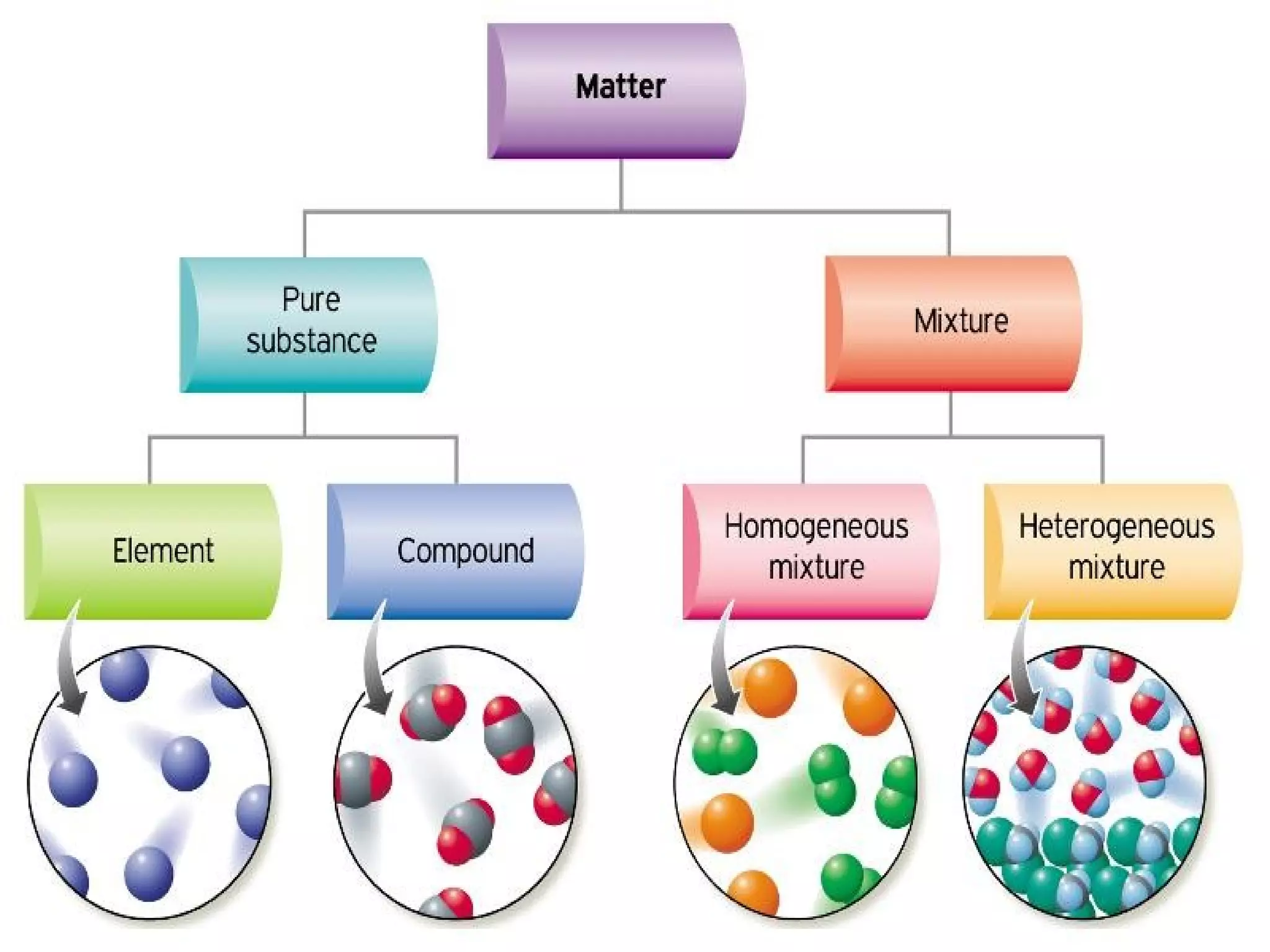
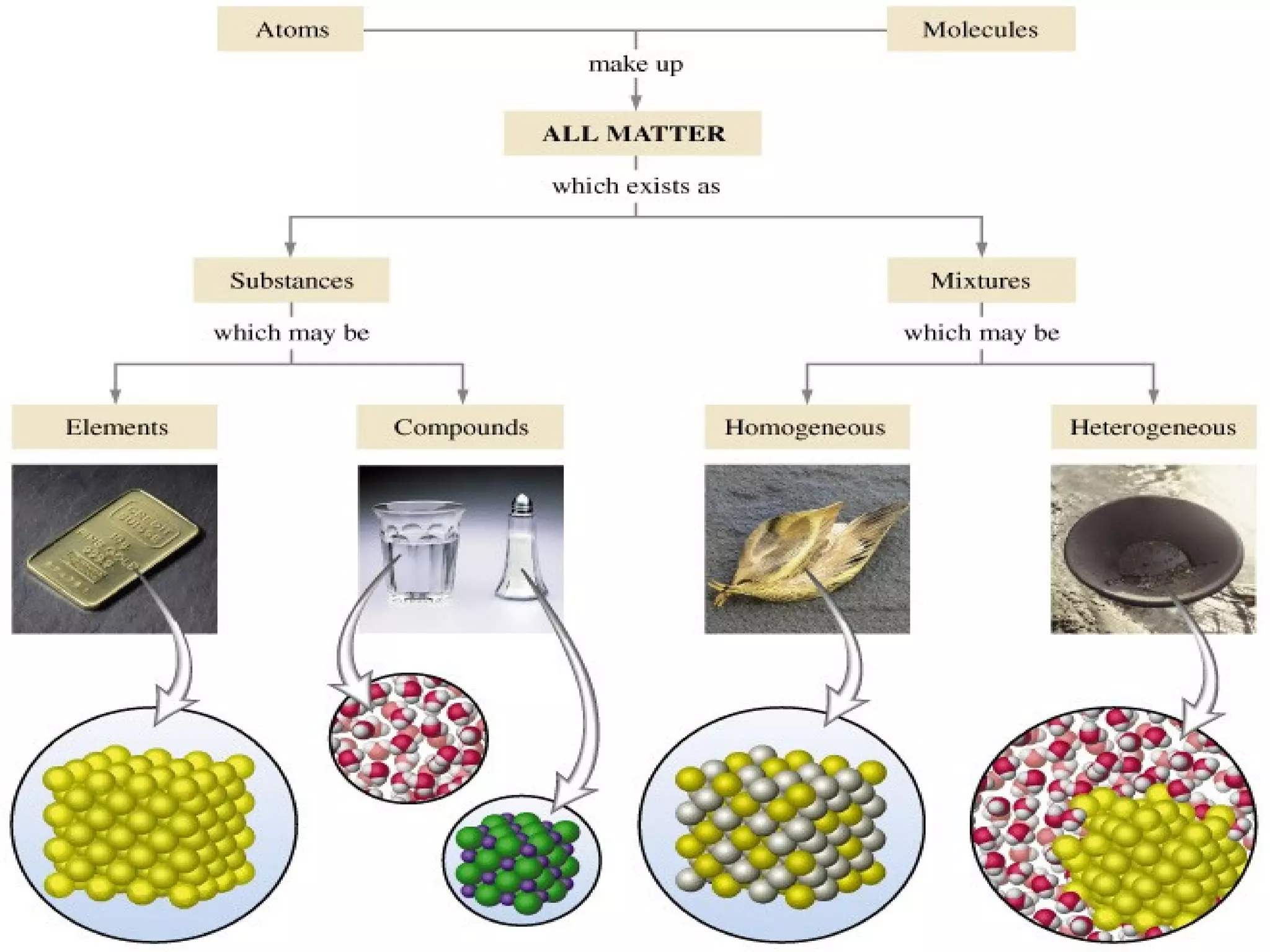
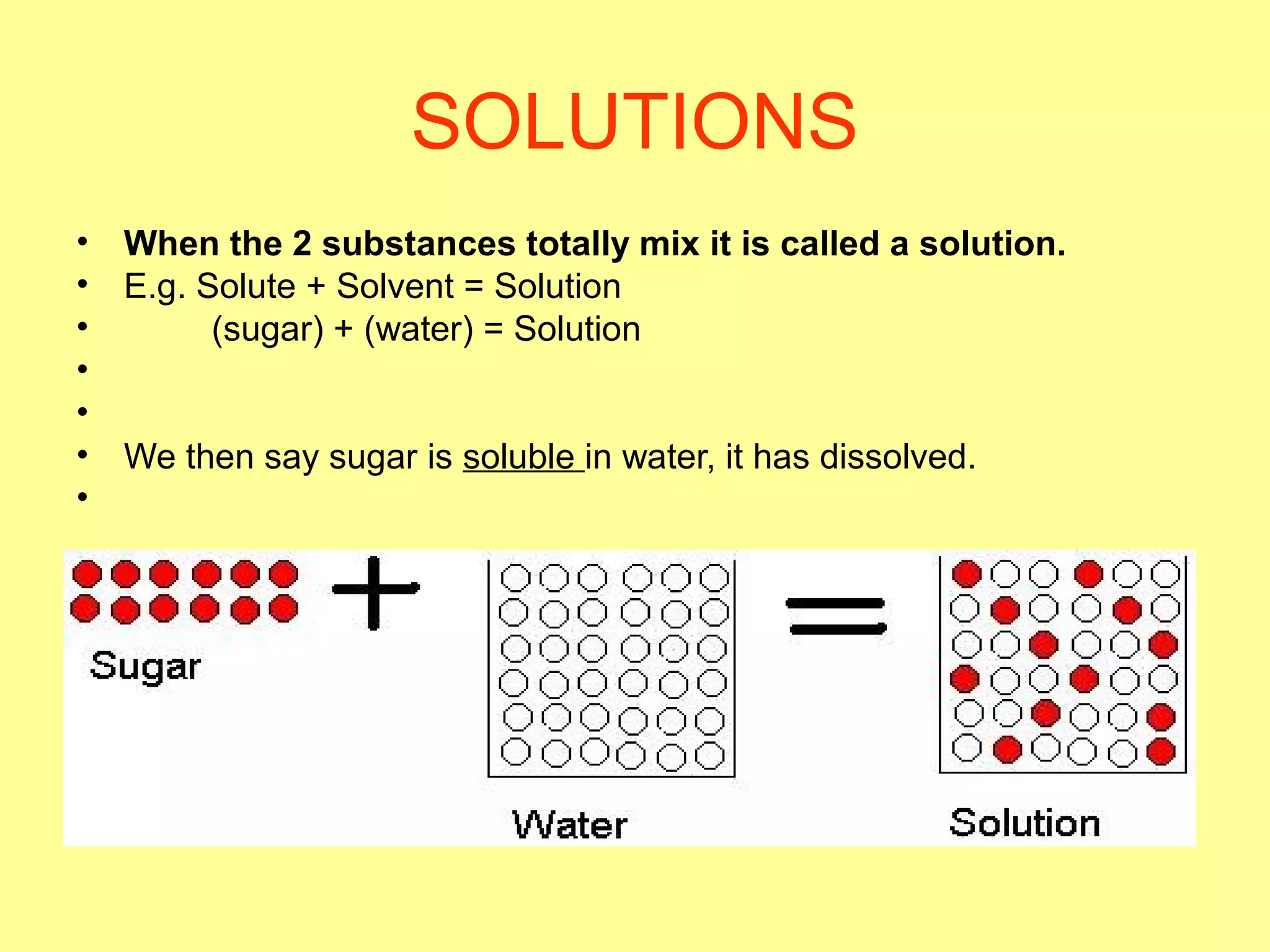
