Embed presentation
Downloaded 285 times


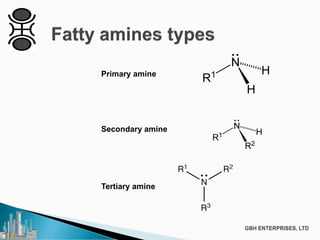

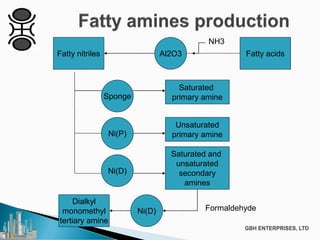


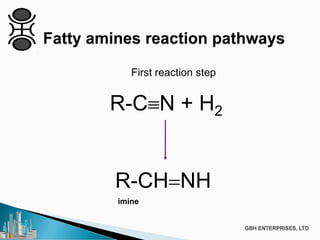
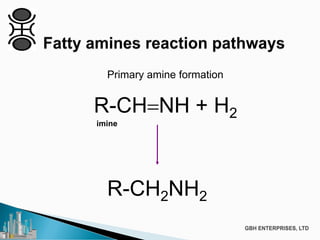
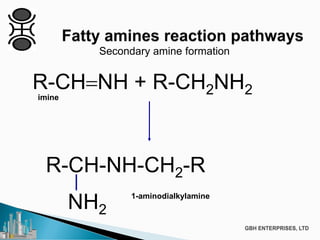
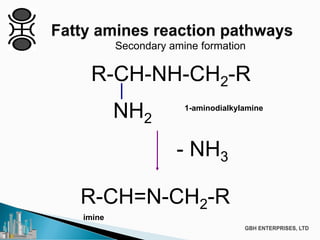
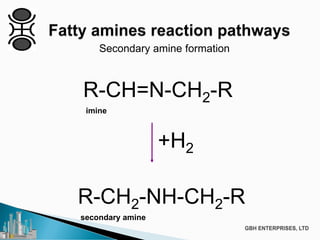
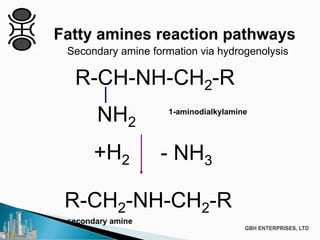

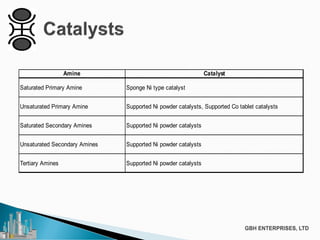


The document discusses the production of fatty amines, focusing on the different types of amines (primary, secondary, and tertiary) and their applications such as corrosion inhibitors and fabric softeners. It details the reaction conditions, catalyst choices, and specific temperature and pressure ranges for each type of amine production. The document outlines chemical processes involved in forming these amines, including reactions with ammonia and other compounds.















