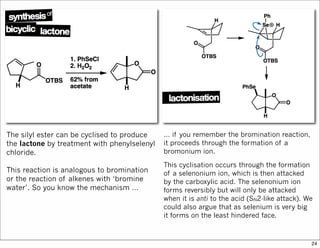
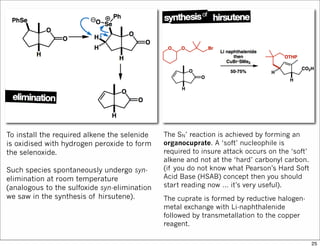
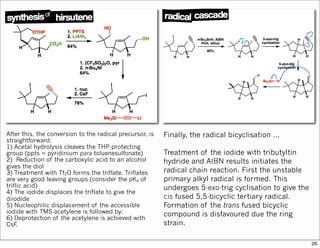
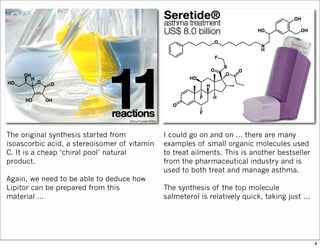
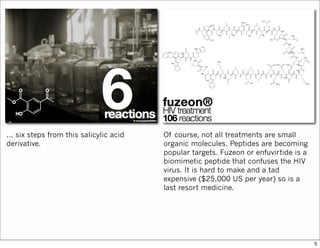
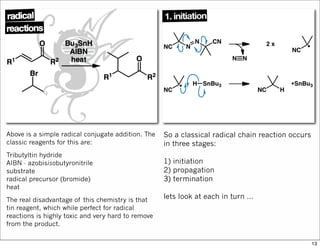
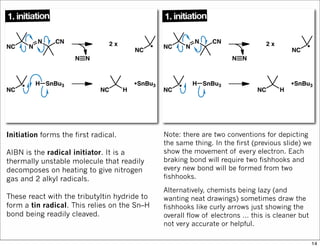
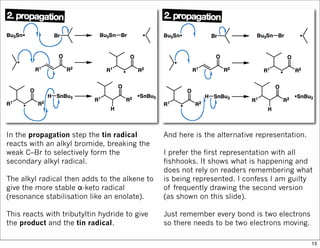
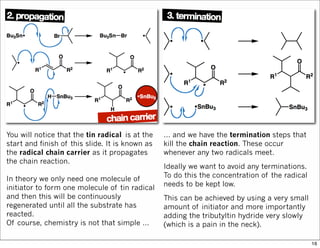
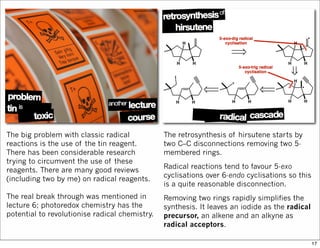
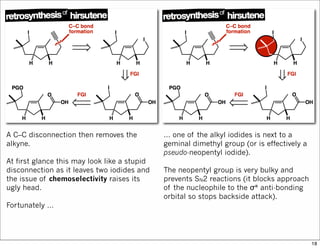
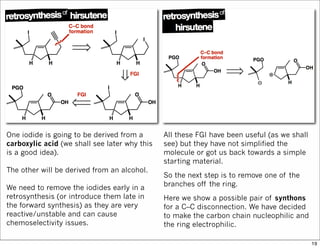
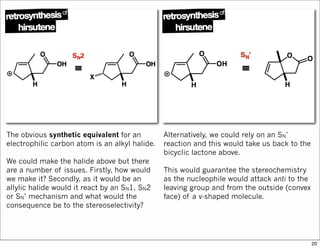
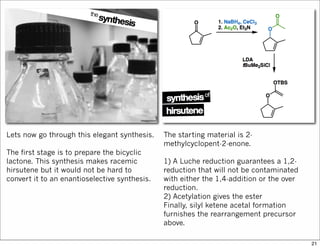
The document outlines a lecture on organic synthesis, focusing on methods for creating complex molecules like oseltamivir and atorvastatin using various synthetic pathways. It discusses the significance of radical chemistry in synthesizing compounds while showcasing specific examples and methodologies, including advantages and challenges associated with these techniques. Additionally, it emphasizes the need for innovative synthetic strategies to enhance access to essential medicines.









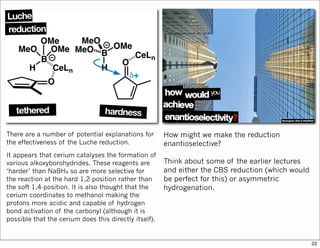
![Heating the silly ketene acetal to reflux
initiates an Ireland-Claisen rearrangement.
This is a pericyclic reaction, specifically a
concerted [3,3]-sigmatropic
rearrangement. Such reactions are
stereospecific.
You can recognise the possibility of such
rearrangements just by looking for two
multiple bonds whose terminals are 6
atoms apart.
Normally, such rearrangements occur
through a chair-like transition state and you
can use this to rationalise the
stereochemical communication.
This is shown above.
More information can be found about such
rearrangements here:
Stereoselective Synthesis - lecture 11 or
Advanced Organic Synthesis - lecture 8
23](https://image.slidesharecdn.com/123713ablecture09-161117235844/85/123713AB-lecture09-23-320.jpg)