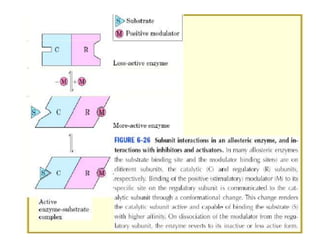
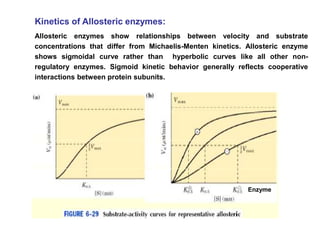
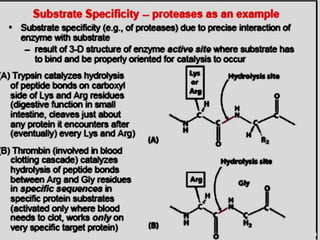
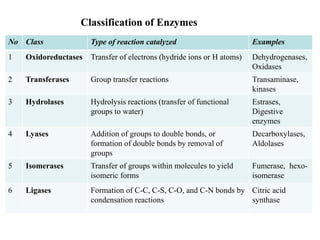
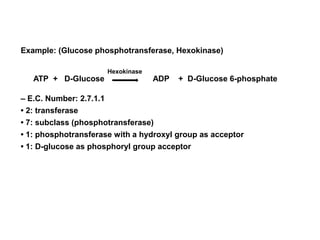
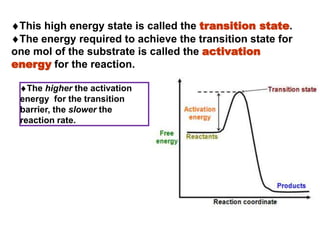
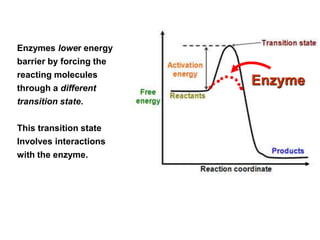
The document discusses enzymes as essential biocatalysts for biochemical reactions, emphasizing their roles in life processes, specificity, catalytic efficiency, and regulation. It covers enzyme structure, classification, and kinetics, including the Michaelis-Menten model, factors affecting reaction velocity, inhibition types, and regulatory mechanisms. Enzymes are vital to biological functions and are affected by various conditions such as substrate concentration, temperature, and pH.












![Factors afecting reaction velocity:
Enzymes can be isolated from cells and their
properties studied in vitro.
Velocity: The rate or velocity of a reaction is the
number of substrate molecules converted to
product per unit time, velocity is usually
expressed as µmol of product formed per
minute.
Factors affecting reaction velocity:
1. Substrate concentration:
The rate of an enzyme-catalyzed reaction
increases with increasing substrate
concentration until a maximal velocity is
reached. The plot of initial reaction velocity
against substrate concentration is hyperbolic.
[S]
Rate](https://image.slidesharecdn.com/enzymes2lect-200421171248/85/Enzymes-17-320.jpg)

![3. pH (acid-base property):
[H+] affects reaction velocity
The catalytic process usually requires specific chemical groups (ionized or un-
ionized) in enzyme or substrate in order to interact.
eg, if a catalytic activity requires an amino group of an enzyme to be a
protonated form (-NH3
+), at alkaline pH this group is deprotonated and
therefore the reaction velocity declines.
Extremes of pH may leads to denaturation of the enzyme because the
structure of the catalytically active protein molecule depends on the ionic
character of the amino acid side chains.
Optimum pH varies for different enzymes: eg pepsin (digestive enzyme in
stomach) maximally active at pH 2.0, whereas enzymes, supposed to work at
neutral pH are denatured by such an acidic environment.](https://image.slidesharecdn.com/enzymes2lect-200421171248/85/Enzymes-19-320.jpg)


![Michaelis-Menten Model or Equation
k1 k3
E + S E●S E+ P
k2
From this kinetic scheme, a relationship can be
derived for the rate or velocity of the reaction:
Michaelis-Menten Equation
Vmax[S]
[S] + Km
V =](https://image.slidesharecdn.com/enzymes2lect-200421171248/85/Enzymes-24-320.jpg)
![v or
rate
[S]
Vmax
Km
½ Vmax
0
Vmax, the maximum rate (plateau)
is k3 x [total enzyme]
Km =(k1 + k3)/ k2, almost a
binding constant
Michaelis-Menten constant
(Km) can be defined as the
concentration of the specific
substrate at which a given
enzyme reaches one-half its
maximum velocity.
Km = [S], where the velocity v
= ½ Vmax,
Km is called the Michaelis
constant.
k1 k3
E + S E●S E+ P
k2](https://image.slidesharecdn.com/enzymes2lect-200421171248/85/Enzymes-25-320.jpg)
![Km reflects the affinity of the
enzyme for a substrate.
•Low Km value reflects a high affinity
of the enzyme for substrate,
because a low concentration of
substrate is needed to half-saturate
the enzyme that is to reach the
velocity at ½ Vmax.
•High Km reflects the low affinity
enzyme for substrate because a
high concentration of substrate is
needed to half-saturate the enzyme.
v
Vmax/2
Km
[S]
Vmax](https://image.slidesharecdn.com/enzymes2lect-200421171248/85/Enzymes-26-320.jpg)
![Lineweaver-Burk plot:
This form of the Michaelis-Menten equation is
called the Lineweaver-Burk equation. For
enzymes obeying the Michaelis-Menten
relationship, a plot of 1/V0 versus 1/[S] yields a
straight line. This line has a slope of Km/Vmax, an
intercept of 1/Vmax on the 1/V0 axis, and an
intercept of 1/Km on the 1/[S] axis. The double-
reciprocal presentation, also called a Lineweaver-
Burk plot, has the great advantage of allowing a
more accurate determination of Vmax, which can
only be approximated from a simple plot of V0
versus [S].](https://image.slidesharecdn.com/enzymes2lect-200421171248/85/Enzymes-27-320.jpg)




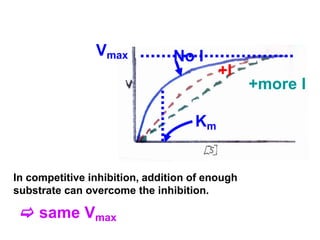
![Effect of competitive inhibitor on Vmax:
The effect of a competitive inhibitor is reversed by increasing [S]. At a
sufficient high substrate concentration, the reaction velocity reaches
the Vmax as observed in the absence of inhibitor.
Effect of competitive inhibitor on Km:
A competitive inhibitor increases the apparent Km for a given substrate
which means that, in the presence of a cmpetitive inhibitor, more
substrate is needed to achieve ½ Vmax.
Statin drugs: competitively inhibits HMG-CoA reductase (rate
limiting enzyme) and thus inhibit synthesis of cholesterol, thereby
lowering plasma cholesterol levels.](https://image.slidesharecdn.com/enzymes2lect-200421171248/85/Enzymes-33-320.jpg)