Length, Bond Energy, Bond Angle for the course Organic Pharmacy I, course code is PHAR-1105 specially for the pharmacy students. Also it can be used for the Biochemistry students and other like as HSC level in Bangladesh or another country. We are the students of Mawlana Bhashani Science and Technology University, department of pharmacy which is located in Tangail.
Thank you for your patient.
Crown ethers

Crown ethers
NOMENCLATURE
GENERAL SYNTHESIS OF CROWN ETHER
AZA CROWN
CRYPTAND
APPLICATIONS
1. SYNTHETIC APPLICTION
Esterification
Saponification
Anhydride formation
Potassium permanganate oxidation
Aromatic substitution reactions
Elimination reactions
Displacement reaction
Generation of carbenes
Superoxide anion
Alkylations – 1. o-alkylations
2. c-alkylations
3. n-alkylations
2. ANALYTICAL APPLICATION
Determination of gold in geological samples
Super critical fluid extraction of trace metal from solid and liquid materials
Application of ionic liquids in analytical chemistry
Oxidation and determination of aldehydes
Crown ethers are used in the laboratory as phase transfer catalyst
OTHER APPLICATION
It is used in photocynation
Resolution of racemic mixture
Benzoin condensation
Hetrocyclisation
Synthesis of furanones
Acetylation of secondary amines in presence of primary amine
Hyperconjugation

Dr. Neelam from the Department of Chemistry presented on the topic of hyperconjugation. Hyperconjugation is the delocalization of σ-electrons from a C-H bond into an adjacent unsaturated system. It can occur in alkenes, alkynes, carbocations, and carbon radicals. The number of possible hyperconjugative structures equals the number of alpha hydrogens on sp3 hybridized carbon atoms. Hyperconjugation explains trends in stability and heats of hydrogenation between different alkenes. It is a permanent effect that does not change hybridization and is distance independent.
Birch reduction

The Birch reduction is a reaction where aromatic compounds undergo partial reduction to unconjugated cyclohexadiene compounds in the presence of alkali metals like sodium or lithium in liquid ammonia. The solvated electrons from the reaction of the metal with liquid ammonia give the solution an intense blue color. The mechanism begins with single electron transfer from the metal to the aromatic ring, forming a radical anion. Regioselectivity in the reduction depends on whether substituents on the aromatic ring are electron donating groups or electron withdrawing groups. The Birch reduction can selectively reduce the less electron-rich ring in bicyclic aromatic compounds.
Bent rule

Bent's rule describes how the hybridization of central atoms in molecules relates to the electronegativity of substituents. More electronegative elements prefer hybrid orbitals with less s character and more p character, while less electronegative substituents prefer orbitals with more s character. This explains differences in bond lengths and angles compared to ideal values, as bond length decreases and angle decreases with increasing p character directed towards more electronegative substituents. Examples of bent's rule justification include the decreased bond angle in fluoromethane compared to methane due to less s character in the C-F bond.
E2 reaction

1. The document outlines different elimination reaction mechanisms including E2, E1, and E1cb.
2. It discusses the regiochemistry and stereochemistry of elimination reactions and how Zaytzeff's rule and Hofmann's rule apply.
3. The key differences between the E2, E1, and E1cb mechanisms are described along with factors that determine whether substitution or elimination will occur for a given reaction.
Reduction reactions

This document discusses reduction reactions and reducing agents. It aims to teach the reader to: 1) exploit differences in reactivity between hydride and neutral reducing agents to achieve chemoselective reductions; 2) use substrate chirality to control syn vs. anti diastereoselectivity in ketone reductions; 3) rationalize reaction outcomes using transition state diagrams; 4) appreciate the versatility of transition metals in reductions; 5) understand the utility of dissolving metal reductions; and 6) use radical chemistry for deoxygenation and halide reduction. It then provides details on various hydride and neutral reducing agents, focusing on their reactivities and applications in selective reductions.
Electrophilic Substitution Reaction

The document discusses different types of electrophilic substitution reactions: SE1, SE2, and SEi. SE1 reactions follow first-order kinetics and involve two steps - rate-determining ionization and fast combination. SE2 reactions also follow first-order kinetics, but occur in a single step through a transition state. SE2 reactions can result in retention or inversion of configuration. SEi reactions are concerted mechanisms where the electrophile assists in removing the leaving group, leading to retention of configuration.
Acids, bases & aromaticity

This document provides information on acids, bases, and aromaticity. It defines acids and bases according to Arrhenius, Bronsted-Lowry, and Lewis theories. Acids are substances that produce H+ ions or accept electron pairs, while bases produce OH- ions or donate electron pairs. The document discusses factors that determine acid and base strength such as conjugate base stability, bond strength, resonance, induction, and hybridization effects. It also provides examples of acid-base reactions and uses pKa values to predict reaction equilibrium and relative acidities.
Organic reaction mechanism

The document discusses different types of substitution reactions including nucleophilic substitution, electrophilic substitution, and free radical substitution. It provides details on the mechanisms, kinetics, stereochemistry and factors affecting the rate of nucleophilic substitution reactions SN1 and SN2. SN1 follows a unimolecular mechanism involving a carbocation intermediate while SN2 follows a bimolecular mechanism with a single concerted transition state. The document also discusses electrophilic aromatic substitution reactions and addition and elimination reactions of alkenes and alkynes.
Migratory aptitudes in rearrangement reaction

The document discusses migratory aptitude in rearrangement reactions. It defines migratory aptitude as the relative ability of a migrating group to migrate in a rearrangement reaction. Factors that affect migratory aptitude include the stability of the carbocation formed and the electron density of the migrating group. Aryl groups generally have higher migratory aptitude than alkyl groups. The document also describes different types of rearrangement reactions and the mechanisms of nucleophilic rearrangements.
Chemical kinetics I

This presentation is based on the Topic " Chemical Kinetics" for the Chemistry undergraduate students of Mumbai University
Nonclassical Carbocation

This document discusses classical and nonclassical carbocations. Nonclassical carbocations have charge delocalization from neighboring bonds like C=C pi bonds. The main difference is that classical carbocations have charge localized on one carbon, while nonclassical carbocations have charge delocalized by double or single bonds not in the allylic position. Examples like the norbornyl carbocation are given to show how neighboring double bonds can stabilize and delocalize charge through 3-center bonds. Reaction rates and product stereochemistry provide evidence for nonclassical intermediates. While some challenged this view, most chemists accept nonclassical interpretations of carbocation reactions.
Aromatic electrophilic substitution

This document provides an overview of aromatic electrophilic substitution reactions (AES). It defines important terms like arenium ions, electrophiles, nucleophiles and discusses the effects of substituents on reactivity. The mechanisms of common AES reactions like nitration, sulfonation, Friedel-Crafts alkylation and acylation are covered. The document also discusses topics like the mesomeric and inductive effects of substituents, the synthesis of tribromobenzene, and the relative reactivities of benzene and substituted benzenes in bromination. Examples of AES on phenols, xylenes, cresols and other aromatic compounds are provided.
Activating and deactivating group

