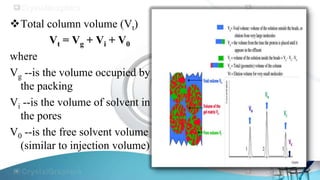
Size exclusion chromatography separates molecules based on their size, with larger molecules eluting from the column first. It uses a stationary phase of porous beads and an aqueous or organic mobile phase. Molecules smaller than the pore size penetrate the beads and take longer to elute, while larger molecules are excluded from the pores and elute more quickly. It is commonly used to determine the molecular weight distribution of polymers and to separate biomolecules like proteins.