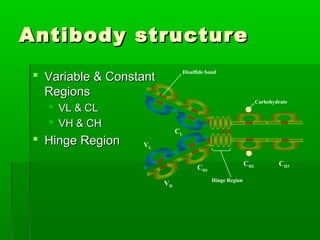
The plasma is the liquid portion of blood that contains proteins like albumins, globulins, fibrinogen, and immunoglobulins. The main classes of globulins are alpha, beta, and gamma globulins. Immunoglobulins, also called antibodies, are gamma globulins that recognize and bind to antigens with high specificity. The five major classes of immunoglobulins in humans are IgG, IgM, IgA, IgD, and IgE, which have different structures and functions. Multiple myeloma is a cancer of plasma cells that results in overproduction of a single immunoglobulin class.