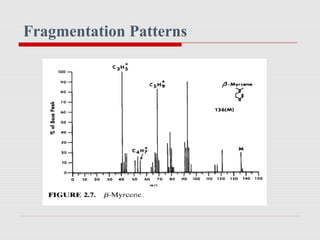
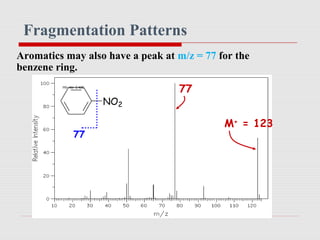
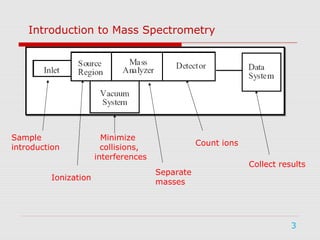
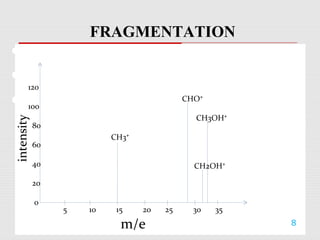
Mass spectrometry is a technique used to identify molecules based on their mass. It works by ionizing chemical compounds to generate molecular or fragment ions and measuring their mass-to-charge ratios. The document discusses the basic principles and components of a mass spectrometer, including ionization, separation of ions based on mass, and detection. It also covers common fragmentation patterns observed for different classes of compounds like hydrocarbons, alcohols, aromatics, and others. General rules for fragmentation are provided along with examples to illustrate how structural information can be determined.

















![iii)Elimination at Hydrogen sulphide (H2S)[M-34] confirms thiols
(mercaptons)
H
C
C
SH
C C + H2S
M - 34
iv)Elimination of Hydrogen cyanide (HCN)[M-27] confirms nitriles.
H
C
C
CN
C C + HCN
M - 27
20](https://image.slidesharecdn.com/massspectroscopy-140929003517-phpapp01/85/Mass-spectroscopy-20-320.jpg)



![NITROGEN RULE:-
It is used for determination of molecular mass of
compounds and its elemental composition
Molecules having odd mass number contain odd
number of nitrogen atoms.
H3C
Molecules having even mass number contain even no
H3C of nitrogen CH3
atoms.
H
MMWW == 5599
((oodddd))
MMWW == 5588
((eevveenn))
IIoonniissaattiioonn
[[MM++HH]]
[[MM++HH]]
MMWW == 6600
MMWW == 5599
CH3
N
H3C CH3
24](https://image.slidesharecdn.com/massspectroscopy-140929003517-phpapp01/85/Mass-spectroscopy-24-320.jpg)