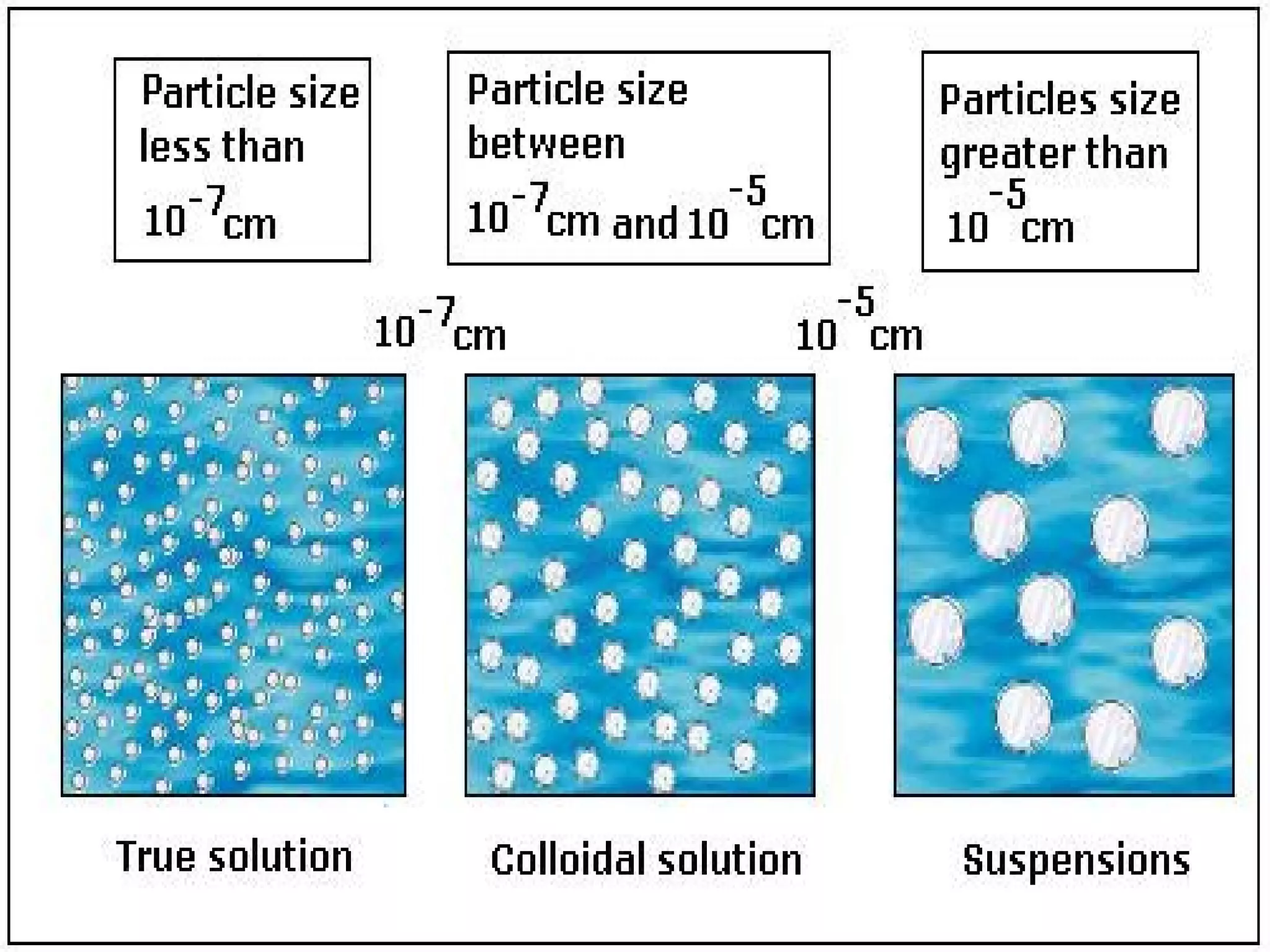
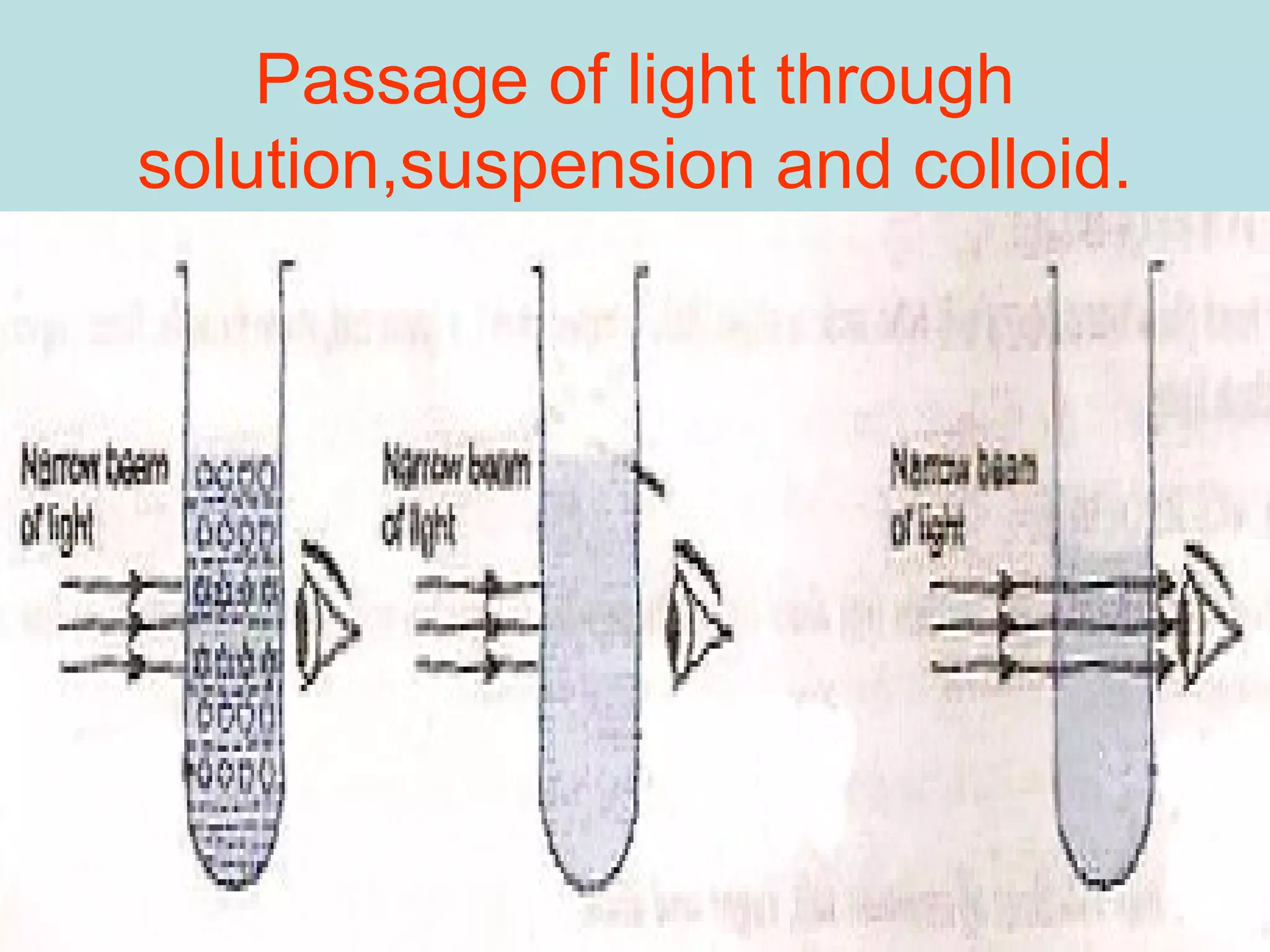
This document defines pure substances and mixtures. A pure substance is homogeneous and has definite properties, while a mixture contains two or more substances mixed together without chemical change. Solutions are homogeneous mixtures where particle size is molecular. Suspensions are heterogeneous mixtures where particle size is larger, allowing settling. Colloids have intermediate particle sizes that do not settle. The document discusses types of mixtures and their distinguishing characteristics.