Activating groups donate electrons to the benzene ring through resonance or inductive effects, increasing its electron density and reactivity toward electrophilic substitution. Common ortho-para directing and activating groups include -NH2, -NHR, -O-, -OH, -OCH3, and -CH3. Deactivating groups withdraw electrons from the benzene ring through resonance or inductive effects, decreasing its electron density and reactivity. Common meta directing and deactivating groups are halogens (-Cl, -Br) and nitro (-NO2). The direction and degree of activation or deactivation depends on whether the resonance or inductive effects dominate for a particular substituent.
Neighbouring Group Participation.pptx

(i) Non-classical carbocations display delocalization of sigma bonds through 3-center-2-electron bonds in bridged systems. Neighboring group participation can assist reactions by donating electrons through lone pairs, pi bonds, aromatic rings, or sigma bonds.
(ii) The pinacol-pinacolone rearrangement involves the migration of an alkyl group from one carbon to another after the loss of a leaving group from a vicinal diol. The migration is assisted by delocalization of the carbocation intermediate onto the oxygen atom.
(iii) In asymmetrical glycols, the group with greater ability for carbocation delocalization, such as phenyl, will migrate preferentially over
Factors affecting stability of metal complexes

1. The stability of metal complexes is affected by factors such as the nature of the central metal ion, the coordinating ligand, and the presence of ring structures. The charge, size, and ionization energy of the metal ion influence stability, as do the size, charge, and basic strength of the ligand.
2. Chelation, where a multidentate ligand bonds to the metal ion at multiple sites, generally enhances stability. Five-membered rings formed by chelation are most stable due to reduced strain. An increase in the number of chelate rings or delocalized π-electrons in ring structures also increases stability.
3. Other factors like forced ligand configurations, the solvent environment, and
Addition reaction sm

Addition reactions occur when two reactants combine to form a new product with no leftover atoms. In an addition reaction, new groups are added to the starting material, breaking a pi bond and forming two sigma bonds. Addition reactions involve the addition of electrophiles, radicals, or nucleophiles across multiple bonds such as carbon-carbon double or triple bonds.
Organic Acid and Bases - Swathi S Rao

- An organic acid is an organic compound that contains a carboxyl group (-COOH) which gives it acidic properties. The strength of an organic acid depends on the stability of its conjugate base.
- Organic bases usually contain nitrogen atoms that can accept protons. Common organic bases include amines, which have a lone pair of electrons on the nitrogen that can accept protons.
- Acid strength is determined by the amount of hydronium (H3O+) ions produced when the acid dissociates in water. Base strength is determined by the amount of hydroxide (OH-) ions produced upon dissolution in water. Factors like bond strength, stability of the conjugate base, and inductive effects influence
Electrophilic Substitution reactions

This document provides an overview of electrophilic substitution reactions. It defines electrophilic substitution as a reaction where a functional group is attached to a compound by replacing another functional group, often a hydrogen atom. It describes two main types: electrophilic aromatic substitution reactions, where an atom attached to an aromatic ring is replaced; and electrophilic aliphatic substitution reactions, where hydrogen in an aliphatic compound is usually replaced. The document also outlines the three step mechanism of electrophilic substitution reactions: 1) generating an electrophile, 2) forming a carbocation, and 3) eliminating a proton to restore aromaticity.
Demjanov rearrangement

The document summarizes the Tiffeneau–Demjanov rearrangement reaction. It was discovered in the early 1900s by French chemist Marc Émile Pierre Adolphe Tiffeneau and Russian chemist Nikolay Yakovlevich Demyanov. The reaction involves treating 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone through a 1,2-carbon migration. This ring expansion reaction is useful for increasing the size of amino-substituted cyclic compounds from four to eight-membered rings. The mechanism involves diazotization of the amine to form a diazonium ion that undergoes 1,2-alkyl shift accompanied by nitrogen loss to form
Solvation effects on reactions

Solvation can be defined as any stabilizing interaction of a solute (or solute moiety) and the solvent. These interactions can be weak, purely electrostatic, as is the case with non-polar solutes and solvents, or more significant, involving the interactions between dipole moments or between dipoles and formal charges.
Contributed by: Anton S. Klimenko (Undergraduate), Department of Chemistry, The University of Utah, 2016
ch1_Basic_concepts_Organic_sem1.ppt

This document discusses various concepts related to organic chemistry including carbocations, carbocation stability, inductive effects, mesomeric effects, hyperconjugation, conjugation, and resonance. It explains that carbocations are carbon-containing species with a positive formal charge that are classified based on the number of carbons bonded to the charged carbon. Tertiary carbocations are the most stable due to effects such as hyperconjugation and inductive/mesomeric effects of alkyl groups that stabilize the positive charge. Inductive and mesomeric effects involve polarization of bonds that can stabilize or destabilize carbocations. Hyperconjugation and conjugation further contribute to stability through resonance structures.
More Related Content
What's hot
Crown ethers

Crown ethers
NOMENCLATURE
GENERAL SYNTHESIS OF CROWN ETHER
AZA CROWN
CRYPTAND
APPLICATIONS
1. SYNTHETIC APPLICTION
Esterification
Saponification
Anhydride formation
Potassium permanganate oxidation
Aromatic substitution reactions
Elimination reactions
Displacement reaction
Generation of carbenes
Superoxide anion
Alkylations – 1. o-alkylations
2. c-alkylations
3. n-alkylations
2. ANALYTICAL APPLICATION
Determination of gold in geological samples
Super critical fluid extraction of trace metal from solid and liquid materials
Application of ionic liquids in analytical chemistry
Oxidation and determination of aldehydes
Crown ethers are used in the laboratory as phase transfer catalyst
OTHER APPLICATION
It is used in photocynation
Resolution of racemic mixture
Benzoin condensation
Hetrocyclisation
Synthesis of furanones
Acetylation of secondary amines in presence of primary amine
Hyperconjugation

Dr. Neelam from the Department of Chemistry presented on the topic of hyperconjugation. Hyperconjugation is the delocalization of σ-electrons from a C-H bond into an adjacent unsaturated system. It can occur in alkenes, alkynes, carbocations, and carbon radicals. The number of possible hyperconjugative structures equals the number of alpha hydrogens on sp3 hybridized carbon atoms. Hyperconjugation explains trends in stability and heats of hydrogenation between different alkenes. It is a permanent effect that does not change hybridization and is distance independent.
Birch reduction

The Birch reduction is a reaction where aromatic compounds undergo partial reduction to unconjugated cyclohexadiene compounds in the presence of alkali metals like sodium or lithium in liquid ammonia. The solvated electrons from the reaction of the metal with liquid ammonia give the solution an intense blue color. The mechanism begins with single electron transfer from the metal to the aromatic ring, forming a radical anion. Regioselectivity in the reduction depends on whether substituents on the aromatic ring are electron donating groups or electron withdrawing groups. The Birch reduction can selectively reduce the less electron-rich ring in bicyclic aromatic compounds.
Bent rule

