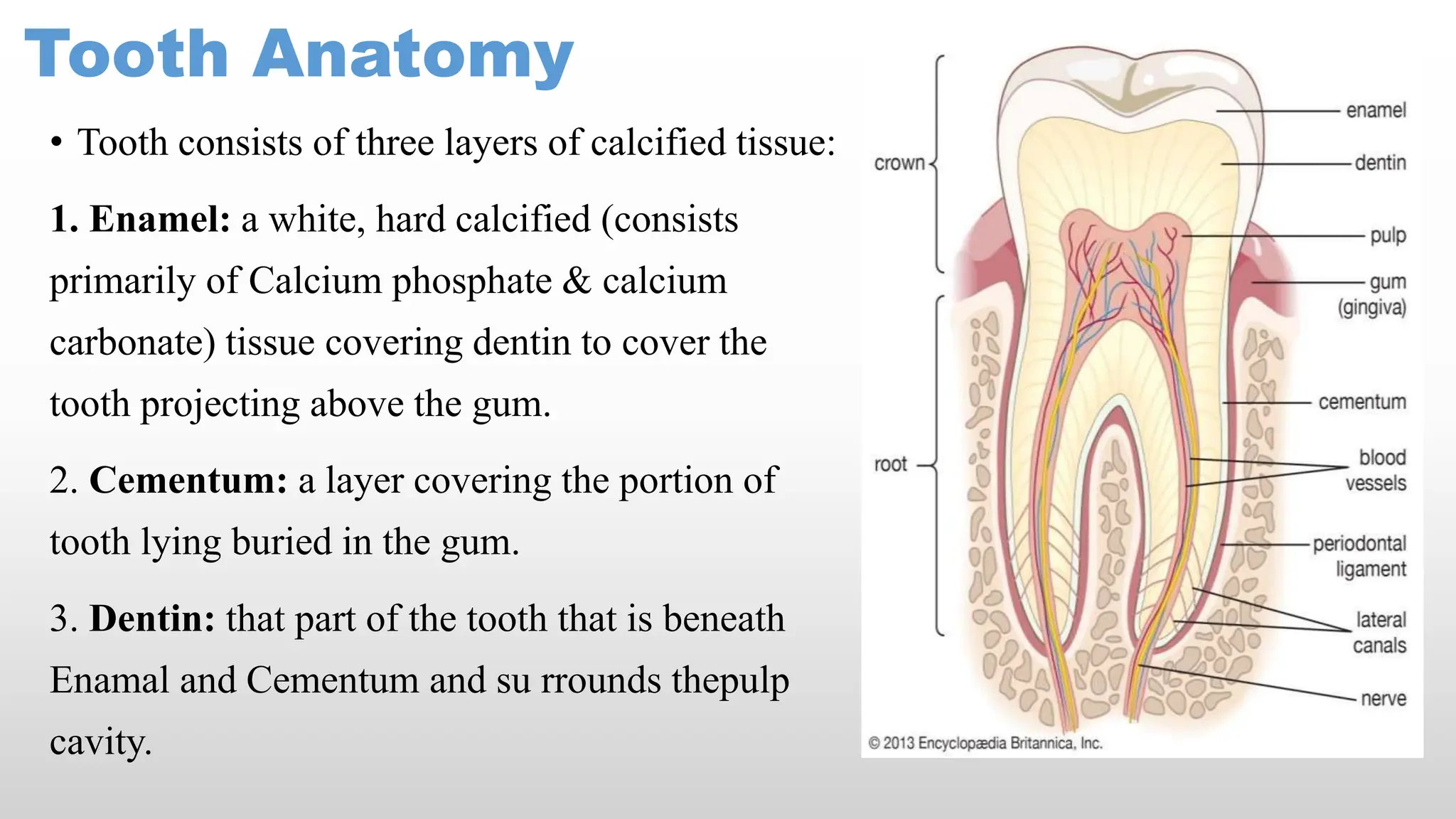
The document discusses various dental products essential for dental hygiene, including anticaries agents, cleaning agents, desensitizing agents, and cement and fillers, emphasizing their roles in preventing dental decay and maintaining oral health. It details the anatomy of teeth and the significance of vitamins and minerals in their formation, alongside the mechanisms of fluoride and its impact on dental caries. Additional information on the preparation, properties, and uses of specific chemicals like sodium fluoride, stannous fluoride, and dental cements is also provided.