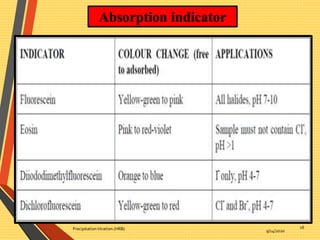
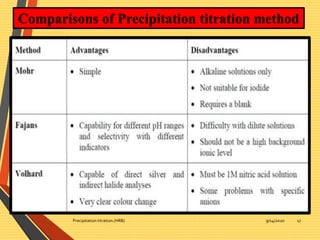
This document discusses precipitation titration methods. It describes the principles of precipitation titration where a titrant forms an insoluble precipitate with the analyte. Common methods like Mohr's, Volhard's, and Fajans are summarized. Factors affecting precipitate solubility and limitations of precipitation titration are also outlined. The document serves to introduce various techniques in precipitation titration.











![9/14/2020Precipitation titration.(HRB) 12
In this method, there is formation of colored compound at
the end point.
Silver ion is titrated with thiocyanate in an acid solution
using ferric ion as an indicator.
Ag+ (aq) + SCN– (aq) → AgSCN(s)
Initially silver thiocyanate is precipitated. After the
equivalence point, when no Ag+ is present, the excess of
thiocyanate added reacts with Fe3+ to give reddish brown
ferric thiocyanate.
Fe3+ (aq) + SCN– (aq) → [FeSCN]2
+ (aq)](https://image.slidesharecdn.com/pa-iunit-iiiprecipitationtirationhrb1-200916072948/85/PA-I-Precipitation-titration-HRB-12-320.jpg)