Bent's rule describes how the hybridization of central atoms in molecules relates to the electronegativity of substituents. More electronegative elements prefer hybrid orbitals with less s character and more p character, while less electronegative substituents prefer orbitals with more s character. This explains differences in bond lengths and angles compared to ideal values, as bond length decreases and angle decreases with increasing p character directed towards more electronegative substituents. Examples of bent's rule justification include the decreased bond angle in fluoromethane compared to methane due to less s character in the C-F bond.
E2 reaction

1. The document outlines different elimination reaction mechanisms including E2, E1, and E1cb.
2. It discusses the regiochemistry and stereochemistry of elimination reactions and how Zaytzeff's rule and Hofmann's rule apply.
3. The key differences between the E2, E1, and E1cb mechanisms are described along with factors that determine whether substitution or elimination will occur for a given reaction.
Reduction reactions

This document discusses reduction reactions and reducing agents. It aims to teach the reader to: 1) exploit differences in reactivity between hydride and neutral reducing agents to achieve chemoselective reductions; 2) use substrate chirality to control syn vs. anti diastereoselectivity in ketone reductions; 3) rationalize reaction outcomes using transition state diagrams; 4) appreciate the versatility of transition metals in reductions; 5) understand the utility of dissolving metal reductions; and 6) use radical chemistry for deoxygenation and halide reduction. It then provides details on various hydride and neutral reducing agents, focusing on their reactivities and applications in selective reductions.
Electrophilic Substitution Reaction

The document discusses different types of electrophilic substitution reactions: SE1, SE2, and SEi. SE1 reactions follow first-order kinetics and involve two steps - rate-determining ionization and fast combination. SE2 reactions also follow first-order kinetics, but occur in a single step through a transition state. SE2 reactions can result in retention or inversion of configuration. SEi reactions are concerted mechanisms where the electrophile assists in removing the leaving group, leading to retention of configuration.
Acids, bases & aromaticity

This document provides information on acids, bases, and aromaticity. It defines acids and bases according to Arrhenius, Bronsted-Lowry, and Lewis theories. Acids are substances that produce H+ ions or accept electron pairs, while bases produce OH- ions or donate electron pairs. The document discusses factors that determine acid and base strength such as conjugate base stability, bond strength, resonance, induction, and hybridization effects. It also provides examples of acid-base reactions and uses pKa values to predict reaction equilibrium and relative acidities.
Organic reaction mechanism

The document discusses different types of substitution reactions including nucleophilic substitution, electrophilic substitution, and free radical substitution. It provides details on the mechanisms, kinetics, stereochemistry and factors affecting the rate of nucleophilic substitution reactions SN1 and SN2. SN1 follows a unimolecular mechanism involving a carbocation intermediate while SN2 follows a bimolecular mechanism with a single concerted transition state. The document also discusses electrophilic aromatic substitution reactions and addition and elimination reactions of alkenes and alkynes.
Migratory aptitudes in rearrangement reaction

The document discusses migratory aptitude in rearrangement reactions. It defines migratory aptitude as the relative ability of a migrating group to migrate in a rearrangement reaction. Factors that affect migratory aptitude include the stability of the carbocation formed and the electron density of the migrating group. Aryl groups generally have higher migratory aptitude than alkyl groups. The document also describes different types of rearrangement reactions and the mechanisms of nucleophilic rearrangements.
Chemical kinetics I

This presentation is based on the Topic " Chemical Kinetics" for the Chemistry undergraduate students of Mumbai University
Nonclassical Carbocation

This document discusses classical and nonclassical carbocations. Nonclassical carbocations have charge delocalization from neighboring bonds like C=C pi bonds. The main difference is that classical carbocations have charge localized on one carbon, while nonclassical carbocations have charge delocalized by double or single bonds not in the allylic position. Examples like the norbornyl carbocation are given to show how neighboring double bonds can stabilize and delocalize charge through 3-center bonds. Reaction rates and product stereochemistry provide evidence for nonclassical intermediates. While some challenged this view, most chemists accept nonclassical interpretations of carbocation reactions.
Aromatic electrophilic substitution

This document provides an overview of aromatic electrophilic substitution reactions (AES). It defines important terms like arenium ions, electrophiles, nucleophiles and discusses the effects of substituents on reactivity. The mechanisms of common AES reactions like nitration, sulfonation, Friedel-Crafts alkylation and acylation are covered. The document also discusses topics like the mesomeric and inductive effects of substituents, the synthesis of tribromobenzene, and the relative reactivities of benzene and substituted benzenes in bromination. Examples of AES on phenols, xylenes, cresols and other aromatic compounds are provided.
Activating and deactivating group

Activating groups donate electrons to the benzene ring through resonance or inductive effects, increasing its electron density and reactivity toward electrophilic substitution. Common ortho-para directing and activating groups include -NH2, -NHR, -O-, -OH, -OCH3, and -CH3. Deactivating groups withdraw electrons from the benzene ring through resonance or inductive effects, decreasing its electron density and reactivity. Common meta directing and deactivating groups are halogens (-Cl, -Br) and nitro (-NO2). The direction and degree of activation or deactivation depends on whether the resonance or inductive effects dominate for a particular substituent.
Neighbouring Group Participation.pptx

(i) Non-classical carbocations display delocalization of sigma bonds through 3-center-2-electron bonds in bridged systems. Neighboring group participation can assist reactions by donating electrons through lone pairs, pi bonds, aromatic rings, or sigma bonds.
(ii) The pinacol-pinacolone rearrangement involves the migration of an alkyl group from one carbon to another after the loss of a leaving group from a vicinal diol. The migration is assisted by delocalization of the carbocation intermediate onto the oxygen atom.
(iii) In asymmetrical glycols, the group with greater ability for carbocation delocalization, such as phenyl, will migrate preferentially over
Factors affecting stability of metal complexes

1. The stability of metal complexes is affected by factors such as the nature of the central metal ion, the coordinating ligand, and the presence of ring structures. The charge, size, and ionization energy of the metal ion influence stability, as do the size, charge, and basic strength of the ligand.
2. Chelation, where a multidentate ligand bonds to the metal ion at multiple sites, generally enhances stability. Five-membered rings formed by chelation are most stable due to reduced strain. An increase in the number of chelate rings or delocalized π-electrons in ring structures also increases stability.
3. Other factors like forced ligand configurations, the solvent environment, and
Addition reaction sm

Addition reactions occur when two reactants combine to form a new product with no leftover atoms. In an addition reaction, new groups are added to the starting material, breaking a pi bond and forming two sigma bonds. Addition reactions involve the addition of electrophiles, radicals, or nucleophiles across multiple bonds such as carbon-carbon double or triple bonds.
Organic Acid and Bases - Swathi S Rao

- An organic acid is an organic compound that contains a carboxyl group (-COOH) which gives it acidic properties. The strength of an organic acid depends on the stability of its conjugate base.
- Organic bases usually contain nitrogen atoms that can accept protons. Common organic bases include amines, which have a lone pair of electrons on the nitrogen that can accept protons.
- Acid strength is determined by the amount of hydronium (H3O+) ions produced when the acid dissociates in water. Base strength is determined by the amount of hydroxide (OH-) ions produced upon dissolution in water. Factors like bond strength, stability of the conjugate base, and inductive effects influence
Electrophilic Substitution reactions

