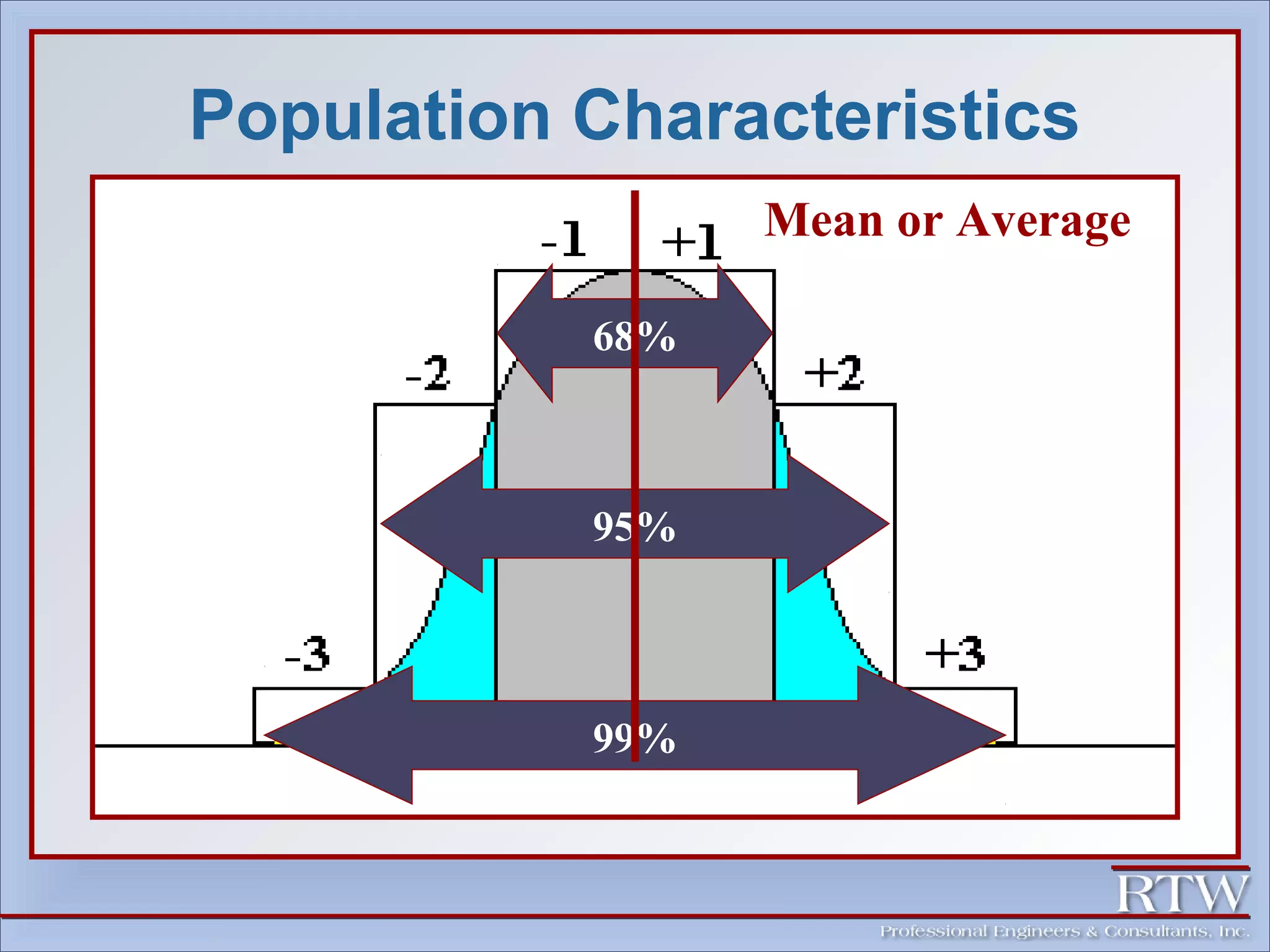
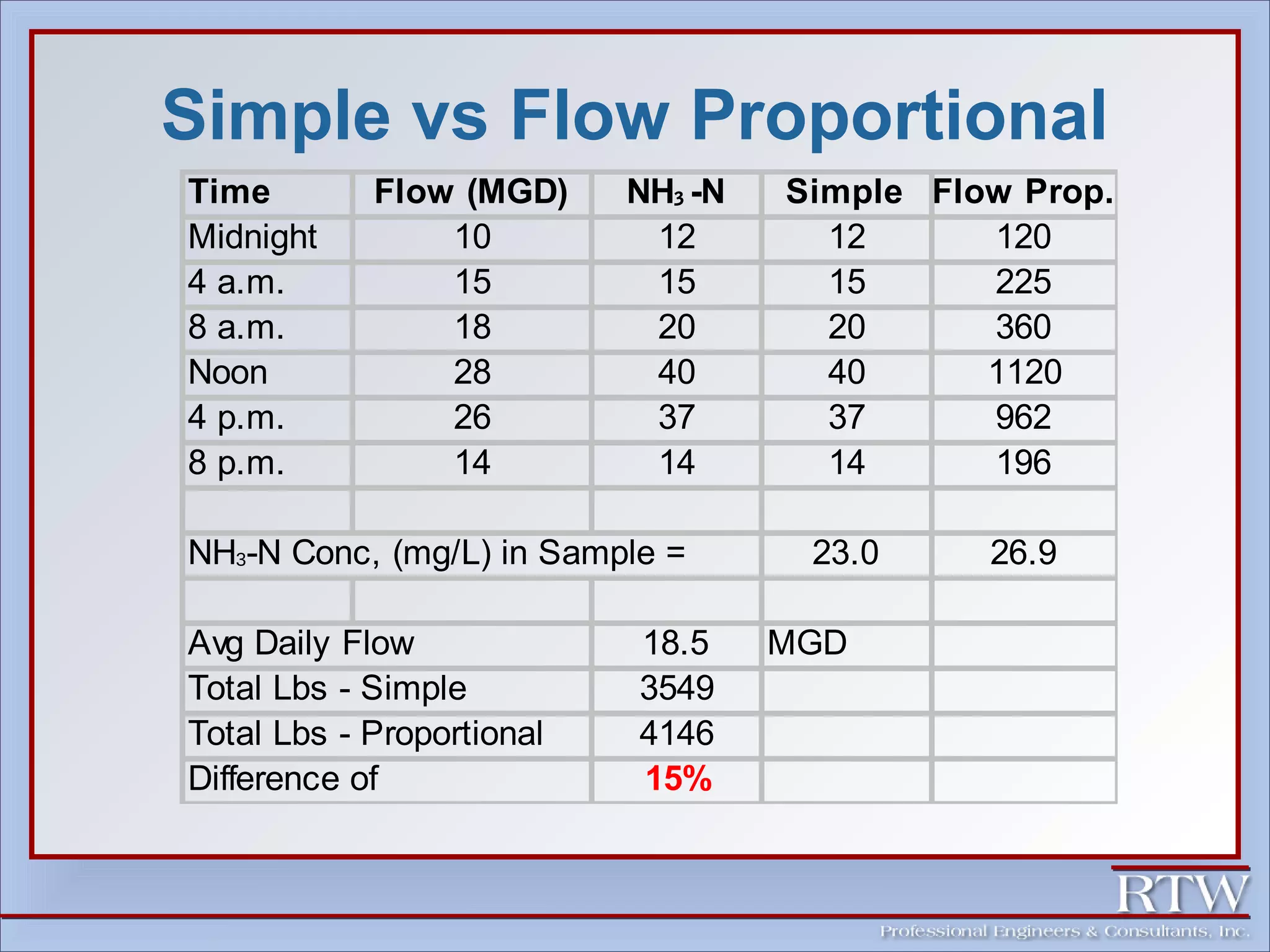
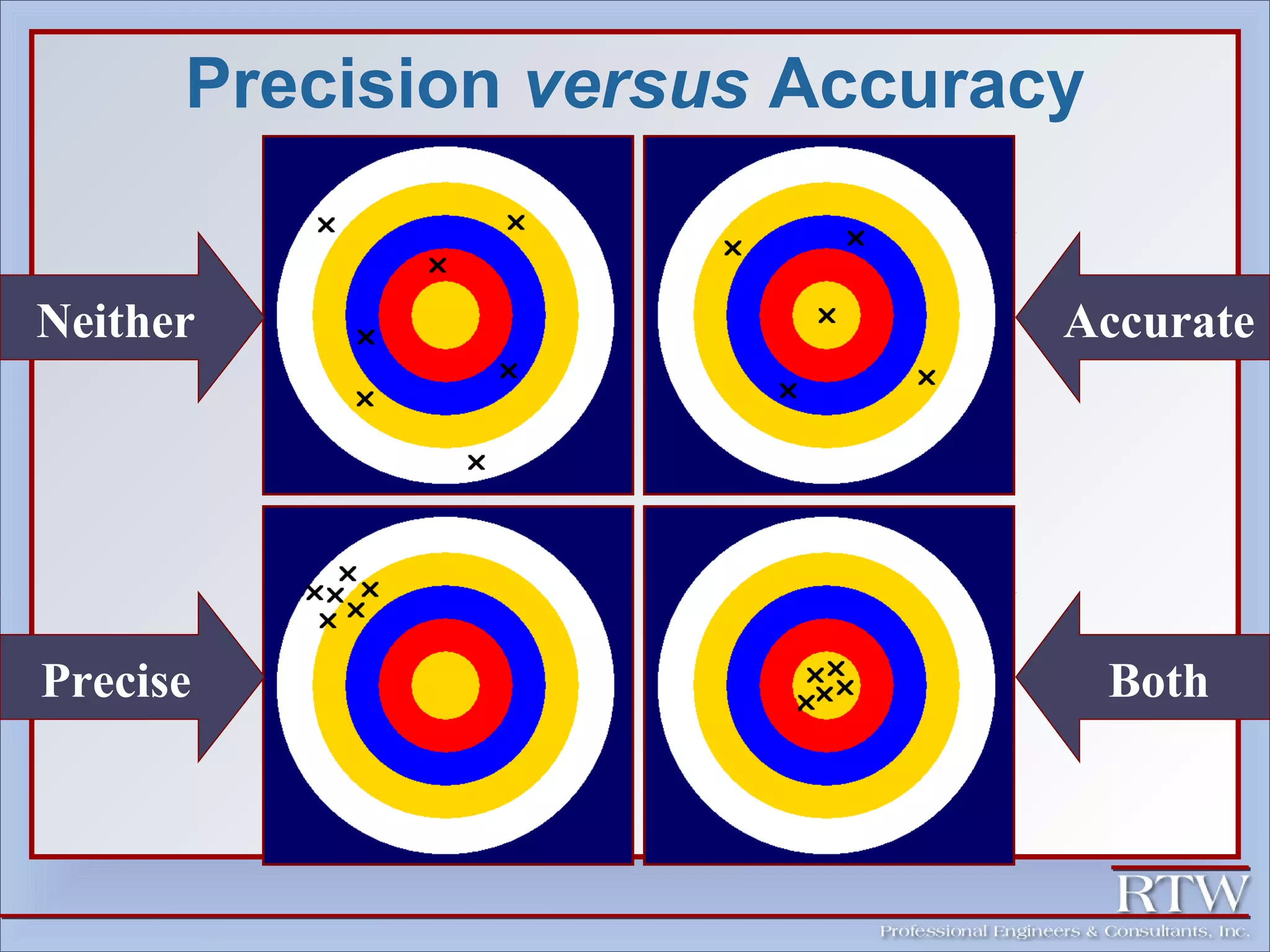
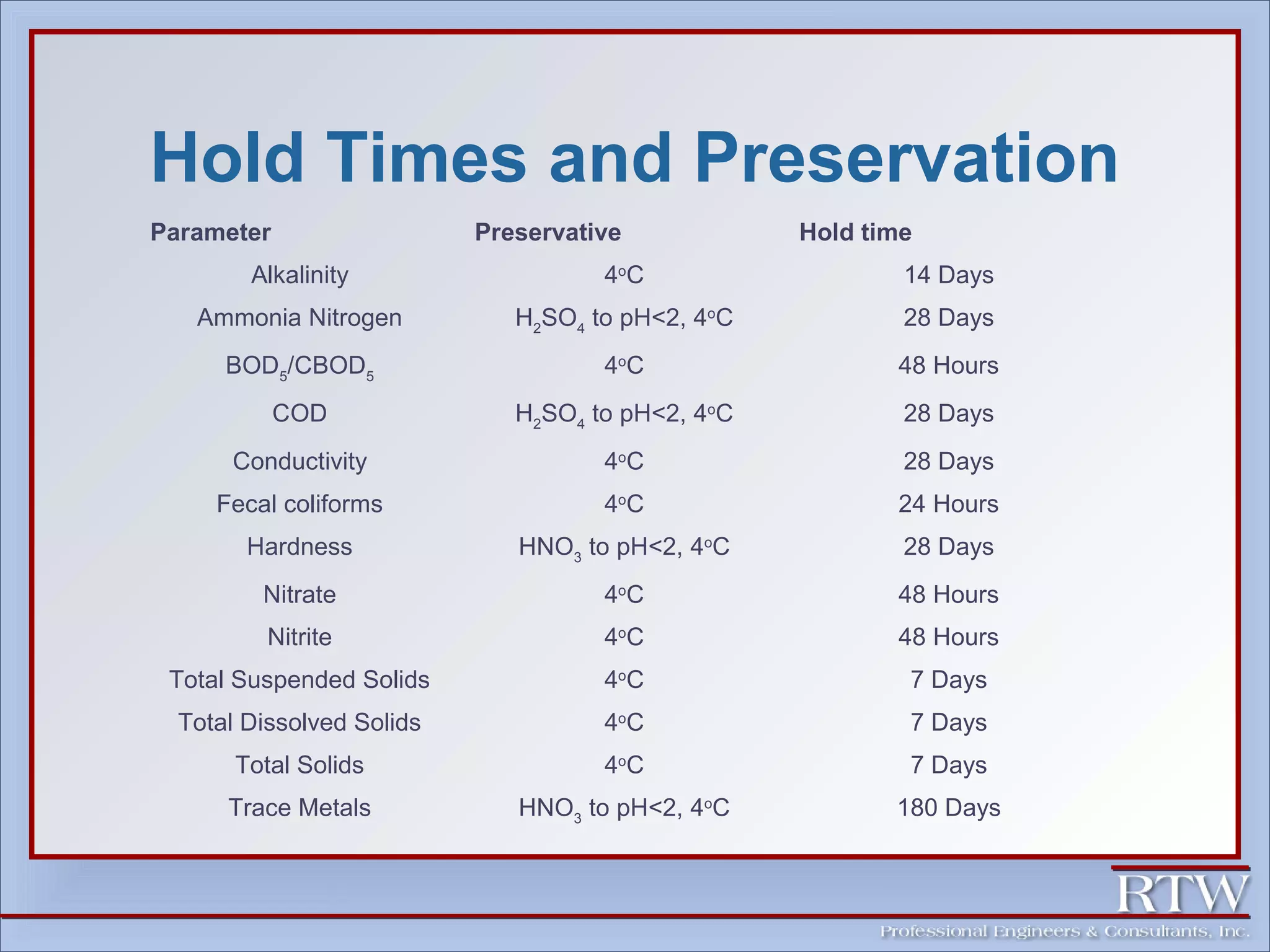
This document discusses representative sampling and quality assurance/quality control procedures. It covers topics such as types of samples, ideal sampling locations, data quality objectives, and examples of proper and improper sampling techniques. Quality control measures like blanks, duplicates, and standards are described to ensure sample accuracy, precision, and to check for contamination in the sampling and analysis process. Maintaining proper sample handling and preservation techniques as well as adhering to hold times for analysis are also important aspects of quality control.