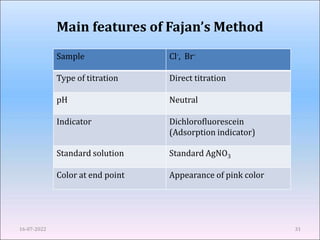
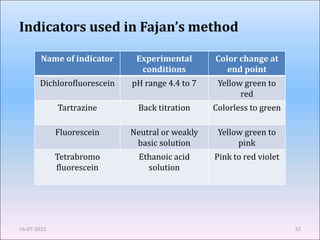
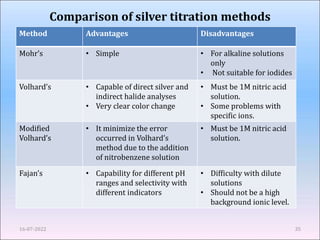
This document provides an overview of precipitation titration methods. It discusses Mohr's method, Volhard's method, and Fajan's method. Mohr's method uses potassium chromate as an indicator and detects the endpoint when a brick red precipitate of silver chromate forms. Volhard's method titrates excess silver ions with thiocyanate using ferric ion as an indicator. Fajan's method employs adsorption indicators that change color when adsorbed onto the precipitate surface at the endpoint. The document explains the principles, procedures, applications and limitations of each precipitation titration method.







![The solubility mainly depends upon several factors such as ions, pH,
temperature and solvents etc.
1) Common ion effect: The solubility of any slightly soluble salt can be
decreased by adding an excess of either of its ions.
Ex.- The dissociation of a slightly soluble salt BA is
BA(S) ⇌ B+ + A-
At equilibrium,
Ksp = [B+] [A-]
Ksp = Solubility product which is a constant
16-07-2022 8](https://image.slidesharecdn.com/precipitationtitrations-220716191504-76044619/85/Sahil-8-320.jpg)
![• If, an excess of either B+ or A- ions are added in the form of another salt
(whose solubility is greater than that of BA), then the product of ionic
concentrations [B+][A-] will exceed the solubility product and hence BA
will precipitate, The common ion effect provides a valuable method for
controlling (the concentration of the ions furnished by a weak
electrolyte.
• Effect of pH in solubility : The solubility of a salt will be decreased by
an increase in pH and vice versa and mainly depends on the anion of the
salt and it is a conjugate base of a weak acid or cation which is conjugate
acid of a weak base.
16-07-2022 9](https://image.slidesharecdn.com/precipitationtitrations-220716191504-76044619/85/Sahil-9-320.jpg)