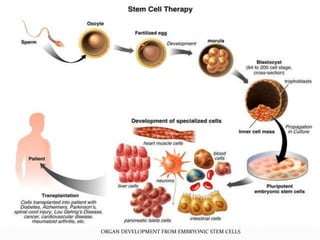
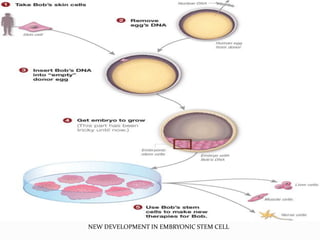
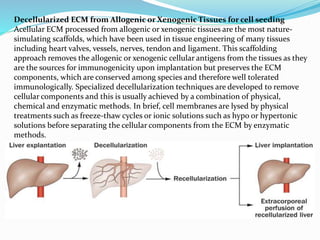
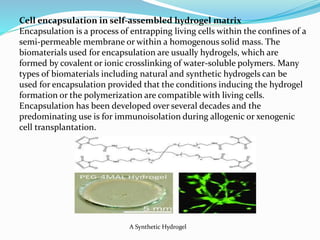
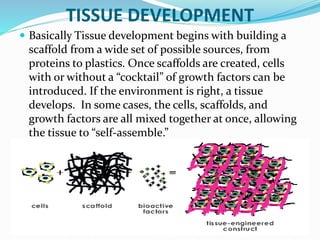
The document discusses tissue engineering, focusing on the combination of scaffolds, cells, and biochemical factors to create functional tissues for the repair and regeneration of damaged tissues or organs. It highlights the significance of stem cells, particularly embryonic stem cells and mesenchymal stem cells, in developing tissues and various scaffolding approaches utilized in tissue engineering. The document concludes that ongoing advancements in tissue engineering and regenerative medicine may lead to enhanced clinical applications for tissue replacement therapies.