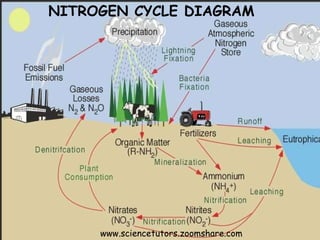
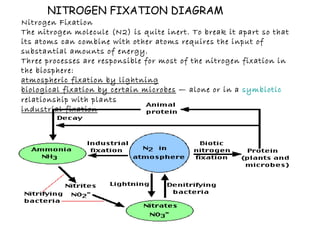
The document provides an overview of the nitrogen cycle, including processes such as nitrogen fixation, nitrification, and denitrification, as well as human impacts on these processes. It discusses natural and industrial methods of nitrogen fixation and highlights the negative effects of human activities, such as fossil fuel combustion and the use of synthetic fertilizers, on nitrogen levels in ecosystems. Additionally, it addresses issues like nitrogen overload leading to problems like eutrophication and acid rain.