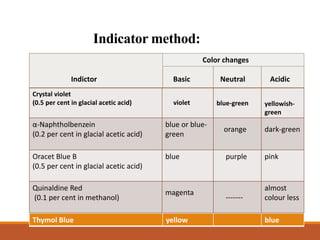
Non-aqueous titration involves titrating substances that are not soluble or do not have a sharp endpoint in water using a non-aqueous solvent. The choice of solvent can affect whether a substance acts as a strong acid or base. Various types of non-aqueous solvents exist including aprotic, protogenic, protophilic, and amphiprotic solvents. Visual indicators are typically used to detect the endpoint in non-aqueous titrations. Some common indicators include crystal violet, oracet blue B, and quinaldine red. Non-aqueous titrations allow for the analysis of organic acids and bases that cannot be easily analyzed using aqueous titration.