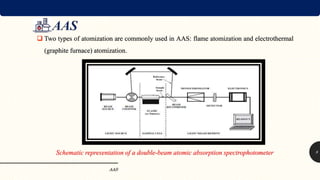
The document discusses Atomic Absorption Spectroscopy (AAS), an analytical method to measure mineral elements by their absorption of ultraviolet or visible radiation in a gaseous state. It details the principles of atomization, instrumentation, and operational procedures, highlighting the advantages of AAS over traditional methods for trace element analysis in food and other applications. Various atomization techniques and their components, including flame and electrothermal atomization, are described, alongside the role of hollow cathode lamps and monochromators in the process.