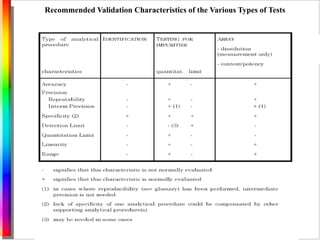
This document summarizes guidelines for analytical method validation from various regulatory agencies. It discusses the purpose of validation to verify method suitability for submissions. Key validation characteristics covered include specificity, linearity, range, accuracy, precision, detection/quantitation limits, robustness, and system suitability testing. It notes that revalidation may be necessary if the analytical method or drug product is changed.





















![The United State Pharmacopoeia 24; The National
Formulary 19; 2000: [1225] VALIDATION OF
COMPENDIAL METHODS.
http://www.labcompliance.com/methods/meth_val.
htm#introduction
http://www.fda.gov/cder/guidance/2396dft.htm
www.fda.gov/ohrms/dockets/
ac/02/slides/3841s1_07_lachman.PPT
http://www.fda.gov/cder/guidance/ameth.htm](https://image.slidesharecdn.com/analyticalmethodvalidation-140928235808-phpapp02/85/Analytical-method-validation-25-320.jpg)

