lecture 3: 123.101
- 1. Unit One Parts 3 & 4: molecular bonding
- 2. Unit One Parts 3&4 H O H3C Br O Br H CH3 Locating electrons Describing bonds Pages Shape of molecules 34 & 43
- 3. Unit One 3&4 if we know where Parts electrons are we can predict reactions and shape...they really are key to understanding chemistry H O H3C Br O Br H CH3 Locating electrons Describing bonds Pages Shape of molecules 34 & 43
- 4. Unit One Parts 3&4 H O H3C Br O Br H CH3 as I’ve taken the material out of order, I’ll give you some Locating electrons page numbers Describing bonds Pages Shape of molecules 35 & 45
- 6. Na Cl Na Cl here we have an atom of sodium (Na) and an atom of Ionic bonds chlorine (Cl) Pg 34
- 7. if we take one electron from Na and give it to Cl... Na Cl Na Cl Ionic bonds Pg 34
- 8. Na Cl Na+ Cl- we get 2 charged species (cation = positive charge & anion = negative charge) Ionic bonds Pg 34
- 9. Na+ Cl- NaCl opposite charges attract and give us an ionic bond Ionic bonds Pg 34
- 10. covalent bonds H H if we bring 2 atoms together and they... Pg 34
- 11. covalent bonds H H share their 2 electrons we have a covalent bond Pg 34
- 12. covalent bonds H H this is the bond we’ll be dealing with most often and is represented by the black line H H 2 electrons per bond Pg 34
- 13. covalent bonds H H H H please remember 2 that this line is 2 electrons electrons per bond Pg 34
- 14. chemistry apain these are just extremes
- 15. reality is in the middle
- 16. where do we find electrons?
- 17. ONE DOES NOT SIMPLY
- 18. Aufbau Principle THIS IS THE LONG VERSION...NOT THE VERSION I DO IN THE LECTURES lowest energy orbital
- 19. Aufbau Principle don’t worry about the name...just that electrons like to have lowest energy possible... lowest energy orbital
- 20. rather like many students...
- 21. 1 18 1 H H He 2 13 14 15 16 17 Li Be B C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s1 energy 2s 2px 2py 2pz hydrogen Pg 1s 43
- 22. 1 18 1 H H He 2 13 14 15 16 17 Li Be B C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s1 energy 2s 2px 2py 2pz just one electron hydrogen so in first orbital Pg 1s 43
- 23. Pauli Exclusion Principle no two electrons are identical
- 24. 1 18 2 H He He 2 13 14 15 16 17 Li Be B C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s2 energy 2s 2px 2py 2pz helium Pg 1s 43
- 25. 1 18 2 H He He 2 13 14 15 16 17 Li Be B C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s2 energy 2s 2px 2py 2pz one electron has spin +½ (up) and the other spin –½ (down) helium Pg 1s 43
- 26. 1 18 2 H He He 2 13 14 15 16 17 Li Be B C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s2 energy 2s 2px 2py 2pz doesn’t matter what it means...just remember helium an electron can only be up or down Pg 1s 43
- 27. 1 18 2 H He He 2 13 14 15 16 17 Li Be B C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s2 energy 2s 2px 2py 2pz so can only ever have two electrons per orbital helium Pg 1s 43
- 28. 1 18 H He 2 13 14 15 16 17 3 Li Be B C N O F Ne Li Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 2px 2py 2pz 1s22s1 energy 2s lithium Pg 1s 43
- 29. 1 18 H He 2 13 14 15 16 17 3 Li Be B C N O F Ne Li Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 2px 2py 2pz 1s22s1 energy lithium obeys both rules...fill lowest orbital 2s first (until full) then fill next lowest) lithium Pg 1s 43
- 30. 1 18 H He 2 13 14 15 16one more ...adding 17 4 electron is easy... Li Be Be B C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 2px 2py 2pz 1s22s2 energy 2s beryllium Pg 1s 43
- 31. 1 18 H He 2 13 14 15 16 17 5 Li Be B B C N O F Ne Na Mg ...and another... Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 2px 2py 2pz 1s22s22p1 energy 2s boron Pg 1s 43
- 32. 1 18 H He 2 13 14 15 16 17 5 Li Be B B C N O F Ne Na Mg it could go in any of Al Si P S Cl Ar 3 4 5 6 7 8 9 10 x,11 y12 2pz, 2p 2p or they’re identical...well K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn they are As Se Br Kr energetically Ga Ge 2px 2py 2pz 1s22s22p1 energy 2s boron Pg 1s 43
- 33. 1 18 H He 2 13 14 15 16 17 5 Li Be B B C N O F Ne but, where does Na Mg the next (and most Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 important as its K Ca Sccarbon) go?? Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Ti V Cr 2px 2py 2pz 1s22s22p1 energy 2s boron Pg 1s 43
- 34. Hund's rule electrons as far apart as possible (de ge n er a t e o rb i tals) (as long as it doesn’t violate any of the previous rules!)
