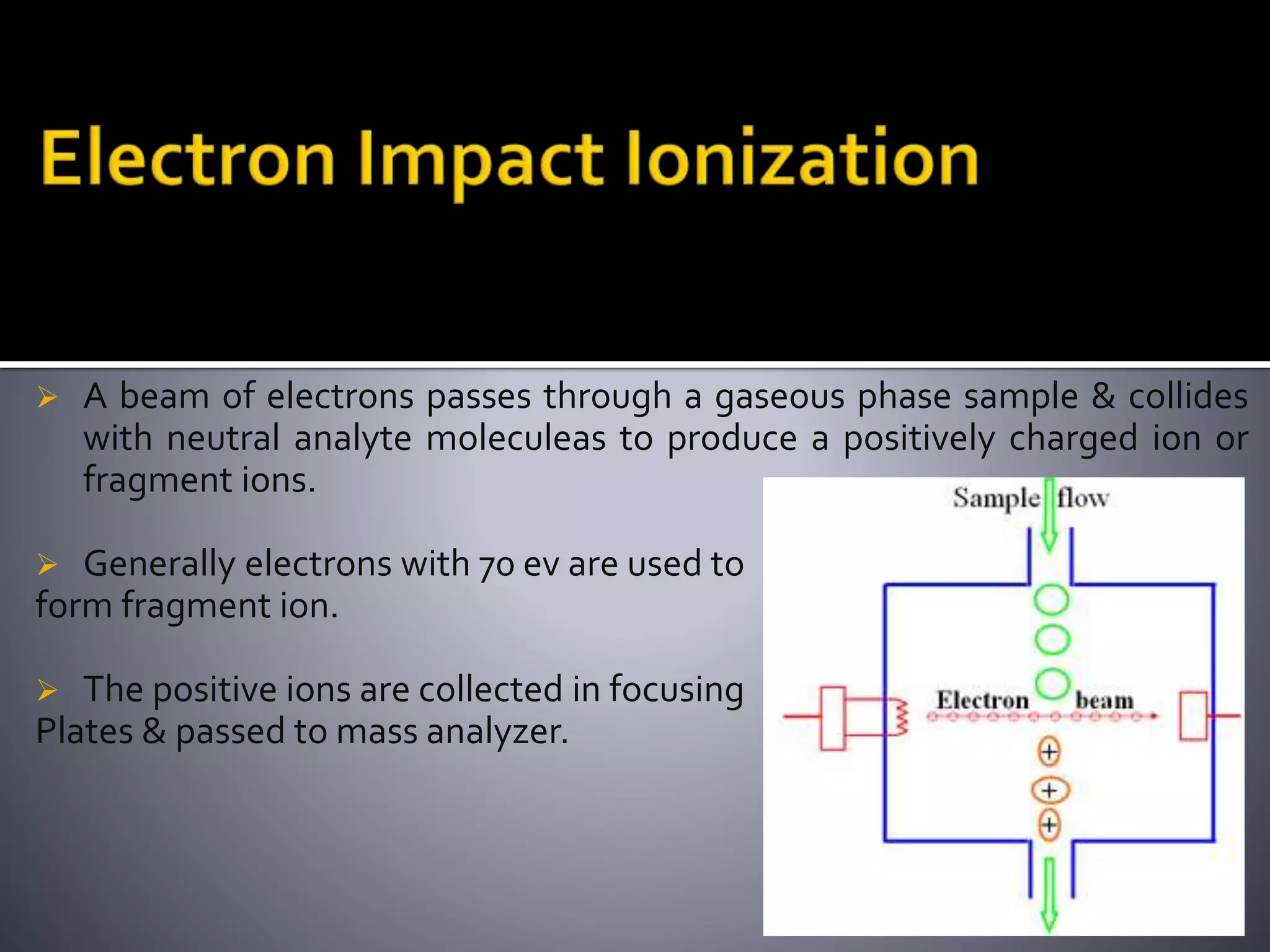
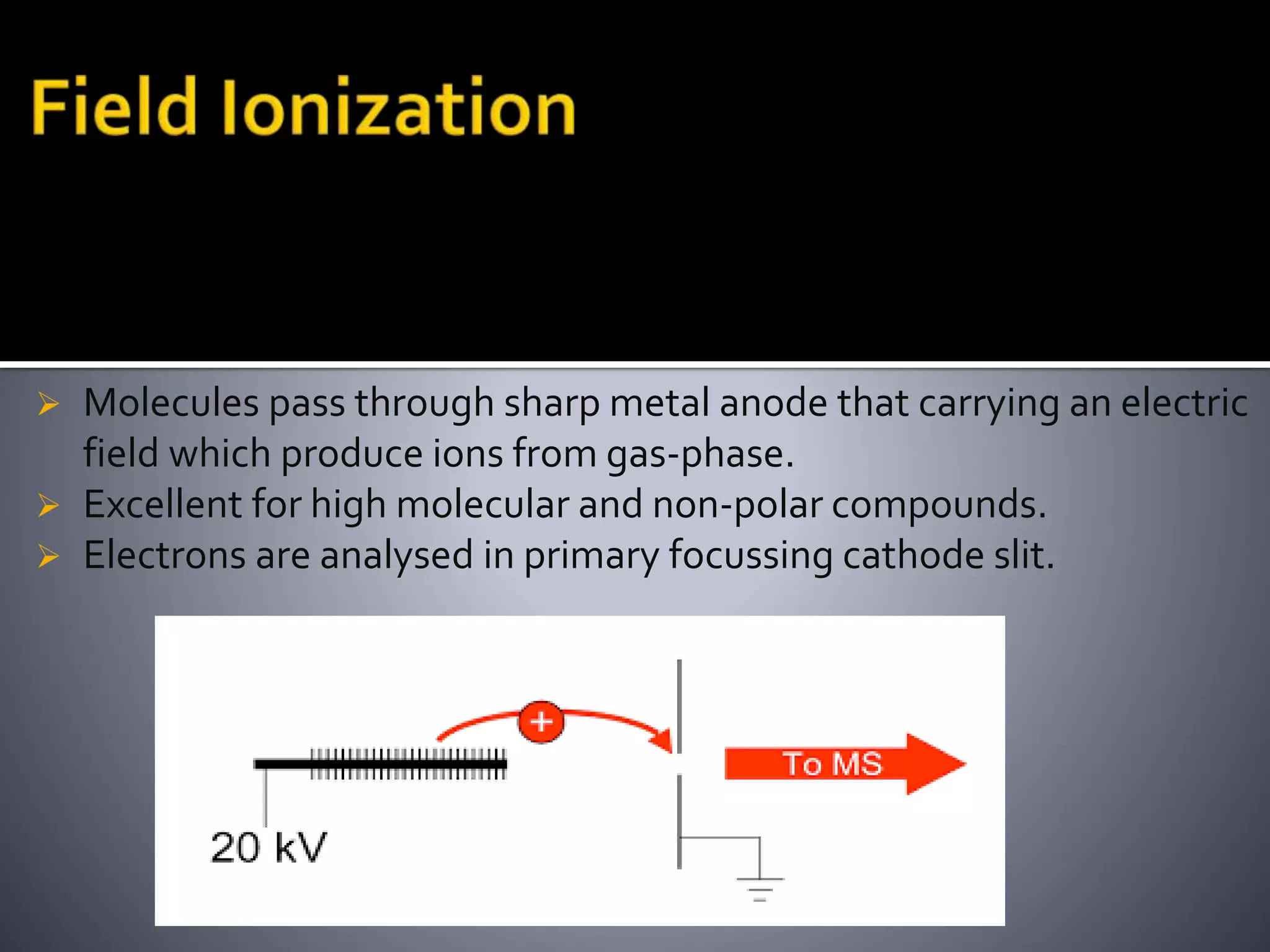
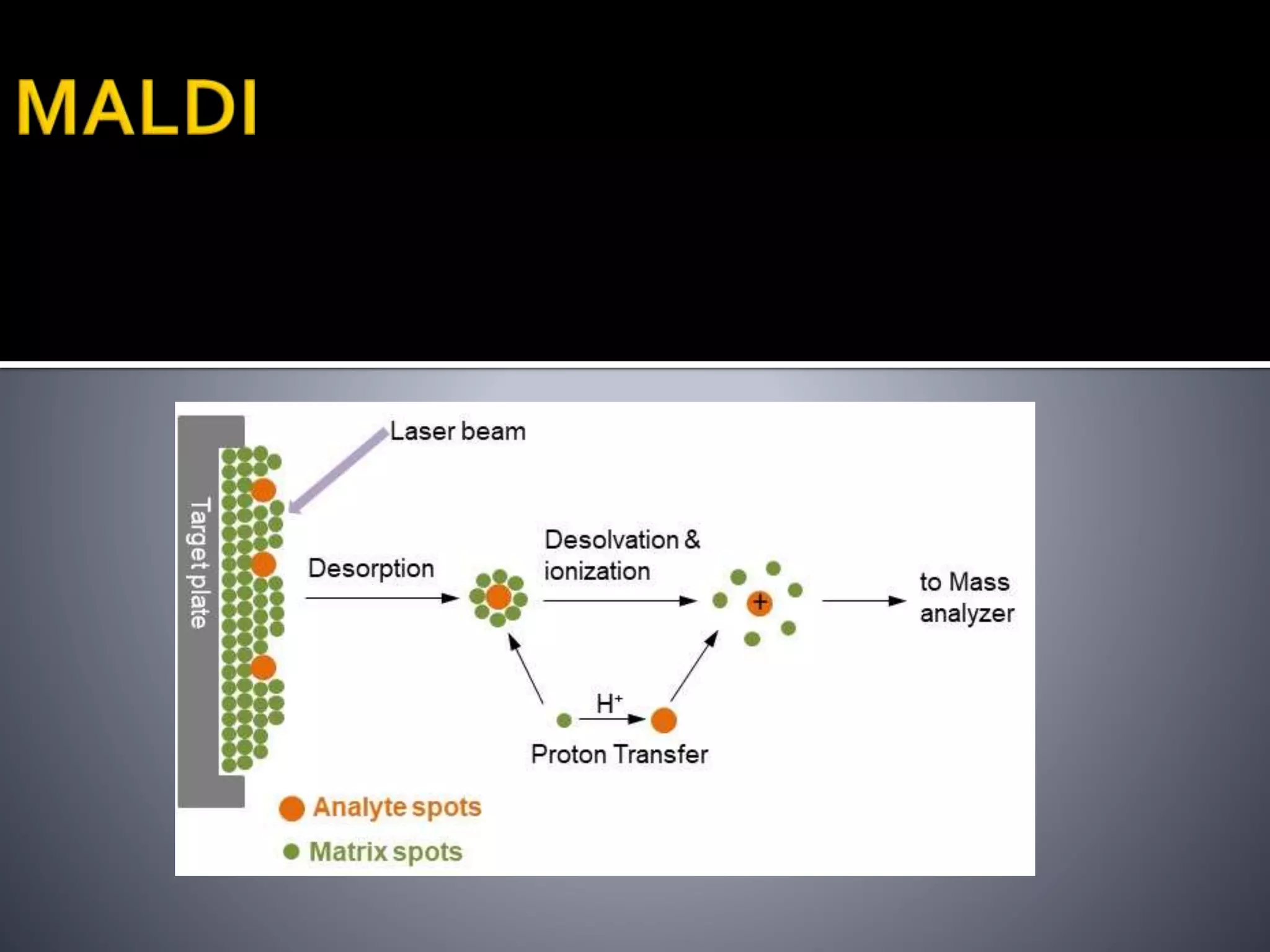
