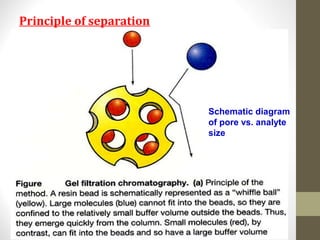
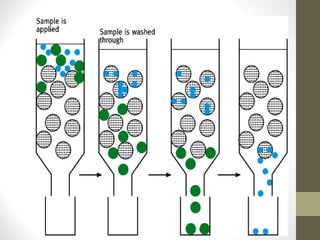
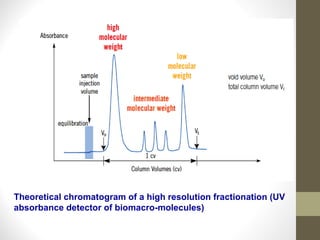
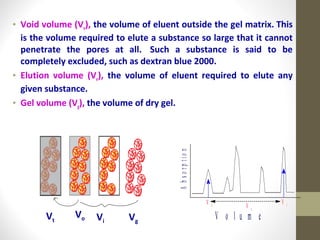
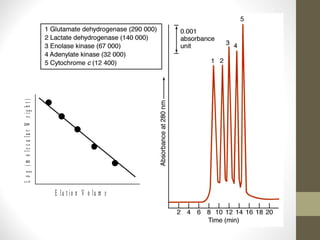
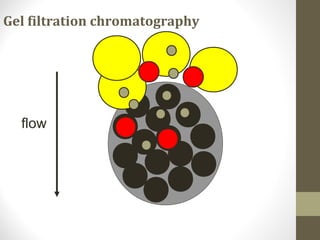
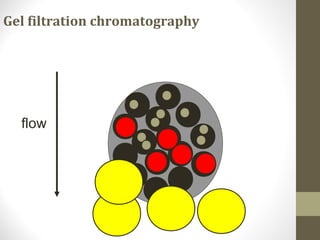
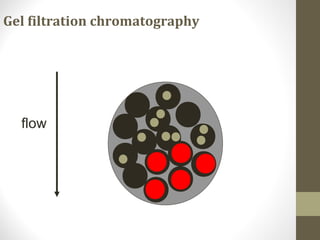
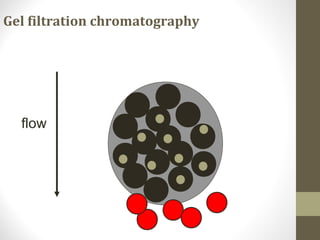
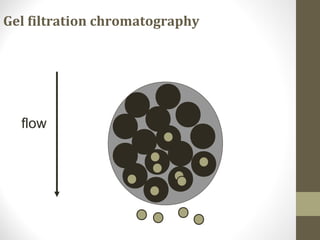
This document discusses gel chromatography, a technique used to separate biomolecules based on differences in size. The stationary phase consists of porous gel beads, which allow larger molecules to pass through more quickly while smaller molecules spend more time interacting with pores and elute later. Common applications include determining molecular weights by relating elution volume to log of weight, fractionating mixtures of biomolecules by size, and desalting samples. The document provides details on properties and types of gels used, as well as procedures for column packing and sample separation.