- 35. Hund's rule makes sense as like charges always repel... electrons as far apart as possible (de ge n er a t e o rb i tals)
- 36. 1 18 H He 2 13 14 15 16 17 6 Li Be B C C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s22s22p 12p 1 2px 2py 2pz x y energy 2s 1s22s22p2 carbon Pg 1s 43
- 37. 1 18 H He 2 13 14 15 16 17 6 Li Be B C C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s22s22p 12p 1 2px 2py 2pz could be 2pz, makes no x y difference... energy 2s 1s22s22p2 carbon Pg 1s 43
- 38. that's a lot of electrons... luckily we don’t care about all them...
- 39. all you have to remember is... ©jaci XIII@flickr
- 40. 1 18 H He 2 13 14 15 16 17 6 Li Be B C C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s22s22p2 carbon atomic = number of number electrons Pg 45
- 41. Valence electrons 1 18 H He 2 13 14 15 16 17 6 Li Be B C C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 1s22s22p 12p 1 2px 2py 2pz x y energy 2s 1s22s22p2 carbon Pg 1s 43
- 42. Valence electrons 1 18 H He 2 13 14 15 16 17 6 Li Be B C C N O F Ne Na Mg Al Si P S Cl Ar 3 4 5 6 7 8 9 10 11 12 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr only need consider high energy electrons or those on the outside called the 1s22s22p 12p 1 2px 2py 2pz valence electrons. x y energy 2s 1s22s22p2 carbon Pg 1s 44
- 43. C C if we consider the Bohr model of the atom, the 1s22s22p2 atomwhere we thinkplanet 2 one resembling a of an 2s22p with moons orbiting (or the solar system) N N 1s22s22p3 2s22p3 group 1 2 13 14 15 16 17 18 H He Li Be B C N O F Ne Pg 44
- 44. C C 1s22s22p2 2s22p2 then the valence electrons are those on the outer edge (like Neptune for young-upstarts or Pluto for us oldies) N N 1s22s22p3 2s22p3 group 1 2 13 14 15 16 17 18 H He Li Be B C N O F Ne Pg 44
- 45. C C 1s22s22p2 2s22p2 then the valence electrons are those on the outer edge (like Neptune for young-upstarts or Pluto for us oldies) N N 1s22s22p3 2s22p3 group 1 2 13 14 15 16 17 18 H He Li Be B C N O F Ne Pg 44
- 46. C C 1s22s22p2 2s22p2 N N 1s22s22p3 2s22p3 absolute rubbish...but more group 1 2 13 14 15 16 17 18 comprehendible! H He Li Be B C N O F Ne Pg 41
- 47. C C 1s22s22p2 2s22p2 N N an easy we to remember the number of valence electrons is 1s22s22p3 2s22p3 to take group number... group 1 2 13 14 15 16 17 18 H He Li Be B C N O F Ne Pg 41
- 48. C C 1s22s22p2 2s22p2 N N ...and ignore 1s22s22p3 2s22p3 first ‘1’ valence electrons 1 2 3 4 5 6 7 8 H He Li Be B C N O F Ne Pg 41
- 49. C C 1s22s22p2 2s22p2 N N 1s22s22p3 2s22p3 valence electrons 1 2 3 4 5 6 7 8 so oxygen (group 16) has H He 6 valence Li Be B C N O F Ne electrons Pg 41
- 50. what do valence electrons tell us?
