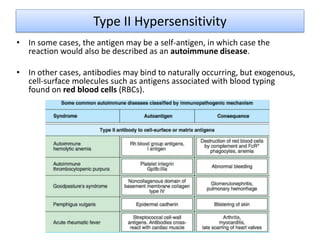
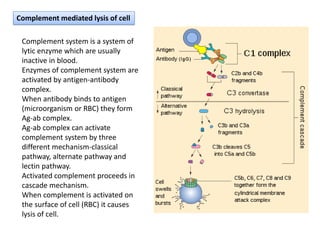
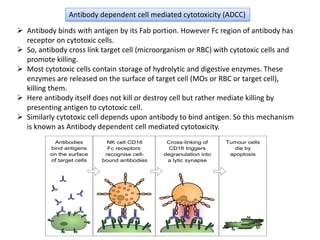
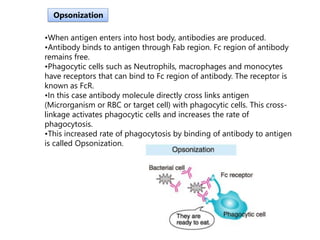
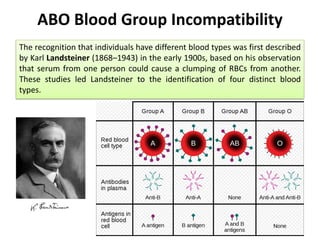
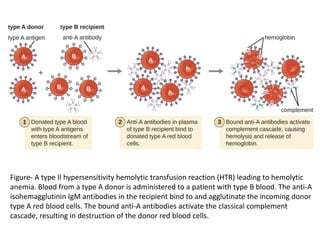
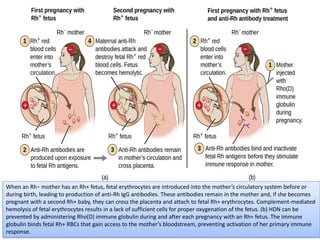
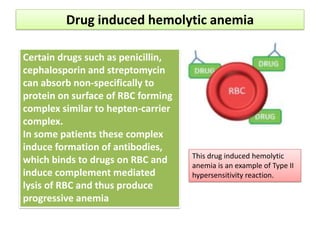
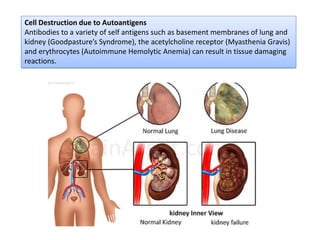
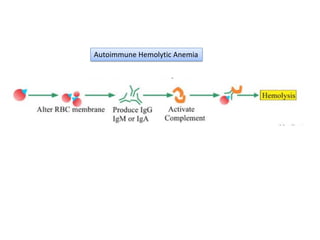
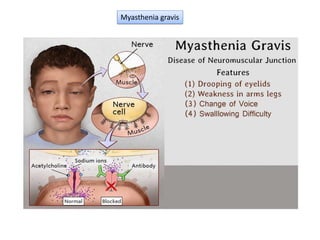
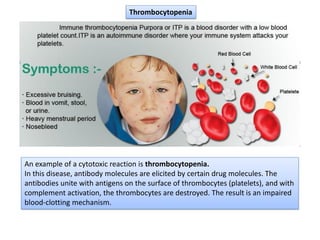
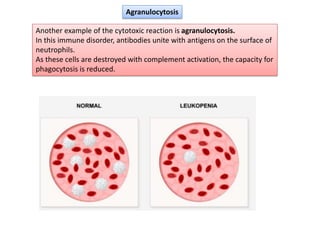
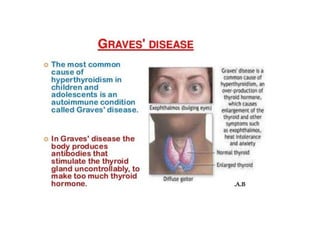
Type II hypersensitivity is an antibody-mediated immune reaction primarily involving IgG and IgM antibodies that target cellular antigens, leading to cell damage and destruction through mechanisms such as complement activation, antibody-dependent cell-mediated cytotoxicity (ADCC), and opsonization. This hypersensitivity can manifest in various clinical conditions including transfusion reactions, hemolytic disease of the newborn, autoimmune disorders, and drug-induced hemolytic anemia. These reactions are typically immediate, occurring within minutes to hours post-exposure.