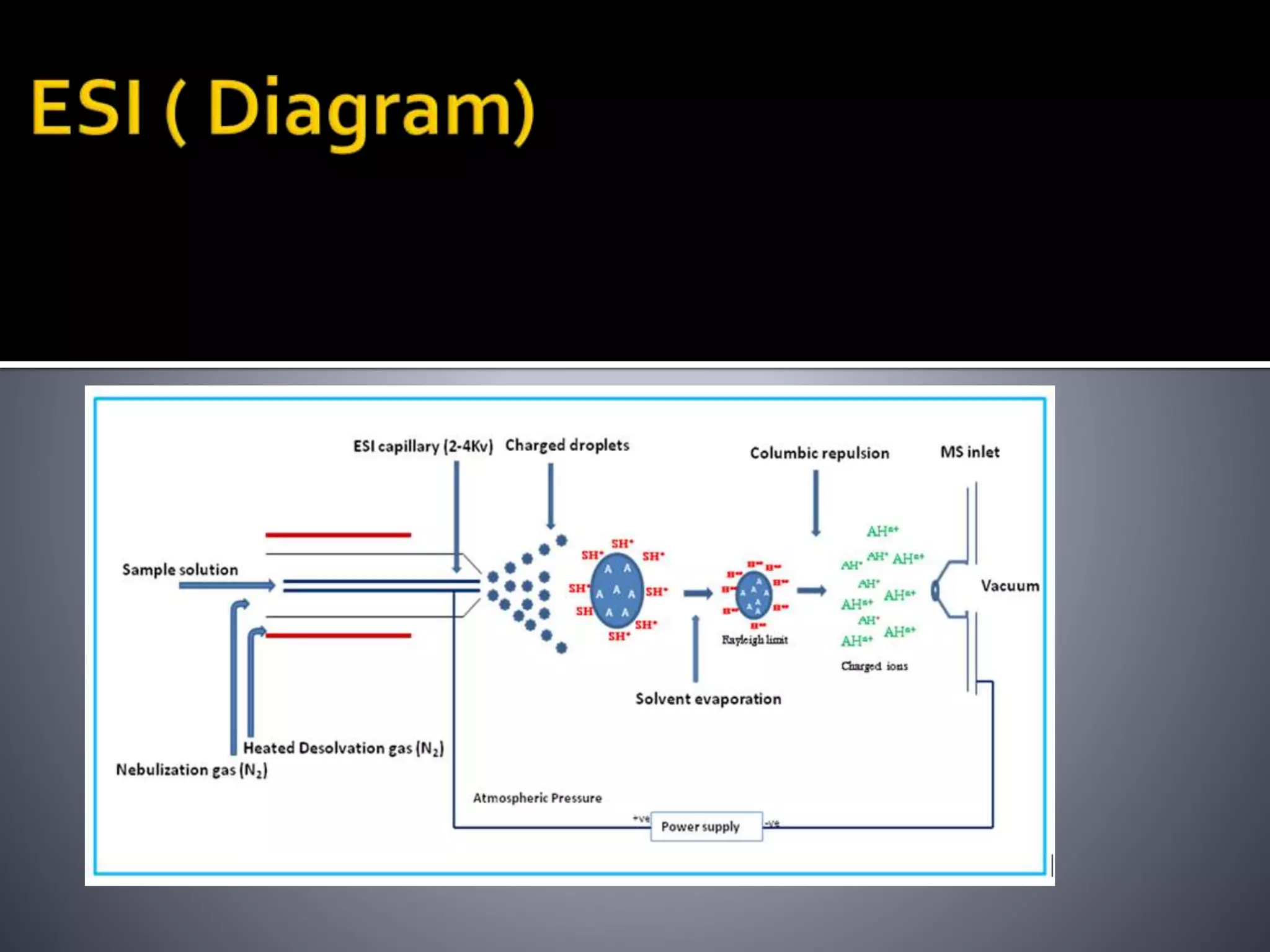
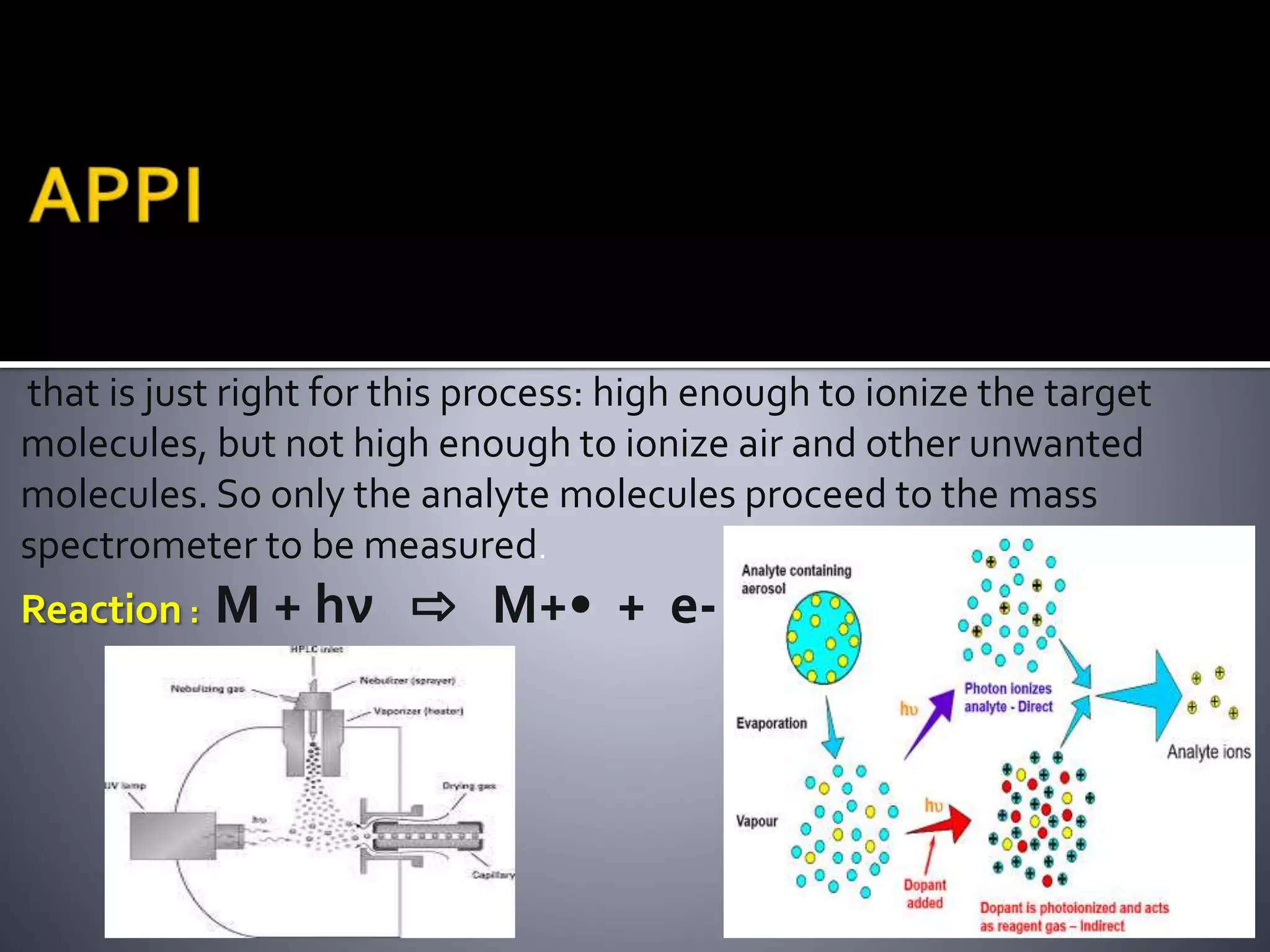
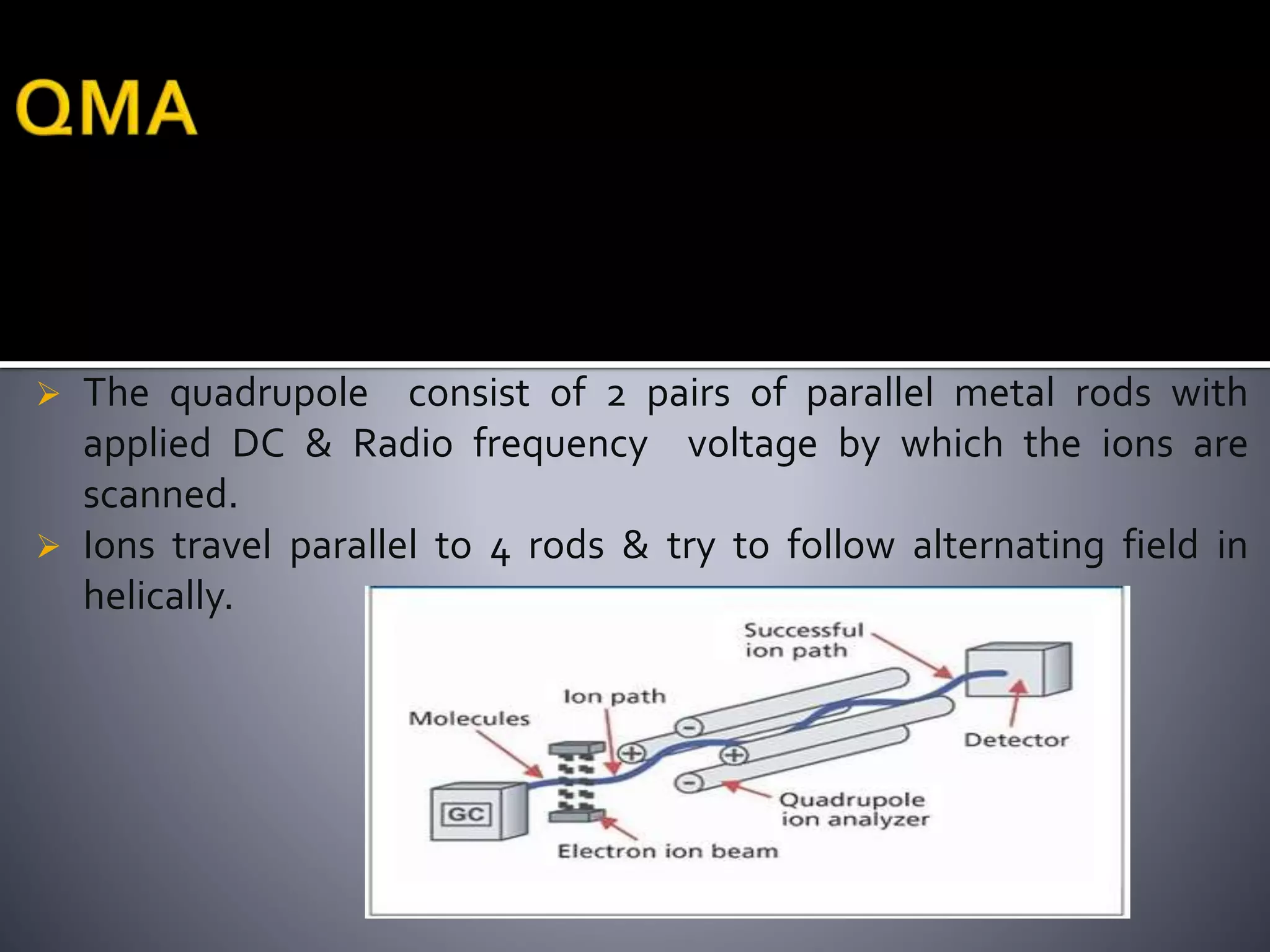
This document discusses several ionization techniques used in mass spectrometry including electron impact ionization, chemical ionization, field ionization, MALDI, FAB, ESI, APCI, APPI, and their applications. It also describes the working of common mass analyzers like quadrupole mass analyzer and time-of-flight analyzer. Finally, it mentions some applications of mass spectrometry like protein characterization, isotope tracking, molecular weight determination, studying reaction mechanisms etc.