- 51. H 1 2 H H 4 3 O H the number of bonds
- 52. the shape of molecules 109°
- 53. ? how
- 54. atoms are happy if they have a full valence shell... Ne 1s22s22p6 noble gas
- 55. Ne ...commonly this means 8 electrons 1s22s22p6 noble gas
- 56. 8 electrons Ne full shell
- 57. C 1s22s22p2 4 bonds N 1s22s22p3 3 bonds O 1s22s22p4 2 bonds Pg 36
- 58. Pg 45 C 4 valence electrons so for carbon to get to 8 it needs 4 more electrons 1s22s22p2 Pg 45
- 59. Pg 46 C or 4 new covalent bonds 4 bonds Pg 46
- 60. C nitrogen has 5 valence1s22s22p2 electrons...so 4 bonds needs 3 more... N 1s22s22p3 3 bonds O 1s22s22p4 2 bonds Pg 36
- 61. N 5 valence electrons 1s22s22p3 Pg 46
- 62. so forms 3 covalent bonds N 3 bonds Pg 46
- 63. C 1s22s22p2 4 bonds oxygen needs 2 more electrons so forms 2 covalent bonds N 1s22s22p3 3 bonds O 1s22s22p4 2 bonds Pg 36
- 64. O 6 valence electrons 1s22s22p4 Pg 46
- 65. O 2 bonds Pg 46
- 66. C 1s22s22p2 4 bonds hopefully, you can see 3 this is where those magic numbers in lecture one came N bonds from! 1s22s22p3 O 1s22s22p4 2 bonds Pg 34
- 67. Pg 8 36 H H C H H Octet rule: 8 valence electrons
- 68. Pg 8 37/46 H H H C N O H H Octet rule: 8 valence electrons
- 69. Pg Lewis structures 37/46 Hydrofluoric acid HF H + F H F ≡ H F use octet rule to draw Methanol CH OH 3 the structure of stable molecules... H H C + O + 4H H C O H ≡H C O H H H
- 70. Pg Lewis structures 41 Hydrofluoric acid HF H–F easy..H = 2 electrons (full s orbital) & F = 8... H + F H F ≡ H F Methanol CH3OH H H C + O + 4H H C O H ≡H C O H H H
- 71. Pg Lewis structures 37/46 Lewis structure shows all valence electrons Hydrofluoric acid HF represented by our simple diagram H–F H + F H F ≡ H F Methanol CH3OH H H C + O + 4H H C O H ≡H C O H H H
- 72. Pg Lewis structures 37/46 Hydrofluoric acid HF H + F H F ≡ works for more complex H F molecules Methanol CH3OH H H C + O + 4H H C O H ≡H C O H H H
- 73. Pg Lewis structures 37/46 Hydrofluoric acid HF H + F H F ≡ H F Methanol CH3OH Note: it helps to leave lone pairs (of electrons) on diagram...this is where a lot of chemistry occurs... H H C + O + 4H H C O H ≡H C O H H H
- 74. Acetone CH3COCH3 3 C + O + 6H how do we deal with more complex molecules? Pg 44
- 75. Acetone CH3COCH3 3 C + O + 6H first draw all the atoms where you think O they might go... H H C H C C H H H Pg 44
- 76. Acetone CH3COCH3 3 C + O + 6H now join all the atoms together...some of the atoms have full valence shells so we can draw them in as on O the next slide... H C H C C H H H H Pg 44
- 77. Acetone CH3COCH3 3 C + O + 6H the central C and O both have only 7 valence electrons... O H C H C C H H H H Pg 44
- 78. Acetone CH3COCH3 3 C + O + 6H O ...but if they share 4 electrons they both have H C H 8 valence electrons...this C C gives us a double bond H H (alkene) H H O O C ≡ Pg H3C CH3 44
- 79. – Borohydride anion BH4 – what happens if we have a negative charge (anion)? Pg 44
- 80. – Borohydride anion BH4 – take the atoms as normal and... B + 3H + H Pg 44
- 81. – Borohydride anion BH4 – add electron B + 3H + H ...add an electron Pg 44
- 82. – Borohydride anion BH4 – add electron H H B + 3H + H H B H ≡ H B H H H Pg 44
- 83. – Borohydride anion BH4 – add electron does it matter which atom we give the electron to? H H B + 3H + H H B H ≡ H B H H H Pg 44
- 84. – Borohydride anion BH4 – add electron does it matter which atom we give the electron to? H H B + 3H + H H B H ≡ H B no! (but in this case H H H– makes more H chemical sense) Pg 44
