Embed presentation



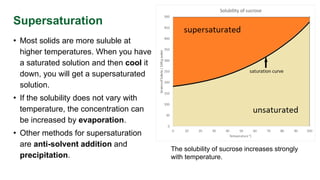





This document provides an introduction to the process of crystallization. It explains that crystallization involves arranging atoms or molecules into rigid crystals from solutions or melts. Crystallization is widely used for separation and purification in industry. The key steps of crystallization are achieving supersaturation of a solution, nucleation of seed crystals, and crystal growth until saturation is reached. Common methods to supersaturate solutions include changing temperature, evaporation, or adding anti-solvents. The objectives of crystallization are typically to achieve high yields, narrow crystal size distributions, maximum purity, and specific morphologies in an economic process.









