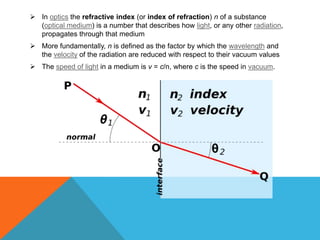
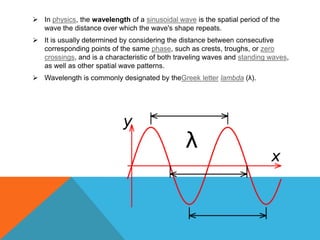
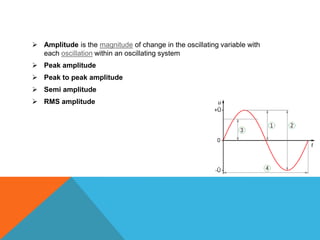
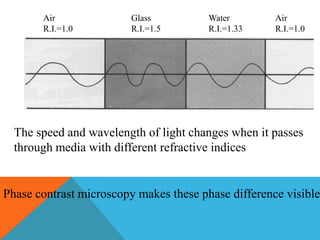
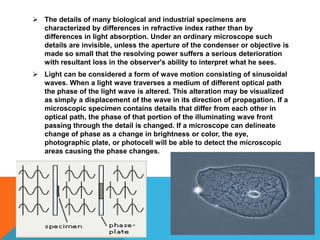
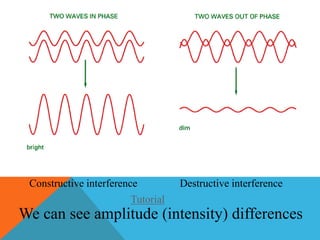
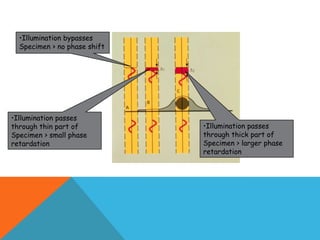
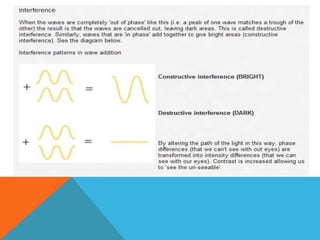
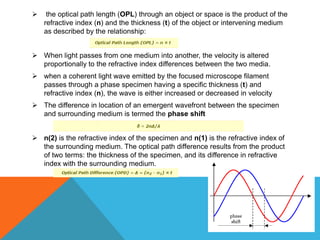
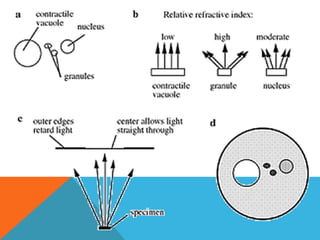
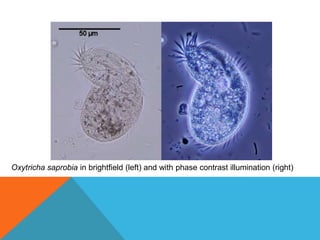
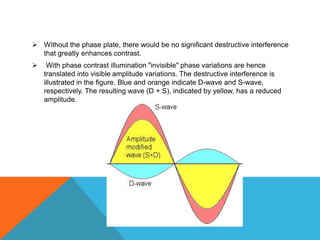
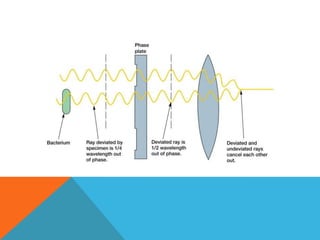
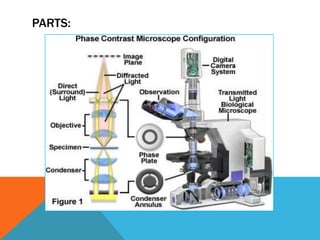
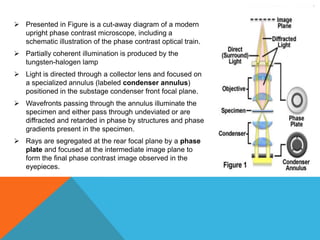
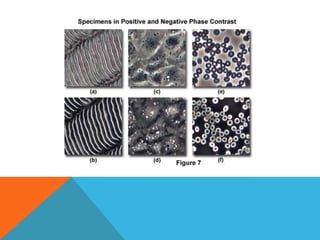
The document provides a comprehensive overview of phase contrast microscopy, highlighting its principles and advancements since its inception. It discusses the role of optical path differences and refractive indices in enhancing the visibility of microscopic specimens, as well as the historical contributions of key figures like Zernike, who received a Nobel Prize for his work in this field. Additionally, it details the technical aspects and components necessary for implementing phase contrast techniques in microscopy, distinguishing between types of phase contrast and equipment setups.