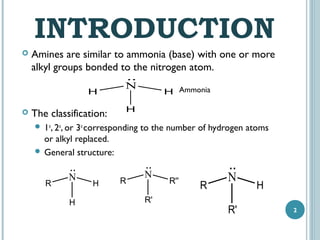
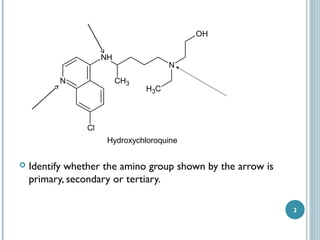
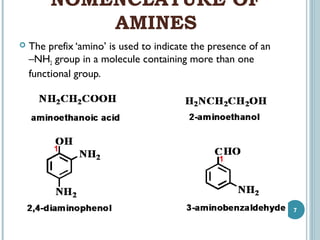
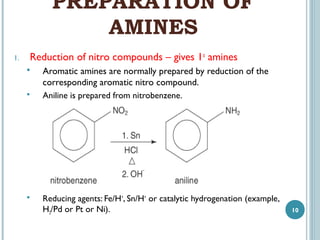
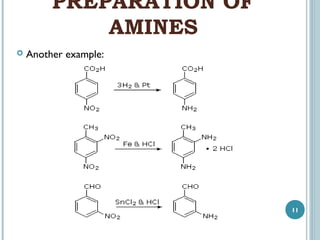
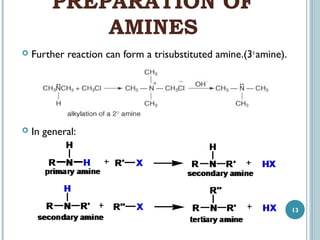
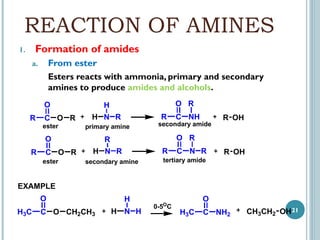
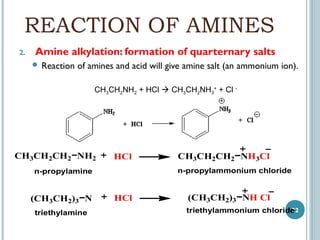
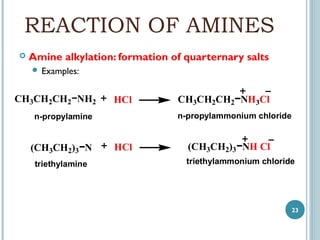
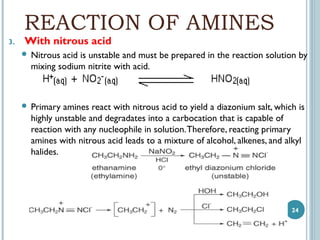
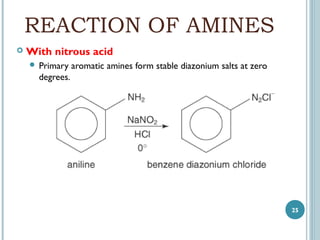
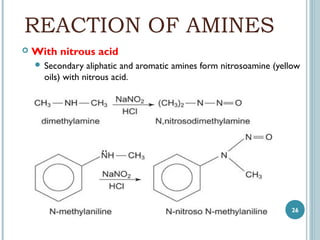
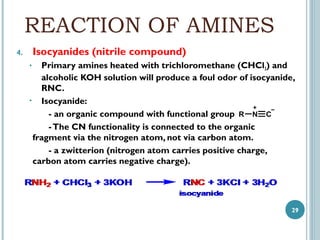
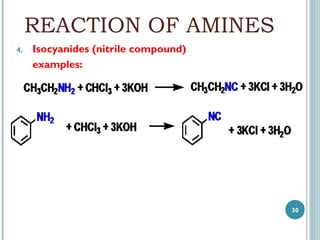
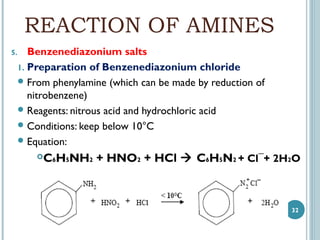
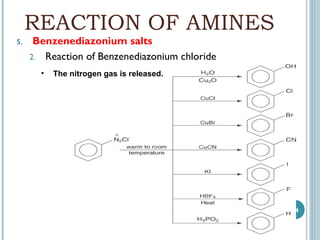
1. Amines are organic compounds derived from ammonia by replacing one or more hydrogen atoms with alkyl groups. This document discusses the nomenclature, preparation, and reactions of amines.
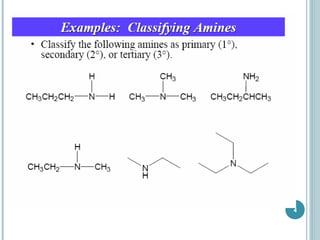
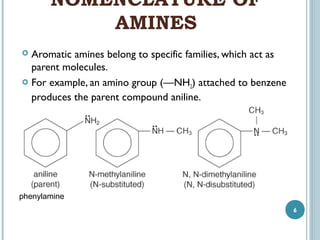
2. Amines are named based on whether they contain one, two, or three alkyl groups bonded to the nitrogen atom (primary, secondary, tertiary). Aromatic amines are named after the parent aromatic compound with the suffix -amine.
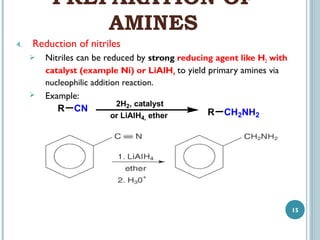
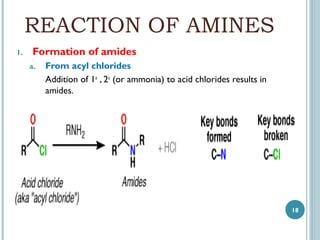
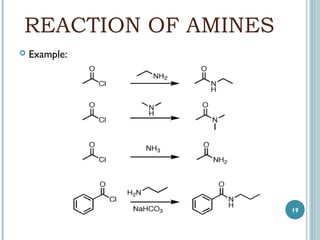
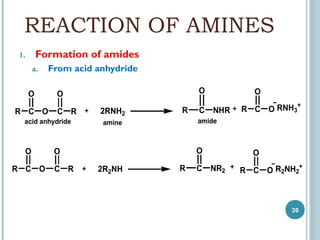
3. Amines can be prepared through reduction of nitro compounds, halides, amides, nitriles, or amides via Hoffman degradation. Common reducing agents include lithium aluminum hydride and catalytic hydrogenation.