- 85. + Ammonium cation NH4 + lose electron if we have a positive charge (cation) we do the opposite... Pg 44
- 86. + Ammonium cation NH4 + lose electron start with our normal atoms... N + 3H + H Pg 44
- 87. + Ammonium cation NH4 + lose electron N + 3H + H then remove an electron Pg 44
- 88. + Ammonium cation NH4 + lose electron H H N + 3H + H H N H ≡ H N H H H Pg 44
- 89. where is the charge? is it on one atom?
- 90. all over the molecule... No, its all over the molecule! But...
- 91. but the truth isn't useful, so...
- 92. formal charges localise charge on an atom...
- 93. this is ‘electron book- keeping’...we are just assigning charge to one atom to help explain chemistry... formal charges localise charge on an atom...
- 94. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons ...on an atom Pg 47
- 95. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons ...according to the atoms position in the periodic table Pg 47
- 96. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons ...in lone pairs... Pg 47
- 97. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons ...or the number of bonds to that atom Pg 47
- 98. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons H H N + 3H + H H N H ≡ H N H H H cation N fc = 5-0-½(8)=+1 Pg 47
- 99. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons H H N + 3H + H H N H ≡ H N H H H no charge on H as: cation H = 1-0-½(2) = 0 N fc = 5-0-½(8)=+1 Pg 47
- 100. formal number of number of number of charge = valence – unshared – bonds (fc) electrons electrons H H the simplified N + 3Hformula of bonds) + (just use number H H N H ≡ H N H H H cation N fc = 5-0-4=+1 Pg 47
- 101. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons O O + O + O O O O ≡ O O O3 neutral ozone Pg 47
- 102. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons O O + O + O O O O ≡ O O O3 neutral ozone lhs O; fc = 6-4-½(4)=0 Pg 47
- 103. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons O O + O + O O O O ≡ O O O3 neutral ozone lhs O; fc = 6-4-½(4)=0 central O; fc = 6-2-½(6)=+1 rhs O; fc = 6-6-½(2)=-1 Pg 47
- 104. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons O O + O + O O O O ≡ O O O3 neutral ozone lhs O; fc = 6-4-½(4)=0 central O; fc = 6-2-½(6)=+1 rhs O; fc = 6-6-½(2)=-1 Pg 47
- 105. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons O O + O + O O O O ≡ O O ≡ O O O O3 neutral atom's formal ozone charges lhs O; fc = 6-4-½(4)=0 central O; fc = 6-2-½(6)=+1 rhs O; fc = 6-6-½(2)=-1 Pg 47
- 106. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons O O + O + O O O O ≡ O O ≡ O O O ozone neutral as O3 neutral atom's formal + & – cancel each ozone charges other out lhs O; fc = 6-4-½(4)=0 central O; fc = 6-2-½(6)=+1 rhs O; fc = 6-6-½(2)=-1 Pg 47
- 107. formal number of number of ½ number charge = valence – unshared – of shared (fc) electrons electrons electrons these charges explain why ozone is so reactive! O O + O + O O O O ≡ O O ≡ O O O O3 neutral atom's formal ozone charges lhs O; fc = 6-4-½(4)=0 central O; fc = 6-2-½(6)=+1 rhs O; fc = 6-6-½(2)=-1 Pg 47
- 108. formal number of number of number of charge = valence – unshared – bonds (fc) electrons electrons O O + O + O O O O ≡ O O ≡ O O O O3 neutral atom's formal the simplified ozone charges formula (just use number of bonds) lhs O; fc = 6-4-2=0 central O; fc = 6-2-3=+1 rhs O; fc = 6-6-1=-1 Pg 47
- 109. a bond O 2 H O is electrons
- 110. a bond 2 is electrons
- 111. simple model
- 112. quantum model more accurate...
