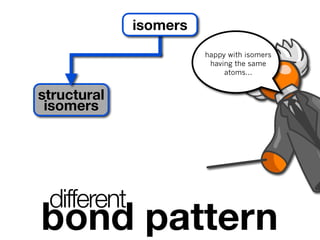
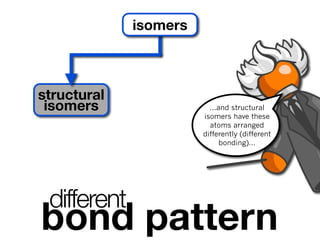
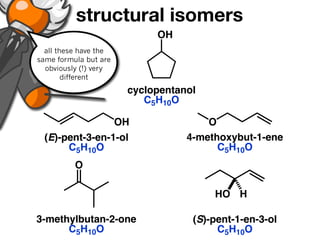
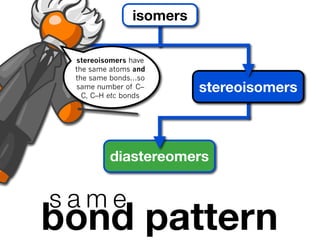
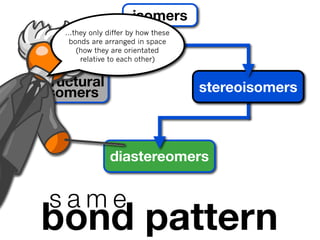
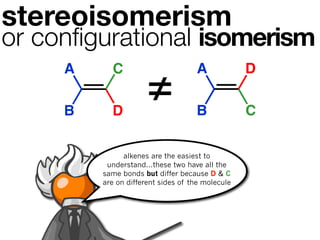
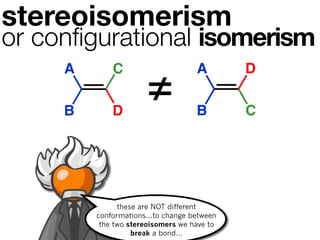
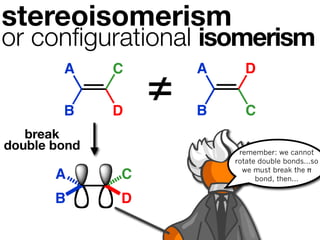
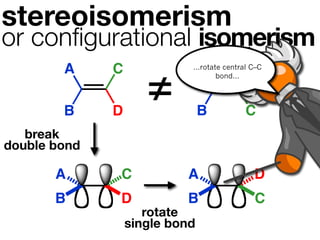
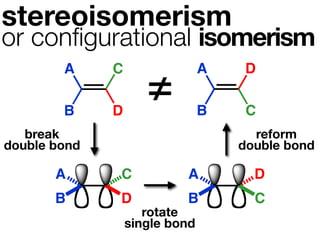
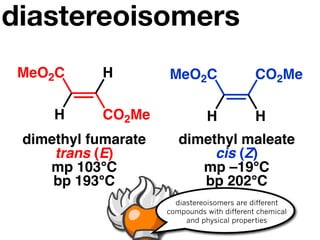
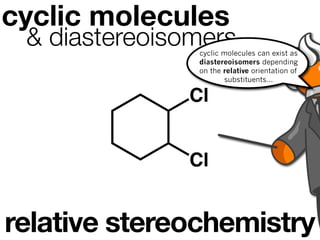
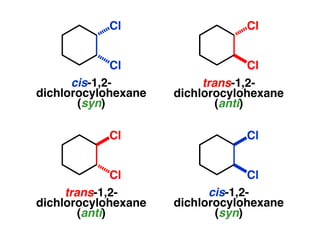
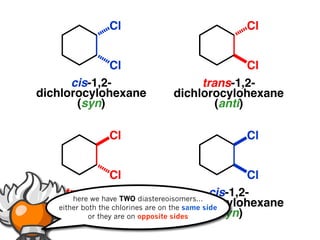
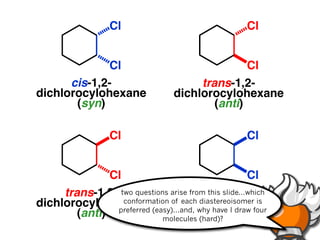
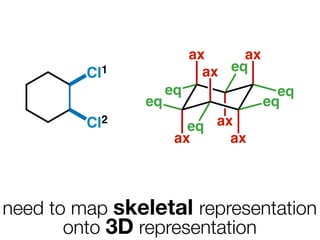
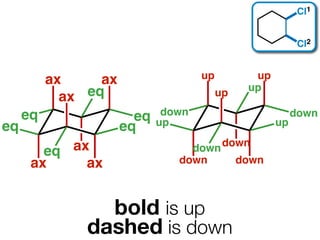
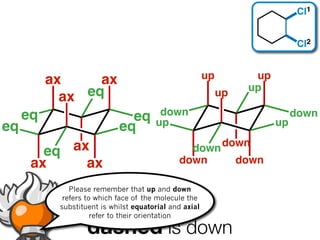
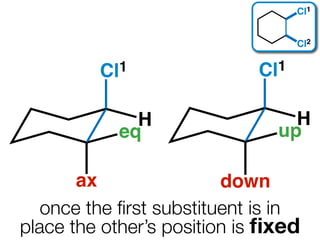
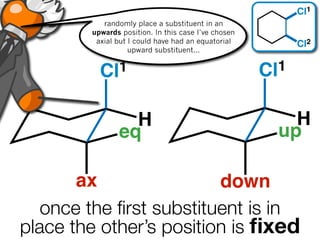
