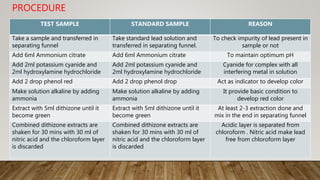
The document describes the limit test for lead, which determines the allowable limit of heavy metal lead in a sample. The test involves reacting the sample with dithizone, which forms a violet-colored lead dithizonate complex in the presence of lead. The intensity of color in the sample is compared to that of a standard lead solution treated the same way. If the sample solution is less colored than the standard, the sample passes the lead limit test. The test is useful for detecting trace amounts of lead impurity from sources like equipment, storage containers, or packaging materials used during manufacturing or storage of medical compounds.