- 113. Atomic orbitals it's a quantum world... we’ve looked at a nice simple model so far...
- 114. Atomic orbitals it's a quantum world... mathematicians and physicists have shown it’s a bit more complicated in ‘reality’
- 115. but I don't like maths... so...here's some pretty pictures...
- 116. 90% atomic orbital atomic orbital is the volume of space in which there is a 90% chance of finding an electron Pg 36
- 117. 2 remember: a atomic orbital maximum of 2 electrons per orbital electrons Pg 37
- 118. a 1s orbital is also a sphere...just a lot smaller 2s Pg 38 Pic: Dr. Jonathan Gutow
- 119. let’s ignore this nasty little effect of maths... 2s Pg 38 Pic: Dr. Jonathan Gutow
- 120. px z py z pz z y y y x x x 2p Pg 38
- 121. px z py z pz z y y y x x x 2p each of the three 2p orbitals is dumbbell shaped... Pg 38
- 122. px z py z pz z y y y x x x 2p ...they are identical in all ways except... Pg 38
- 123. px z py z pz z y y y x x x 2p ...they point in different directions (hence the names) Pg 38
- 124. px z py z pz z y y y this is one orbital (just has two different x x coloured areas) x 2p Pg 34
- 125. afraid?
- 126. you will be...
- 127. our simple Lewis model helps explain a lot of chemistry...especially reactions... what is a bond?
- 128. what is a bond? ...but it fails to explain such fundamental concepts as shape...
- 129. ...actually, it can explain shape if we use VSEPR theory...but anyways, lets use those orbitals what is a bond?
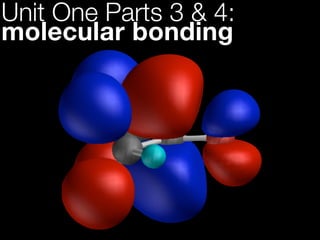
- 130. single (σ) bond H• + H• H H energy here we have 2 hydrogen atoms (each with 1 electron in a 1s orbital) Pg H• 1s H• 1s 37
- 131. single (σ) bond H• + H• H H σ* to form a covalent bond they must energy share their electrons... σ Pg H• 1s H–H H• 1s 35
- 132. single (σ) bond H• + H• H H σ* energy ...this is achieved by combining the two atomic Pg orbitals to give... σ H• 1s H–H H• 1s 35
- 133. single (σ) bond H• + H• H H σ* ...a new molecular orbital, a sigma σ orbital (or bond) energy σ Pg H• 1s H–H H• 1s 35
- 134. single (σ) bond H• + H• H H ...this bonding σ* orbital is lower in energy than the atoms...so a bond will form energy σ Pg H• 1s H–H H• 1s 35
- 135. single (σ) bond H• + H• H H a consequence of the maths is we also get an anti-bonding sigma orbital (σ*)...2 orbitals σ* must give 2 new orbitals energy σ Pg H• 1s H–H H• 1s 37
- 136. single (σ) bond H• + H• H H σ* energy ...but lets ignore this confusing little devil for the time being! σ Pg H• 1s H–H H• 1s 37
- 137. single (σ) bond it is called a σ orbital as is symmetrical along bond axis (you can rotate it like a cylinder and it doesn’t change) Pg H H 47
- 138. single (σ) bond all bonds to H are sigma (as all are like a cylinder)...here we overlap 1s of H with 2p of C and get sigma bond) C• + H• C H Pg 37
- 139. Pg single (σ) bond 38 σ* energy if we take two 2p orbitals and combine them head-to-head C• σ C• 2py C–C 2py
- 140. Pg single (σ) bond 38 σ* ...we get a sigma σ bonding orbital...it is still energy like a cylinder... C• σ C• 2py C–C 2py