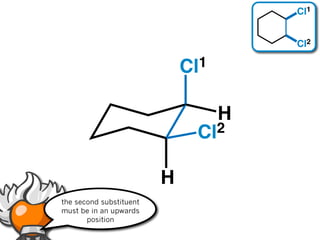
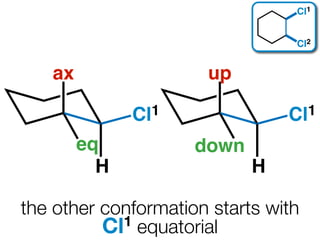
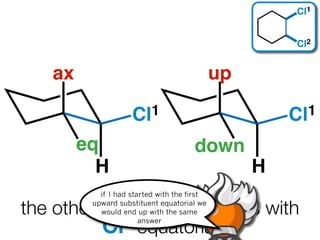
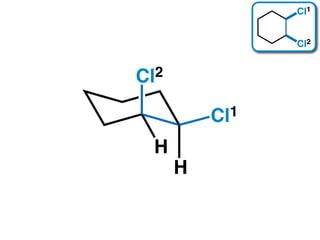
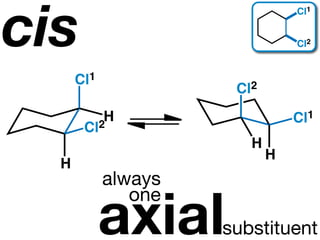
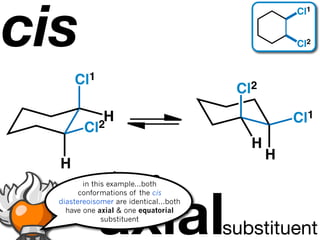
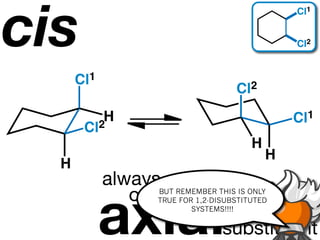
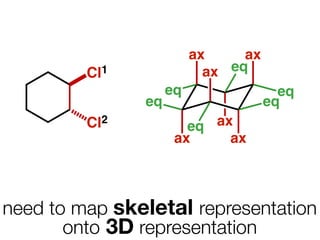
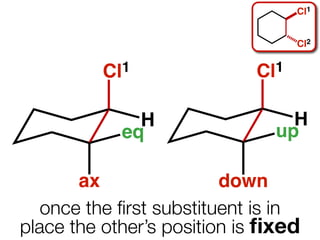
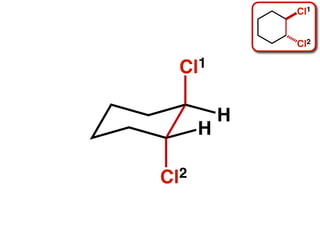
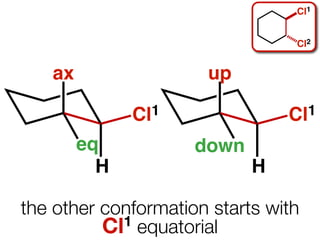
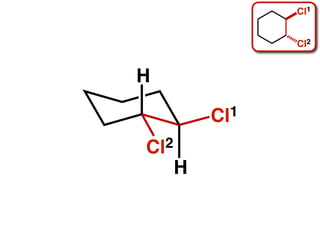
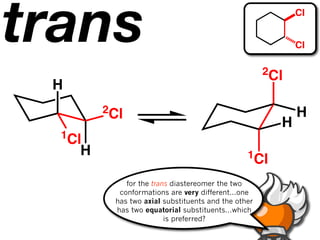
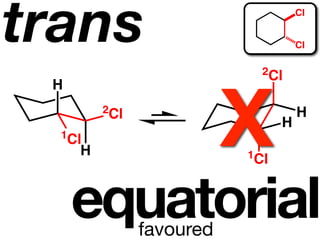
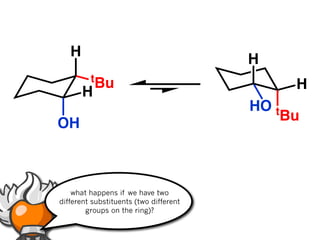
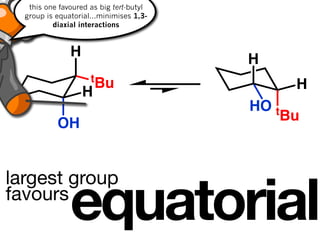
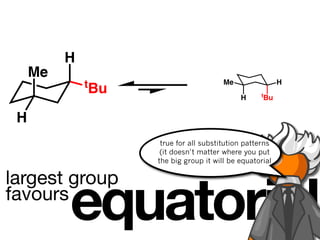
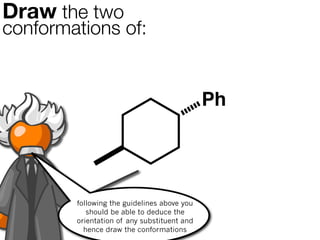
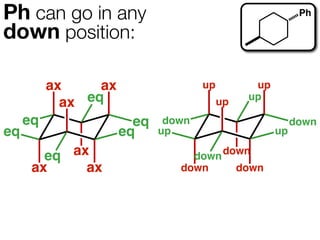
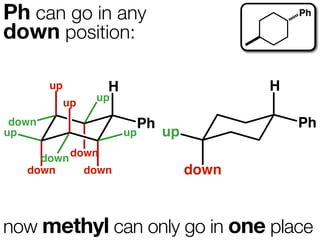