This document provides an overview of electrophilic substitution reactions. It defines electrophilic substitution as a reaction where a functional group is attached to a compound by replacing another functional group, often a hydrogen atom. It describes two main types: electrophilic aromatic substitution reactions, where an atom attached to an aromatic ring is replaced; and electrophilic aliphatic substitution reactions, where hydrogen in an aliphatic compound is usually replaced. The document also outlines the three step mechanism of electrophilic substitution reactions: 1) generating an electrophile, 2) forming a carbocation, and 3) eliminating a proton to restore aromaticity.
Demjanov rearrangement

The document summarizes the Tiffeneau–Demjanov rearrangement reaction. It was discovered in the early 1900s by French chemist Marc Émile Pierre Adolphe Tiffeneau and Russian chemist Nikolay Yakovlevich Demyanov. The reaction involves treating 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone through a 1,2-carbon migration. This ring expansion reaction is useful for increasing the size of amino-substituted cyclic compounds from four to eight-membered rings. The mechanism involves diazotization of the amine to form a diazonium ion that undergoes 1,2-alkyl shift accompanied by nitrogen loss to form
What's hot (20)
Similar to Inductive effects
Solvation effects on reactions

Solvation can be defined as any stabilizing interaction of a solute (or solute moiety) and the solvent. These interactions can be weak, purely electrostatic, as is the case with non-polar solutes and solvents, or more significant, involving the interactions between dipole moments or between dipoles and formal charges.
Contributed by: Anton S. Klimenko (Undergraduate), Department of Chemistry, The University of Utah, 2016
ch1_Basic_concepts_Organic_sem1.ppt

This document discusses various concepts related to organic chemistry including carbocations, carbocation stability, inductive effects, mesomeric effects, hyperconjugation, conjugation, and resonance. It explains that carbocations are carbon-containing species with a positive formal charge that are classified based on the number of carbons bonded to the charged carbon. Tertiary carbocations are the most stable due to effects such as hyperconjugation and inductive/mesomeric effects of alkyl groups that stabilize the positive charge. Inductive and mesomeric effects involve polarization of bonds that can stabilize or destabilize carbocations. Hyperconjugation and conjugation further contribute to stability through resonance structures.
Unit 1 State of matter.pptx

Forces of attraction determine the different states of matter. Intramolecular forces like ionic bonds and covalent bonds occur within molecules. Intermolecular forces like van der Waals forces, hydrogen bonding, and London dispersion forces occur between molecules. These intermolecular forces are responsible for solids, liquids, and gases having distinct properties based on the strength of attraction between molecules. In solids, strong intermolecular forces create a rigid structure. In liquids, weaker intermolecular forces allow molecules to flow freely past one another. In gases, intermolecular forces are very weak, causing molecules to move randomly in all directions.
A friedel crafts alkylation love x love begin from here

This document discusses how substituents influence the reactivity of aromatic rings towards electrophilic substitution. It describes two main effects - inductive and resonance effects. Inductive effects result from differences in electronegativity between substituents and hydrogen, withdrawing or donating electrons. Resonance effects occur when substituents can conjugate with the aromatic ring, again donating or withdrawing electrons. Electron-donating groups increase reactivity through resonance, while electron-withdrawing groups decrease it, usually due to stronger inductive effects outweighing resonance donation. Substituents are classified as activating or deactivating depending on their net effect.
chapter_11au.ppt

The document discusses intermolecular forces, which are the attractions between molecules. It describes the different types of intermolecular forces - dipole-dipole interactions, hydrogen bonding, and London dispersion forces. These intermolecular forces are weaker than intramolecular bonds but still influence physical properties like boiling points. The document also discusses how states of matter depend on a balance between kinetic energy and intermolecular attractions. It examines properties related to intermolecular forces like vapor pressure, heat of fusion/vaporization, and phase diagrams.
An overview of organic reaction mechanisms

The document provides an overview of organic reaction mechanisms. It discusses the main types of organic reactions - substitution, addition, elimination, and rearrangement - and describes common reaction intermediates and steps. Reaction mechanisms are explained including radical, polar, nucleophilic, and electrophilic reactions. Activating and deactivating groups are described along with their inductive and mesomeric effects.
Reaction mechanisms

This document discusses organic reactions and mechanisms. It defines key terms like substrate, reagent, products and defines the steps in a reaction mechanism. It describes factors that influence reactions like inductive and resonance effects. It also discusses types of reaction intermediates like carbonium ions, carbanions and free radicals. The document outlines different types of organic reactions including substitution, addition, elimination and rearrangement. It provides details on the mechanism of substitution reactions including free radical, electrophilic and nucleophilic substitution reactions.
Reaction mechanisms

This document discusses organic reactions and mechanisms. It defines key terms like substrate, reagent, products, and mechanism. It describes how factors like inductive and mesomeric effects can influence reactions by altering electron density. It also discusses different types of reaction intermediates that can form, such as carbonium ions, carbanions, free radicals, and carbenes. The document classifies reagents as electrophiles or nucleophiles and describes their behaviors. It explains concepts like activation energy and the transition state that systems must go through for a reaction to occur.
Reaction.pptx

This document discusses structural theory in organic chemistry. It covers topics like bond fission and its types (homolysis and heterolysis), organic reagents (electrophiles, nucleophiles, free radicals), types of organic reactions (substitution, elimination, addition, rearrangement), inductive effect and its applications, and mesomeric effect and its applications. Examples are provided to illustrate key concepts. Essay and short answer questions are also included at the end to test understanding of topics covered.
Therotical Organic Chemistry.pptx

This document discusses structural theory in organic chemistry. It covers topics like bond fission and its types (homolysis and heterolysis), organic reagents (electrophiles, nucleophiles, free radicals), types of organic reactions (substitution, elimination, addition, rearrangement), inductive effect and its applications, and mesomeric effect and its applications. Bond fission can occur through homolysis, where the electron pair is split equally forming neutral radicals, or heterolysis where one atom takes both electrons forming charged species like carbocations or carbanions. The document defines different organic reagents and reactions. It also explains inductive and mesomeric effects which influence molecular stability and reactivity in organic compounds.
LGIS-Theme 1a.pptx

This document provides an overview of organic chemistry concepts including:
- The origin of organic chemistry as the study of compounds from living organisms.
- The role of carbon in organic compounds and their importance to human biology.
- Principles of atomic structure including electron shells and orbitals.
- Bond formation through sharing or transferring electrons to attain stable configurations.
- Drawing Lewis structures and accounting for formal charges and resonance.
- Molecular geometry from hybrid atomic orbitals and how this relates to bond angles.
- Intermolecular forces between molecules and various effects that influence molecular properties.
reactivity in chemisorption and catalysis of monometallic particles