- 141. Pg single (σ) bond 38 σ* energy ...this is the normal single bond we observe in alkanes etc. C• σ C• 2py C–C 2py
- 142. Pg single (σ) bond 38 σ* this is one orbital NOT three energy C• σ C• 2py C–C 2py
- 143. single (σ) bond the blue bit is the sigma orbital...ignore Pg the red orbitals for the time being... 35
- 144. single bond or the simple C C version... THIS IS ALL YOU NEED TO KNOW σ (sigma) bond
- 145. single bond σ (sigma) bond
- 146. single bond σ (sigma) bond
- 147. sp3 C an atom with 4 σ bonds is called an sp3 atom (as 1 x s and 3 x Pg p used in bonding) 38
- 148. sp3 1 YOU NEED TO KNOW THIS 4 2 4 points 3
- 149. tetrahedral H Br C Br H 109° sp3 Pg 41
- 150. tetrahedral H C Br Br H sp3 atoms are tetrahedral in shape (the bonds stay as far apart as possible) 109° sp3 Pg 41
- 151. tetrahedral sp3 maximum separation of four points maximum separation of four valence electron pairs
- 152. Pg double (σ + π) bonds 38 C C C=C π* energy C=C π two 2p orbitals can combine side-to-side carbon carbon 2pz C C 2pz
- 153. Pg double (σ + π) bonds 38 C C C=C π* energy the new bond is a pi π bond C=C π carbon carbon 2pz C C 2pz
- 154. Pg double (σ + π) bonds 38 C C C=C π* energy C=C π here we have a C–C σ bond and carbon a pi π bond carbon 2pz C C 2pz
- 155. Pg double (σ + π) bonds 38 C C C=C π* energy C=C π the pi π bond is one orbital (with two bits to it) carbon carbon 2pz C C 2pz
- 156. double (π) bond Pg 38
- 157. double (π) bond it is called a pi π orbital as rotation around the C–C axis causes a change (from red to blue) so no longer like a cylinder Pg 38
- 158. double (π) bond remember: this is ONE orbital (just two different coloured halves) Pg 35
- 159. double (π) bond we have an inner σ bond (the rod) and an outer π bond (the orbital) hence it is a double bond Pg 38
- 160. no rotation
- 161. H3C CH3 CH3 CH3 CH3 the p bond prevents O H alkenes from rotating (the two bonds can’t twist pass multistep enzyme- each other)... light isomerises catalysed reverse complexed process cis-retinal H3C CH3 CH3 CH3 O H CH3 Pg 38
- 162. H3C CH3 CH3 CH3 CH3 this can effect O H shape of molecule multistep enzyme- light isomerises catalysed reverse complexed process cis-retinal H3C CH3 CH3 CH3 O H CH3 Pg 38
- 163. H3C CH3 CH3 CH3 CH3 O H we must break π bond before multistep enzyme- light isomerises catalysed reverse complexed alkene can rotate process cis-retinal H3C CH3 CH3 CH3 O H CH3 Pg 38
- 164. H3C CH3 CH3 CH3 CH3 the change in O H shape initiates the visual cascade and multistep enzyme- light isomerises our sight catalysed reverse complexed process cis-retinal H3C CH3 CH3 CH3 O H CH3 Pg 38
- 165. H3C CH3 CH3 CH3 CH3 O H multistep enzyme- light isomerises catalysed reverse complexed process cis-retinal H3C CH3 CH3 CH3 O why do you think red path is easy H but blue hard? CH3 Pg 38
- 166. double bond or the simple version... THIS IS ALL YOU NEED TO KNOW π (pi) bond
- 167. double bond π (pi) bond
- 168. sp2 an atom with three σ orbitals and one π orbital is called an sp2 atom (we only count the C orbitals used in making Pg s orbitals) 38
- 169. sp2 1 3 3 2 1 double bond and 2 single bonds and we points have an sp2 atom
