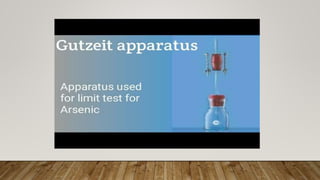
This document describes the limit test for arsenic using the Gutzeit method. Arsenic in a sample is converted to arsine gas, which reacts with mercuric chloride paper to produce a stain that is compared to a standard stain. The test uses an apparatus with two glass tubes, where the sample is placed below zinc and hydrochloric acid to produce arsine gas, which passes through mercuric chloride paper to produce a stain after 40 minutes. By comparing the intensity of this test stain to the standard stain produced in the same way from a solution of known arsenic concentration, the document determines if the sample passes or fails the limit test for arsenic.