This document discusses the properties and reactivity of nanoparticles for catalysis applications. It notes that nanoparticles have a high surface area to volume ratio, which allows them to be more reactive. The physical and electronic properties, including reactivity, of particles varies significantly as size is reduced to the nanoscale. Studies have shown that properties like chemisorption and catalytic activity depend on particle size and number of atoms. The reactivity of very small clusters, even when supported, relates to their distinct electronic structure based on atom number. Particle shape, support interactions, and alloy composition also influence catalytic properties at the nanoscale.
🔬 Need Expert Chemistry Assignment Help? We've Got You Covered! 🔬

Stuck on tricky chemistry assignments? Your search ends here! ChemistryAssignmentHelp.com offers specialized assistance in solving your chemistry assignments with precision and expertise. Our team of experienced PhD-level professionals ensures accurate solutions for diverse topics, from equations to lab reports. With a commitment to originality and confidentiality, we guarantee plagiarism-free work and data security. Say farewell to assignment anxiety and hello to top-notch grades! Visit https://www.chemistryassignmenthelp.com/ to submit your assignments and experience a new level of academic success in chemistry. 📚🧪📊
chap2_orgChem.ppt

This document discusses polar covalent bonds and acid-base chemistry. It introduces electronegativity and how differences in electronegativity between atoms leads to polar covalent bonds. Bond polarity can be quantified using dipole moments. Resonance structures are discussed as ways to represent delocalized bonding. Brønsted-Lowry acids and bases are defined as proton donors and acceptors. Acid strength is quantified using acidity constants (Ka) and their logarithmic form, pKa values. Organic acids and bases are introduced. Lewis acids and bases are also defined in terms of electron pair acceptance and donation.
Basic rxns in org chem

Organic reactions occur between organic molecules containing carbon and hydrogen. There are several types of organic reactions including addition, elimination, substitution, and rearrangement. Organic reactions are also classified by reaction type such as acid-base reactions and redox reactions. Reactions proceed through the formation of unstable intermediates like carbocations, carbanions, free radicals, and radical ions before products form. Factors like energetics, electronic effects, steric effects, stereoelectronic effects, solvent effects influence organic reactions. Reactions require activation energy to reach a transition state before products form.
Inductive effect notes

Inductive effect refers to the unequal sharing of electrons in a molecule due to electron-withdrawing or electron-donating groups, creating a permanent dipole. Electron-withdrawing groups (-I effect) increase acidity by stabilizing the conjugate base, while electron-donating groups (+I effect) decrease acidity by destabilizing the conjugate base. The inductive effect also influences the stability and basicity of molecules.
Gardner_OzonePosterAGU2016

The document summarizes an experiment investigating how photon energy affects isotopic ratios during ozone formation. Oxygen gas was exposed to light from mercury, hydrogen, and planned krypton lamps at different energies, and the resulting ozone was analyzed. Preliminary results found enrichment of 17O and 18O isotopes consistent with literature. Reducing a step in the Chapman cycle reaction had no effect. Future experiments are planned to gain more insight, such as using higher energy krypton light and temperatures, to test the resonance effect hypothesis. The anomalous mass-independent isotope effect seen in ozone formation still lacks a full explanation after decades of research.
Electron Displacement Effect

This document summarizes different types of electron displacement effects including permanent polarization effects like inductive effect, mesomeric effect, and hyperconjugation, as well as temporary polarization effects like electromeric effect. It describes the inductive effect as the displacement of electron density in a covalent bond towards the more electronegative atom. The mesomeric effect involves resonance stabilization of molecules through pi electron delocalization. Hyperconjugation refers to sigma electron delocalization between C-H bonds and adjacent pi systems. The electromeric effect temporarily polarizes multiple bonds during reaction with electrophiles.
Types of Organic Reactions

Organic reactions are chemical reactions involving organic compounds. Organic reactions are used in the construction of new organic molecules. The production of many man-made chemicals such as drugs, plastics, food additives, fabrics depend on organic reactions.
Intermolecular Forces and Physical Properties lesson 2.pptx

The document discusses intermolecular forces and how they determine the physical properties of compounds. It describes the different types of intermolecular forces - dipole-dipole interactions, hydrogen bonding, and London dispersion forces. Compounds with stronger intermolecular forces like hydrogen bonding will have higher melting and boiling points. The document uses examples of isomers and similar compounds to illustrate how small differences in molecular structure can significantly impact physical properties due to varying intermolecular interactions.
Similar to Inductive effects (20)
A friedel crafts alkylation love x love begin from here

A friedel crafts alkylation love x love begin from here
reactivity in chemisorption and catalysis of monometallic particles

reactivity in chemisorption and catalysis of monometallic particles
🔬 Need Expert Chemistry Assignment Help? We've Got You Covered! 🔬

🔬 Need Expert Chemistry Assignment Help? We've Got You Covered! 🔬
Intermolecular Forces and Physical Properties lesson 2.pptx

Intermolecular Forces and Physical Properties lesson 2.pptx
More from Daniel Morton
CH Functionalizations on Electron-Deficient Aromatics in the synthesis of Pi-...

This document discusses the use of C-H functionalization catalysis to incorporate electron-deficient moieties into conjugated materials without needing to first install reactive halides or stannanes. Electron-deficient moieties are important for electronic materials as they can tune optical properties and transport characteristics. Direct C-H functionalization avoids harsh halogenation steps and bypasses instability issues of stannanes. Examples are given of one-pot syntheses using this approach to obtain up to 96% yields of differentially substituted electron-accepting building blocks. Challenges include controlling reaction selectivity when multiple C-H bonds on a moiety could potentially react.
C–H bond hydroxylation at non heme carboxylate-bridged diiron centers

This unit provides an overview of how Nature has inspired the development of novel diiron bridged complexes for use in C–H Hydroxylation.
Contributed by Omar Villanueva and Cora MacBeth, Emory University, 2014
Collagen triple helix stability

Collagen is made up of the repeating pattern Glycine-X-Y, where X and Y are commonly L-proline (Pro) and 4(R)-hydroxy-l-proline (Hyp), respectively. By substituting X and Y with a fluorine probe, stereoelectronic effects can be observed and compared to the effects of hydrogen bonding which has been predicted to be the main contributor to the collagen triple helix strength.
Contributed by: Alexandra Zudova, Samuel Broadbent (Undergraduates), University of Utah, 2013
Bio inspired metal-oxo catalysts for c–h bond functionalization

Metal-oxo complexes are important species in the activation of strong C–H bonds in biological systems. The high reactivity of metal-oxo complexes results from the way their valence electrons are arranged, and this arrangement depends strongly on the geometry around the metal center.
Contributed by: A.S. Borovik and Sarah Cook, University of California-Irvine, 2014
Fischer and schrock carbenes

A carbene is any neutral carbon species which contains a non-bonding valance pair of electrons.
Contributed by Alison Brown & Nathan Buehler, Undergraduates, University of Utah
Hammett parameters

The document discusses the Hammett plot, which is a linear free-energy relationship analysis used to model the electronic effects of substituents on aromatic systems. It describes how σ values are derived from ionization reactions to indicate whether a substituent is electron-donating or electron-withdrawing. These σ values can then be used to analyze reaction mechanisms and optimize reaction conditions for similar processes. Examples are given of how split Hammett plots reveal changing mechanisms depending on the electronic nature of the substituent. Problems involving interpreting ρ values in Hammett plots to determine reaction pathways are also presented.
Aryl fluorination