- 170. trigonal planar 120° sp2 atoms are trigonal planar sp2 (flat and pointing to the corners of a triangle)...again, this is because the orbitals Pg try to be as far apart as possible 41
- 171. trigonal Pg 41 planar sp 2 maximum separation of three points maximum separation of three valence electron pairs
- 172. triple (σ + 2x π) bonds σ H C C H π (2pz + 2pz) σ a triple bond (like an π (2py + 2py) alkyne) is formed from one σ bond and two π bonds (at right angles to each other due to the direct of the p orbitals that made them) σ π H C C π H Pg 39
- 173. triple (σ + 2x π) bonds so...two p orbitals combine head-to-head to give a σ bond and two pairs of p orbitals combine side-to-side to give the two π orbitals (& there are only two π orbitals) σ H C C H π (2pz + 2pz) σ π (2py + 2py) σ π H C C π H Pg 39
- 174. sp an atom with two σ orbitals and two π orbitals is called an sp atom (as two orbitals made the basic σ skeleton) C Pg 39
- 175. sp 1 2 2 points
- 176. linear an atom with two groups on it will be 180° sp Pg linear (a straight line) as the orbitals stay as far apart as possible 40
- 177. linear sp maximum separation of two points maximum separation of two valence electron pairs
- 178. H3C H CO2H OH O O OCH3 H here is a real OH O molecule...we should be dynemicin A able to identify the types of atoms present... Pg 40
- 179. four groups attached so it must be sp3 and as those groups try to stay as far apart as possible it is tetrahedral H3C H CO2H OH O O OCH3 H OH O dynemicin A sp3 tetrahedral Pg 40
- 180. ...only three groups so sp2 and flat, trigonal planar H3C H CO2H OH O O sp2 OCH3 trigonal H planar OH O dynemicin A sp3 tetrahedral Pg 40
- 181. sp linear straight line, two groups must be sp H3C H and linear CO2H OH O O sp2 OCH3 trigonal H planar OH O dynemicin A sp3 tetrahedral Pg 40
- 182. what is oxygen? H3C H CO2H OH O O OCH3 H OH O dynemicin A Pg 40
- 183. ...is it sp as what is attached to two oxygen? carbon atoms? H3C H CO2H OH O O OCH3 H OH O dynemicin A Pg 40
- 184. sp, sp2 or sp3? H O look at a simpler system...water, sp, H sp2 or sp3?
- 185. sp, sp2 or sp3? H O H draw Lewis structure...
- 186. sp, sp2 or sp3? H O H we have FOUR groups around O, two lone pairs & two H atoms. So it is...
- 187. tetrahedral H O H sp3
- 188. tetrahedral H O H sp3 that is why we draw water as a bent molecule...its shape is based on a tetrahedron...
- 189. tetrahedral H O ...any atom with four atoms or lone pairs around it is sp3 with all that entails! H sp3
- 190. sp, sp2 or sp O 3? what kind of atom is the oxygen? C H H
- 191. 1 double bond O ...and two lone pairs, so three groups around the C oxygen so it is... H H
- 192. trigonal planar H O C H sp2
- 193. sp, sp2 or sp3? N C H what kind of atom is the nitrogen?
- 194. 1 triple bond N C H and one lone pair so two groups so it is...
- 195. linear sp N C H
- 196. what have ....we learnt? •e l e c t r o n s where they are •b o n d s what they are Courtesy: National Science Foundation •s h a p e
- 197. Read Pages 36, 41, 48-57 ©rachel_titiriga@flickr