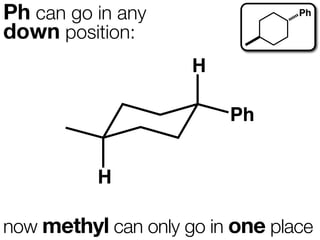
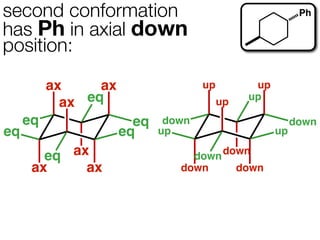
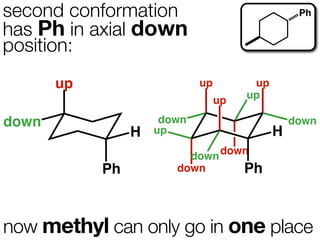
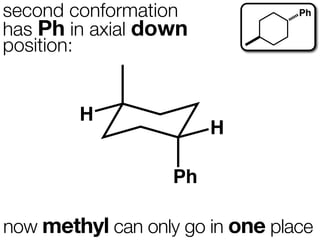
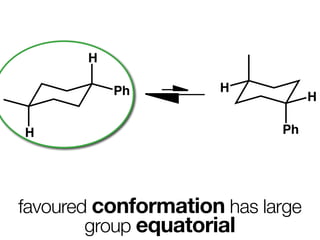
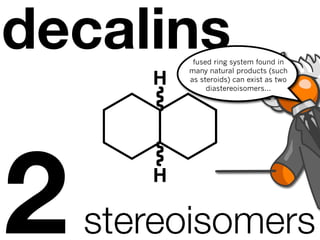
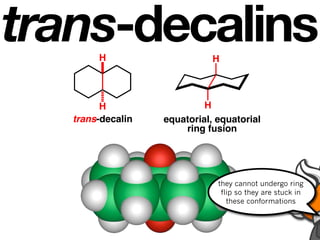
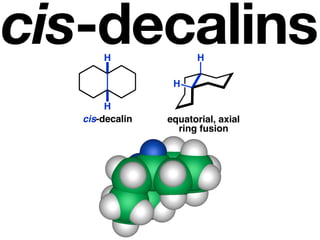
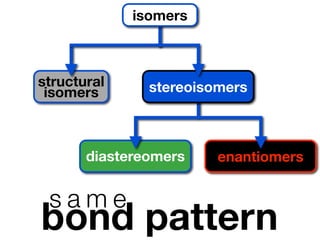
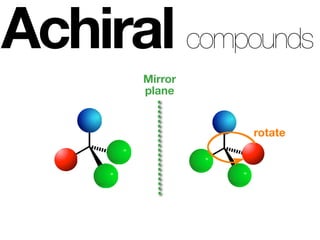
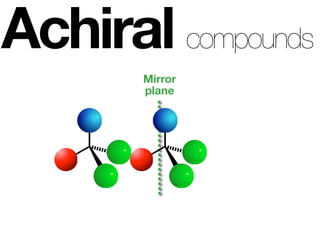
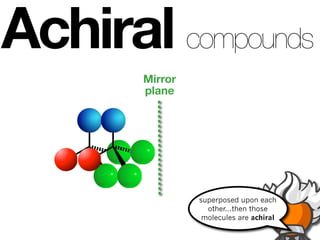
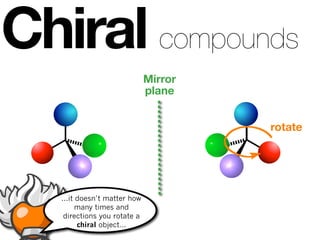
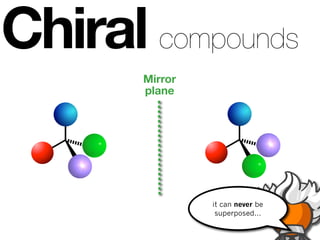
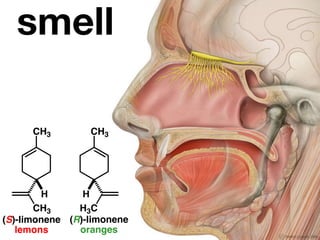
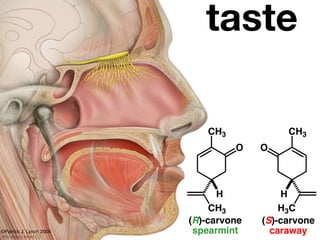
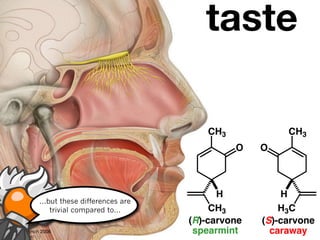
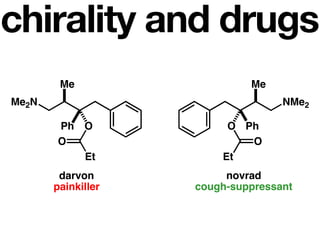
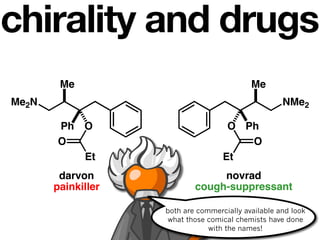
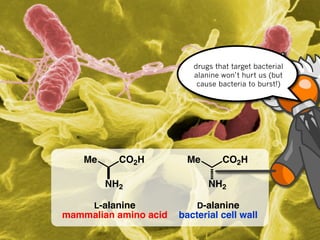