Organic Pedagogical Electronic Network
Aryl Fluorination
This document summarizes aryl fluorination, an important reaction for introducing fluorine groups onto aromatic rings. It notes that two of the top 10 grossing drugs, Lipitor and Reserdal, contain aryl fluorines. The mechanism proceeds through oxidation of an aryl group to a high valent metal species, followed by transmetalation and reductive elimination to introduce the fluorine. Examples are given of stoichiometric and catalytic aryl fluorination reactions using Selectfluor and silver oxide catalysts.
Allylic strain

The allylic position is the atom bound to a double bonded atom. The substituents on the allylic carbon and the doubly bonded atoms can result in allylic strain.
Contributed by: Sophia Robinson, (Undergraduate), Physical Organic Chemistry I, CHEM 7240 (Sigman), University of Utah, 2015
1 3-dipolar-cycloadditions

A 1,3-dipolar cycloaddition is a fascinating and diverse reaction that can be used in stepwise syntheses of large molecules, such as masked aldol reactions, and has potential biological applications. While it is an incredibly useful reactions, is also has a simple mechanism.
Contributed by: Created by Alexandra Kent and Judy Zhu (Undergraduates)Edited by Margaret Hilton
Honors Organic Chemistry
University of Utah
Heck redox relay

This document summarizes work on the Heck reaction and redox-relay Heck reaction. It provides background on the Heck reaction and its applications. It then discusses chain-walking in the Heck reaction where the palladium catalyst migrates down an alkyl chain, producing alkene isomers. It introduces Sigman's work developing a redox-relay Heck reaction where chain-walking of palladium is controlled by an alcohol thermodynamic sink on the substrate, transferring unsaturation to form aldehydes or ketones. It is authored by Margaret Hilton from the Sigman Lab at the University of Utah in 2014.
Stability and reactions of n heterocyclic carbenes

1) N-heterocyclic carbenes (NHCs) are stable two-coordinate carbon compounds that were first synthesized in 1991. Their stability is due to mesomeric and inductive effects from the nitrogen atoms that make the carbenes strong π-donors and weak σ-acceptors.
2) The thermodynamics of dissociation were studied for a bis(benzimidazol-2-ylidene), finding an enthalpy of 13.7 kcal/mol and entropy of 30.4 cal/mol*K. NHCs were also ranked based on their acidity.
3) NHCs form stable complexes with transition metals and have applications in organomet
Pyridine CH functionalization

This unit provides an overview of the different strategies that have been develop to selectively functionalize the 2,3 and 4 positions of pyridine units.
Contributed by the Sarpong Group, UC-Berkeley, 2013
Brønsted catalysis

The Brønsted catalysis relationship is a Linear Free Energy Relationship (LFER) that relates ionization of an acid or base which catalyzes a reaction and the rate of the reaction.
Contributed by: Quincy Davis, Jonathan Greenhalgh, Joshua Visser (Undergraduates), University of Utah, 2016
Quantum Tunneling in Organic Chemistry

Tunneling is a phenomenon of Quantum Mechanics in which particles, with a small amount of probability, are able to “tunnel” or travel through a large, finite potential energy (PE) barrier instead of traveling over the barrier as Classic Mechanics dictates should occur.
Contributed by: Mark O’Dair, Undergraduate, University of Utah, 2014
The Prins-Pinacol Reaction

The Prins-Pinacol reaction is a two-step process that begins with the Prins reaction, where an alkene attacks an aldehyde activated by a Lewis acid to form a cationic intermediate. This is followed by a pinacol rearrangement, where a methyl shift pushes the cation onto an oxygen. The reaction forms complex natural product backbones and allows stereoselective tetrahydropyran synthesis. Driving forces include increased stability of the carbocation intermediate and relief of ring strain. The Lewis acid activates the aldehyde for attack.
Solvation Effects on pKa Values

When substrates are put in solution, the solvent molecules can organize themselves around a charged species to stabilize it. Solvents can stabilize a charge most effectively when the charge on the substrate is easy to get to.
Contributed by: Jamie Allen, Jacqueline Pasek-Allen, Sarah Lefave (Undergraduates), University of Utah, 2016
Hyperconjugation

Hyperconjugation is the donation of a sigma bond into an adjacent empty or partially filled p orbital, which results in an increased stability of the molecule.
Contributed by: Samuel Redstone (Undergraduate), University of Utah, 2016
Hammett Plots in the World of Enzymes

A substituent effect is the change in a molecule’s reactivity when a substituent on the molecule is changed. In 1935, Louis Hammett designed a scale to measure influence of various substituents (X) at the meta- or para- positions on the acidity of benzoic acid.
Contributed by: Erika Aoyama and Megan Browning, University of Utah, 2016
Anomeric effect

The anomeric effect was discovered in 1955 with the work of J.T. Edward, N.-J. Chu, and R.U. Lemieux.
Contributed by: Cody F. Bender, Charles E. Price (Undergraduates), University of Utah, 2016
Marcus theory

An overview of the use of the Marcus Theory to calculate the energies of transition states.
Contributed by: Elizabeth Greenhalgh, Amanda Bischoff, and Matthew Sigman, University of Utah, 2015
More from Daniel Morton (20)
CH Functionalizations on Electron-Deficient Aromatics in the synthesis of Pi-...

CH Functionalizations on Electron-Deficient Aromatics in the synthesis of Pi-...
C–H bond hydroxylation at non heme carboxylate-bridged diiron centers

C–H bond hydroxylation at non heme carboxylate-bridged diiron centers
Bio inspired metal-oxo catalysts for c–h bond functionalization

Bio inspired metal-oxo catalysts for c–h bond functionalization
Stability and reactions of n heterocyclic carbenes

Stability and reactions of n heterocyclic carbenes
Recently uploaded
Applied Science: Thermodynamics, Laws & Methodology.pdf

When I was asked to give a companion lecture in support of ‘The Philosophy of Science’ (https://shorturl.at/4pUXz) I decided not to walk through the detail of the many methodologies in order of use. Instead, I chose to employ a long standing, and ongoing, scientific development as an exemplar. And so, I chose the ever evolving story of Thermodynamics as a scientific investigation at its best.
Conducted over a period of >200 years, Thermodynamics R&D, and application, benefitted from the highest levels of professionalism, collaboration, and technical thoroughness. New layers of application, methodology, and practice were made possible by the progressive advance of technology. In turn, this has seen measurement and modelling accuracy continually improved at a micro and macro level.
Perhaps most importantly, Thermodynamics rapidly became a primary tool in the advance of applied science/engineering/technology, spanning micro-tech, to aerospace and cosmology. I can think of no better a story to illustrate the breadth of scientific methodologies and applications at their best.
Pests of Storage_Identification_Dr.UPR.pdf

InIndia-post-harvestlosses-unscientificstorage,insects,rodents,micro-organismsetc.,accountforabout10percentoftotalfoodgrains
Graininfestation
Directdamage
Indirectly
•theexuviae,skin,deadinsects
•theirexcretawhichmakefoodunfitforhumanconsumption
About600speciesofinsectshavebeenassociatedwithstoredgrainproducts
100speciesofinsectpestsofstoredproductscauseeconomiclosses
Sexuality - Issues, Attitude and Behaviour - Applied Social Psychology - Psyc...

