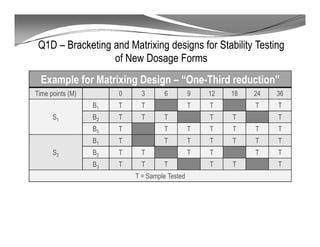
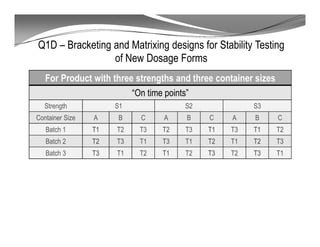
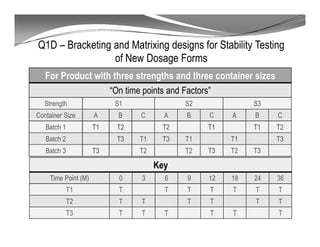
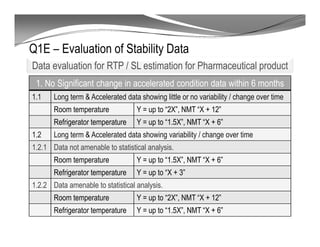
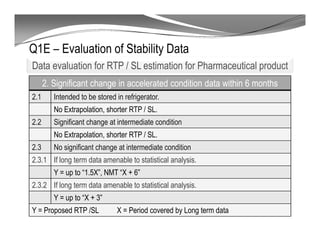
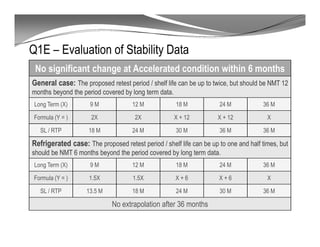
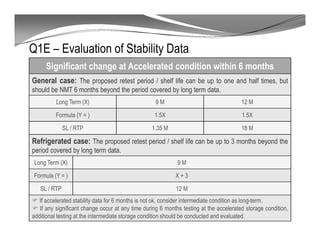
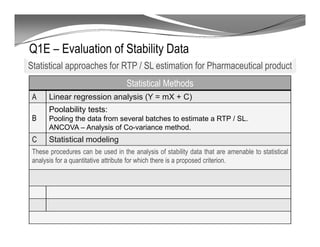
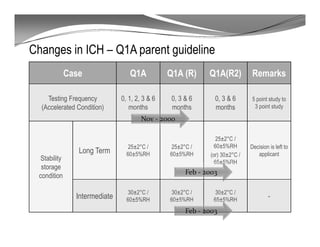
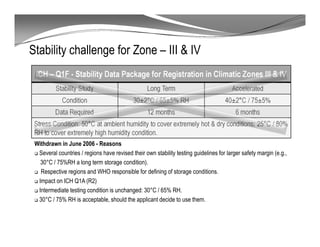
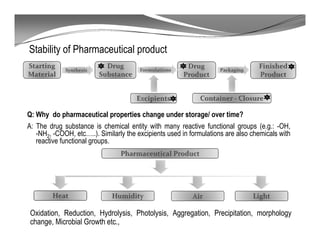
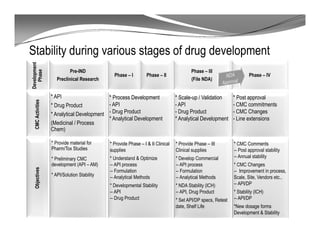
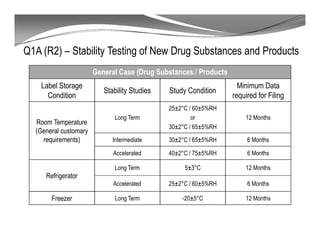
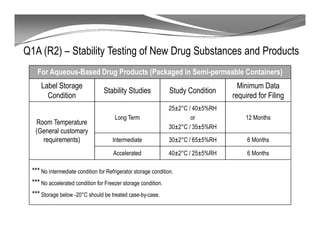
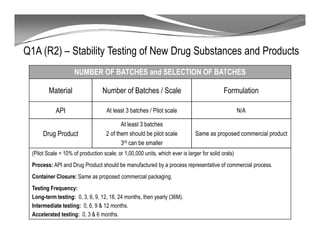
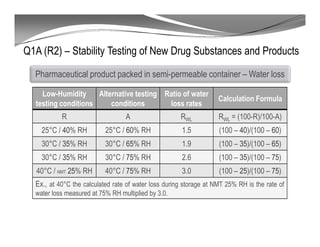
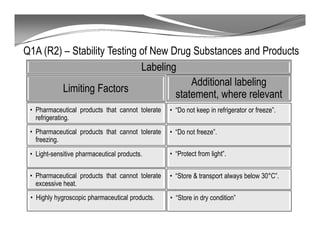
The document discusses ICH stability guidelines for pharmaceutical products. It provides an overview of key ICH guidelines including Q1A(R2) on stability testing of new drug substances and products and Q1B on photo stability testing. Q1A(R2) outlines the core stability data package required, including testing conditions, number of batches, and stability commitments. It also defines criteria for significant changes. Q1B covers photo stability testing conditions and study design. The guidelines aim to provide stability information for marketing applications and ensure quality, safety and efficacy over the shelf life of pharmaceutical products.














![Q1B – Photo Stability Testing of New Drug Substances and Products
Key Features:
-- Covers the photo stability information for application.
-- Photo stability testing conditions for exposing drug substance and products. (Photo sensitivity)
Major points:
-- Light sources
-- Photo stability conditions.
-- Study Design.
-- Number of batches.
Light Sources:
-- Option – 1: Artificial daylight (out put similar to D65 [outdoor daylight] & DI65 [indoor indirect daylight])
combining visible and UV outputs, Xenon or metal halide lamp.
-- Option – 2:
(a) Cool white fluorescent lamp. (b) Near UV Fluorescent lamp with spectral distribution between 320 –
400 nm, Emax emission between 350 – 370 nm.](https://image.slidesharecdn.com/stability-150501144405-conversion-gate02/85/ICH-Stability-Studies-22-320.jpg)