The document discusses the importance and methods of measurement in physics, outlining various physical quantities, their classifications as fundamental or derived, and the systems of units such as the SI, cgs, and fps systems. It explains the rules for writing SI units, the significance of measurements' accuracy and precision, and how to record significant figures. Additionally, it highlights the types of errors in measurement and the importance of eliminating systematic and random errors for accurate experimental results.





































![Seven dimensions of the world
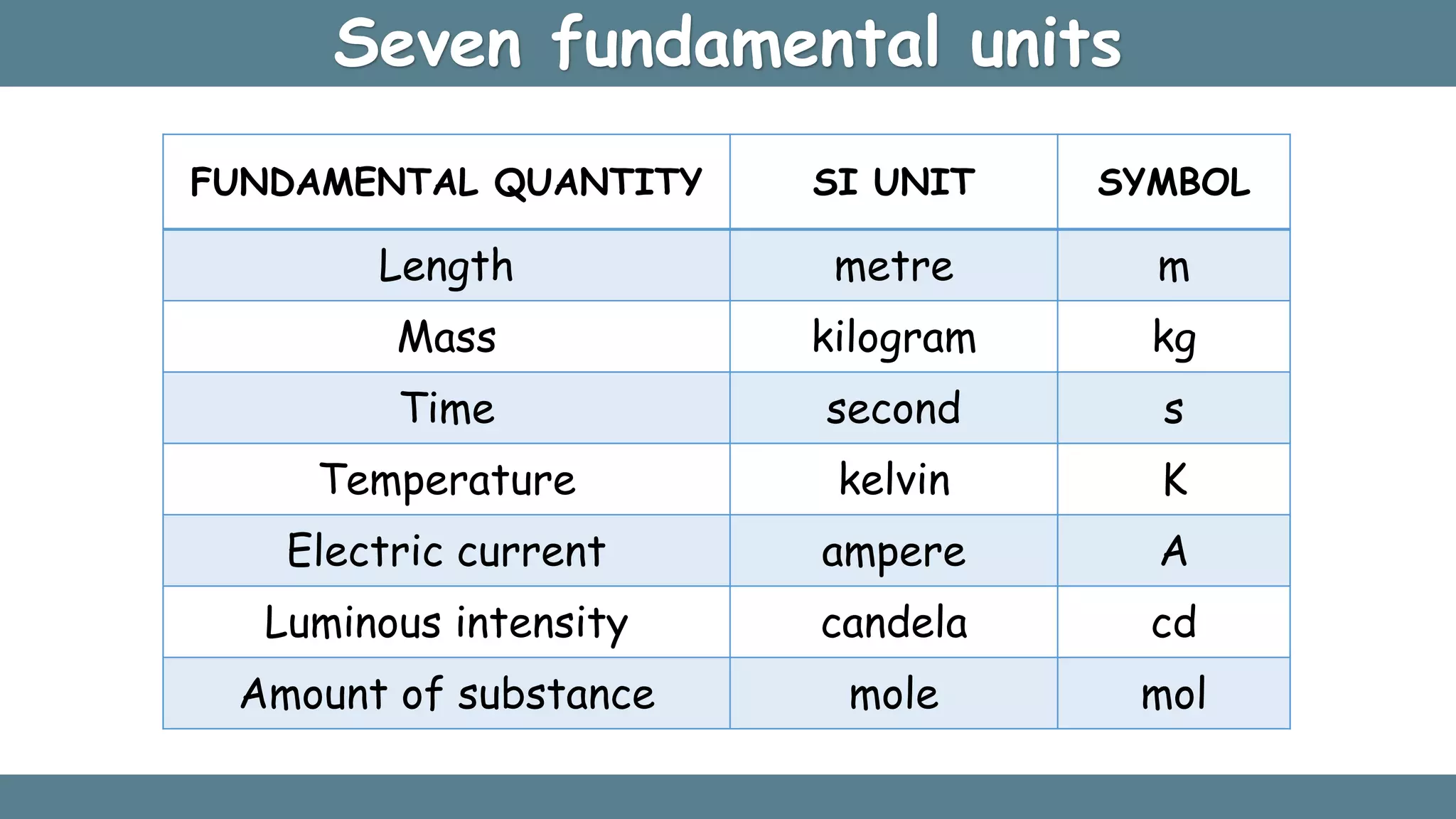
Fundamental quantities
Length
Mass
Time
Temperature
Current
Amount of substance
Luminous intensity
Dimensions
[L]
[M]
[T]
[K]
[A]
[N]
[J]](https://image.slidesharecdn.com/3-measurement-161127184347/75/Units-and-Measurement-41-2048.jpg)

![=
Mass
length × breath × height
[Density] =
[M]
L × L × L
=
[M]
L3
= [ML−3
]
Dimensions of a physical quantity
Density =
Mass
Volume
Example:
Hence the dimensions of density are 1 in mass and − 3 in length.](https://image.slidesharecdn.com/3-measurement-161127184347/75/Units-and-Measurement-43-2048.jpg)

![To check the correctness of equation
∆x = displacement = [L]
Consider the equation of displacement,
By writing the dimensions we get,
vit = velocity × time =
length
time
× time = [L]
at2
= acceleration × time2
=
length
time2
× time2
= [L]
The dimensions of each term are same, hence the equation is
dimensionally correct.
∆x = vit +
1
2
a t2](https://image.slidesharecdn.com/3-measurement-161127184347/75/Units-and-Measurement-45-2048.jpg)
![To convert units
Let us convert newton SI unit of force into dyne CGS unit of force .
The dimesions of force are = [LMT−2
]
So, 1 newton = (1 m)(1 kg)(1 s)−2
and, 1 dyne = (1 cm)(1 g)(1 s)−2
Thus,
1 newton
1 dyne
=
1 m
1 cm
1 kg
1 g
1 s
1 s
−2
=
100 cm
1 cm
1000 g
1 g
1 s
1 s
−2
= 100 × 1000 = 105
Therefore, 1 newton = 105 dyne](https://image.slidesharecdn.com/3-measurement-161127184347/75/Units-and-Measurement-46-2048.jpg)
![To derive a formula
The time period ‘T’ of oscillation of a
simple pendulum depends on length ‘l’
and acceleration due to gravity ‘g’.
Let us assume that,
T ∝ 𝑙a 𝑔b or T = K 𝑙a 𝑔b
K = constant which is dimensionless
Dimensions of T = [L0M0T1]
Dimensions of 𝑙 = [L1
M0
T0
]
Dimensions of g = [L1M0T−2]
Thus, L0M0T1 = K [L1M0T0]a [L1M0T−2]b
= K LaM0T0 LbM0T−2b
L0M0T1 = K La+bM0T−2b
a + b = 0 & − 2b = 1
∴ b = −
1
2
& a =
1
2
T = K 𝑙1/2 𝑔−1/2
∴ T = K
𝑙
𝑔](https://image.slidesharecdn.com/3-measurement-161127184347/75/Units-and-Measurement-47-2048.jpg)