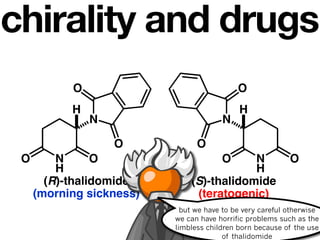
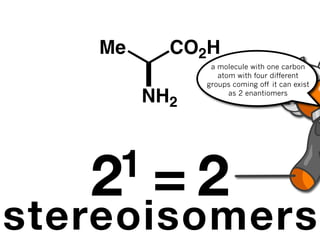
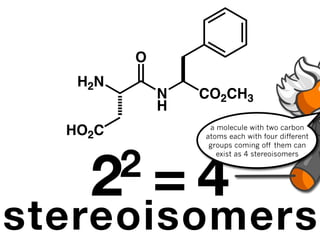
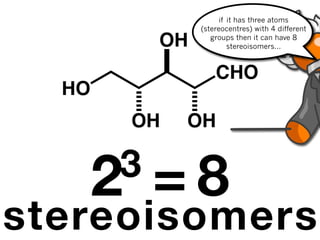
The document discusses stereochemistry and different types of isomers. It introduces structural isomers which have different bonding patterns and stereoisomers which have the same bonding but different spatial arrangements. Stereoisomers can be diastereomers or enantiomers. Diastereomers have different physical properties while enantiomers are non-superimposable mirror images and have identical physical properties. The document uses examples like cyclic molecules and decalins to illustrate these concepts.




















![physical properties
α
light light (λ) polariser plane sample reading
source polarised light cell length l (dm)
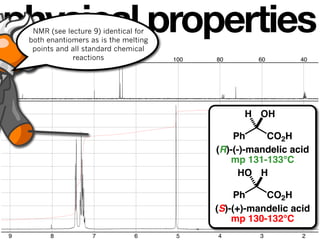
