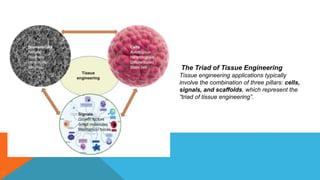
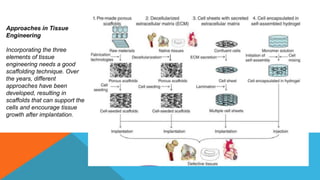
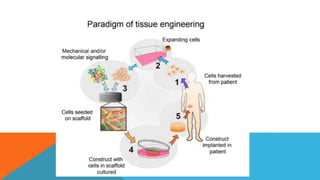
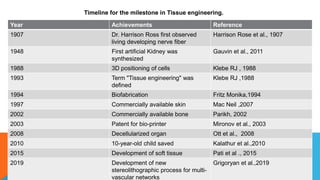
Tissue engineering is an interdisciplinary field that combines engineering and life sciences to create biological substitutes that restore or enhance tissue function. With a growing demand for organ replacements and limited donor availability, tissue engineering aims to mitigate healthcare challenges by using living cells, scaffolds, and signals to regenerate tissues and organs. Applications span various medical fields, including skin, liver, pancreas, heart, blood vessels, nervous system, and bone regeneration, while also extending to drug testing and personalized medicine.