A proprietary approach developed by bringing together the best of learning theories from Psychology, design principles from the world of visualization, and pedagogical methods from over a decade of training experience, that enables you to: Learn better, faster!
Direct Seeded Rice - Climate Smart Agriculture

Direct Seeded Rice - Climate Smart AgricultureInternational Food Policy Research Institute- South Asia Office
PPT on Direct Seeded Rice presented at the three-day 'Training and Validation Workshop on Modules of Climate Smart Agriculture (CSA) Technologies in South Asia' workshop on April 22, 2024.
ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...

ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...Advanced-Concepts-Team
Presentation in the Science Coffee of the Advanced Concepts Team of the European Space Agency on the 07.06.2024.
Speaker: Diego Blas (IFAE/ICREA)
Title: Gravitational wave detection with orbital motion of Moon and artificial
Abstract:
In this talk I will describe some recent ideas to find gravitational waves from supermassive black holes or of primordial origin by studying their secular effect on the orbital motion of the Moon or satellites that are laser ranged.waterlessdyeingtechnolgyusing carbon dioxide chemicalspdf

The technology uses reclaimed CO₂ as the dyeing medium in a closed loop process. When pressurized, CO₂ becomes supercritical (SC-CO₂). In this state CO₂ has a very high solvent power, allowing the dye to dissolve easily.
Immersive Learning That Works: Research Grounding and Paths Forward

We will metaverse into the essence of immersive learning, into its three dimensions and conceptual models. This approach encompasses elements from teaching methodologies to social involvement, through organizational concerns and technologies. Challenging the perception of learning as knowledge transfer, we introduce a 'Uses, Practices & Strategies' model operationalized by the 'Immersive Learning Brain' and ‘Immersion Cube’ frameworks. This approach offers a comprehensive guide through the intricacies of immersive educational experiences and spotlighting research frontiers, along the immersion dimensions of system, narrative, and agency. Our discourse extends to stakeholders beyond the academic sphere, addressing the interests of technologists, instructional designers, and policymakers. We span various contexts, from formal education to organizational transformation to the new horizon of an AI-pervasive society. This keynote aims to unite the iLRN community in a collaborative journey towards a future where immersive learning research and practice coalesce, paving the way for innovative educational research and practice landscapes.
Compexometric titration/Chelatorphy titration/chelating titration

Classification
Metal ion ion indicators
Masking and demasking reagents
Estimation of Magnisium sulphate
Calcium gluconate
Complexometric Titration/ chelatometry titration/chelating titration, introduction, Types-
1.Direct Titration
2.Back Titration
3.Replacement Titration
4.Indirect Titration
Masking agent, Demasking agents
formation of complex
comparition between masking and demasking agents,
Indicators/Metal ion indicators/ Metallochromic indicators/pM indicators,
Visual Technique,PM indicators (metallochromic), Indicators of pH, Redox Indicators
Instrumental Techniques-Photometry
Potentiometry
Miscellaneous methods.
Complex titration with EDTA.
Describing and Interpreting an Immersive Learning Case with the Immersion Cub...

Current descriptions of immersive learning cases are often difficult or impossible to compare. This is due to a myriad of different options on what details to include, which aspects are relevant, and on the descriptive approaches employed. Also, these aspects often combine very specific details with more general guidelines or indicate intents and rationales without clarifying their implementation. In this paper we provide a method to describe immersive learning cases that is structured to enable comparisons, yet flexible enough to allow researchers and practitioners to decide which aspects to include. This method leverages a taxonomy that classifies educational aspects at three levels (uses, practices, and strategies) and then utilizes two frameworks, the Immersive Learning Brain and the Immersion Cube, to enable a structured description and interpretation of immersive learning cases. The method is then demonstrated on a published immersive learning case on training for wind turbine maintenance using virtual reality. Applying the method results in a structured artifact, the Immersive Learning Case Sheet, that tags the case with its proximal uses, practices, and strategies, and refines the free text case description to ensure that matching details are included. This contribution is thus a case description method in support of future comparative research of immersive learning cases. We then discuss how the resulting description and interpretation can be leveraged to change immersion learning cases, by enriching them (considering low-effort changes or additions) or innovating (exploring more challenging avenues of transformation). The method holds significant promise to support better-grounded research in immersive learning.
EWOCS-I: The catalog of X-ray sources in Westerlund 1 from the Extended Weste...

Context. With a mass exceeding several 104 M⊙ and a rich and dense population of massive stars, supermassive young star clusters
represent the most massive star-forming environment that is dominated by the feedback from massive stars and gravitational interactions
among stars.
Aims. In this paper we present the Extended Westerlund 1 and 2 Open Clusters Survey (EWOCS) project, which aims to investigate
the influence of the starburst environment on the formation of stars and planets, and on the evolution of both low and high mass stars.
The primary targets of this project are Westerlund 1 and 2, the closest supermassive star clusters to the Sun.
Methods. The project is based primarily on recent observations conducted with the Chandra and JWST observatories. Specifically,
the Chandra survey of Westerlund 1 consists of 36 new ACIS-I observations, nearly co-pointed, for a total exposure time of 1 Msec.
Additionally, we included 8 archival Chandra/ACIS-S observations. This paper presents the resulting catalog of X-ray sources within
and around Westerlund 1. Sources were detected by combining various existing methods, and photon extraction and source validation
were carried out using the ACIS-Extract software.
Results. The EWOCS X-ray catalog comprises 5963 validated sources out of the 9420 initially provided to ACIS-Extract, reaching a
photon flux threshold of approximately 2 × 10−8 photons cm−2
s
−1
. The X-ray sources exhibit a highly concentrated spatial distribution,
with 1075 sources located within the central 1 arcmin. We have successfully detected X-ray emissions from 126 out of the 166 known
massive stars of the cluster, and we have collected over 71 000 photons from the magnetar CXO J164710.20-455217.
Micronuclei test.M.sc.zoology.fisheries.