H OH HO H
Ph CO2H Ph CO2H
(R)-(-)-mandelic acid (S)-(+)-mandelic acid
[α]23 –153
D [α]23 +153
D](https://image.slidesharecdn.com/lecture8-090308232521-phpapp01/85/Lecture8-123-101-83-320.jpg)
![physical properties
α
each enantiomer rotates plane
polarised light in a different
direction and more importantly...
light light (λ) polariser plane sample reading
source polarised light cell length l (dm)
H OH HO H
Ph CO2H Ph CO2H
(R)-(-)-mandelic acid (S)-(+)-mandelic acid
[α]23 –153
D [α]23 +153
D](https://image.slidesharecdn.com/lecture8-090308232521-phpapp01/85/Lecture8-123-101-84-320.jpg)






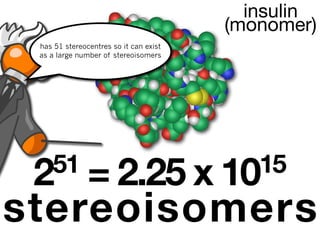





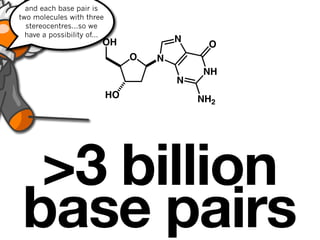




![9
read
part ©[auro]@flickr](https://image.slidesharecdn.com/lecture8-090308232521-phpapp01/85/Lecture8-123-101-112-320.jpg)