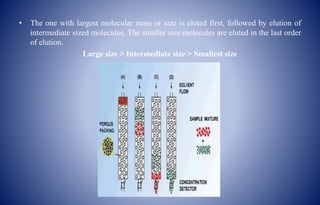
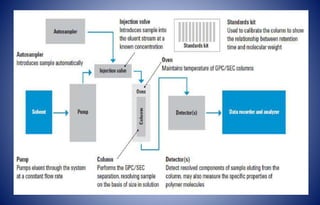
Gel chromatography is a chromatographic technique used to separate and analyze bio-macromolecular substances based on molecular weight or size. The method employs cross-linked polymer gels as a stationary phase, allowing larger molecules to elute faster while smaller ones are retained longer. Its applications include purification, desalting, fractionation, and determination of molecular weight of various biological molecules.