Current Ms word generated power point presentation covers major details about the micronuclei test. It's significance and assays to conduct it. It is used to detect the micronuclei formation inside the cells of nearly every multicellular organism. It's formation takes place during chromosomal sepration at metaphase.
8.Isolation of pure cultures and preservation of cultures.pdf

Isolation of pure culture, its various method.
11.1 Role of physical biological in deterioration of grains.pdf

Storagedeteriorationisanyformoflossinquantityandqualityofbio-materials.
Themajorcausesofdeteriorationinstorage
•Physical
•Biological
•Mechanical
•Chemical
Storageonlypreservesquality.Itneverimprovesquality.
Itisadvisabletostartstoragewithqualityfoodproduct.Productwithinitialpoorqualityquicklydepreciates
The debris of the ‘last major merger’ is dynamically young

The Milky Way’s (MW) inner stellar halo contains an [Fe/H]-rich component with highly eccentric orbits, often referred to as the
‘last major merger.’ Hypotheses for the origin of this component include Gaia-Sausage/Enceladus (GSE), where the progenitor
collided with the MW proto-disc 8–11 Gyr ago, and the Virgo Radial Merger (VRM), where the progenitor collided with the
MW disc within the last 3 Gyr. These two scenarios make different predictions about observable structure in local phase space,
because the morphology of debris depends on how long it has had to phase mix. The recently identified phase-space folds in Gaia
DR3 have positive caustic velocities, making them fundamentally different than the phase-mixed chevrons found in simulations
at late times. Roughly 20 per cent of the stars in the prograde local stellar halo are associated with the observed caustics. Based
on a simple phase-mixing model, the observed number of caustics are consistent with a merger that occurred 1–2 Gyr ago.
We also compare the observed phase-space distribution to FIRE-2 Latte simulations of GSE-like mergers, using a quantitative
measurement of phase mixing (2D causticality). The observed local phase-space distribution best matches the simulated data
1–2 Gyr after collision, and certainly not later than 3 Gyr. This is further evidence that the progenitor of the ‘last major merger’
did not collide with the MW proto-disc at early times, as is thought for the GSE, but instead collided with the MW disc within
the last few Gyr, consistent with the body of work surrounding the VRM.
Recently uploaded (20)
Applied Science: Thermodynamics, Laws & Methodology.pdf

Applied Science: Thermodynamics, Laws & Methodology.pdf
Sexuality - Issues, Attitude and Behaviour - Applied Social Psychology - Psyc...

Sexuality - Issues, Attitude and Behaviour - Applied Social Psychology - Psyc...
Juaristi, Jon. - El canon espanol. El legado de la cultura española a la civi...

Juaristi, Jon. - El canon espanol. El legado de la cultura española a la civi...
ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...

ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...
waterlessdyeingtechnolgyusing carbon dioxide chemicalspdf

waterlessdyeingtechnolgyusing carbon dioxide chemicalspdf
Immersive Learning That Works: Research Grounding and Paths Forward

Immersive Learning That Works: Research Grounding and Paths Forward
GBSN - Biochemistry (Unit 6) Chemistry of Proteins

GBSN - Biochemistry (Unit 6) Chemistry of Proteins
Compexometric titration/Chelatorphy titration/chelating titration

Compexometric titration/Chelatorphy titration/chelating titration
Describing and Interpreting an Immersive Learning Case with the Immersion Cub...

Describing and Interpreting an Immersive Learning Case with the Immersion Cub...
EWOCS-I: The catalog of X-ray sources in Westerlund 1 from the Extended Weste...

EWOCS-I: The catalog of X-ray sources in Westerlund 1 from the Extended Weste...
8.Isolation of pure cultures and preservation of cultures.pdf

8.Isolation of pure cultures and preservation of cultures.pdf
11.1 Role of physical biological in deterioration of grains.pdf

11.1 Role of physical biological in deterioration of grains.pdf
The debris of the ‘last major merger’ is dynamically young

The debris of the ‘last major merger’ is dynamically young
Inductive effects
- 1. Organic Pedagogical Electronic Network Inductive Effects Colin Swenson Tewoderos Ayele University of Utah
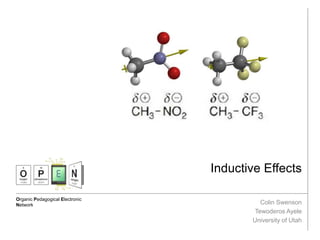
- 2. Inductive Effect Eric Anslyn and Dennis Dougherty, Modern Physical Organic Chemistry, University Science Books; pp 15-18 Overview: An inductive effect occurs when the electrons shared between two atoms or groups of different electronegativities are withdrawn to the more electronegative atom or group through the σ bond resulting in a separation of charge, or dipole. This effect can be felt a few atoms away affecting the reactivity of other groups on the molecule. The yellow arrows show the dipole moment of the molecule, indicating the direction of electron density.
- 3. Inductive Effects on Acidity Reush, W. Carboxylic Acids. https://www2.chemistry.msu.edu/faculty/reusch/VirtTxtJml/crbacid1.htm (Accessed October 2015). The increased acidity of carboxylic acids can be attributed to electrostatic effects, such as induction. More electronegative groups will greatly change the distribution of electron density in the molecule by withdrawing electrons. This effect will make the proton more acidic. The relative distance of the electronegative group also affects the acidity of carboxylic acids. The closer the electron withdrawing substituent is to the reactive proton, the more acidic the molecule becomes. Acidity increases as more electronegative substituents are added to the molecule and withdraw electron density from the acidic proton Compound pKa CH3CO2H 4.74 BrCH2CO2H 2.9 ClCH2CO2H 2.85 FCH2CO2H 2.65 CH3CH2ClCO2H 2.89 CH3ClCH2CO2H 4.05 ClCH2CH2CO2H 4.53
- 4. Case Study Bell, R., Mellot-Draznieks, C., Torrisi, A. J. Chem. Phys. 130, 194703 (2009) Inductive Effects on Aromatics for CO2 Adsorption Carbon dioxide, a side product in the production of hydrogen gas for fuel applications, needs to be separated from the gaseous mixture at very low pressures. An economic approach involves the use of Metal-Organic Framework materials. The quadrupole of CO2 interacts with the delocalized pi- system of aromatics. Substituents supplying inductive effects have been studied for enhancement of the affinity for CO2. Methyl substituents supplying a positive inductive effect are shown to stabilize the CO2-aromatic interaction, whereas halogen substituents supplying a negative inductive effect are shown to destabilize the CO2-aromatic interaction. Delocalized charge on aromatic carbons as a function of the binding energy (BE) to carbon dioxide of different substituent groups.
- 5. Problems Bell, R., Mellot-Draznieks, C., Torrisi, A. J. Chem. Phys. 130, 194703 (2009) 1. Rank the following in increasing acidity (1 is most acidic) a. 1 b. 1 c. 1 2. Explain how induction effects differ from resonance effects. 3. Provide an explanation for the trend seen in the delocalization of charge on the aromatic carbons presented in the case study. 4. Which molecule would you expect to have a greater affinity for CO2 , C6H5NO2 or C6H5NH2 ? Explain.
- 6. Solutions Bell, R., Mellot-Draznieks, C., Torrisi, A. J. Chem. Phys. 130, 194703 (2009) 1 a. b. c. 2. Resonance effects are directly involved with the movement of electrons, whereas inductive effects are a result of a change in electron density. 3. There is less delocalized charge on the aromatic carbons as halogens are placed on the ring because they withdraw electron density from the aromatic system. 4. C6H5NH2 because there will be more delocalized charge on the aromatic system due to the amine group being less withdrawing than the nitro group. 3 2 1 2 3 1 3 1 2
- 7. This work is licensed under a Creative Commons Attribution- ShareAlike 4.0 International License. Contributed by: Colin Swenson and Tewoderos Ayele (Undergraduates) University of Utah, 2015