Cmr
•Download as PPTX, PDF•
0 likes•474 views
13C NMR spectroscopy provides structural and functional details of compounds by showing the different magnetic environments of carbon atoms. It measures the energy difference between spin states of carbon-13 nuclei, which appears as peaks in the spectrum in parts per million (ppm). The number of peaks corresponds to the number of different carbon environments in a molecule. Chemical shifts are influenced by the electronegativity of bonded atoms, with more electronegative atoms causing peaks to appear at higher ppm due to increased deshielding of the carbon nucleus.
Report
Share
Report
Share
Recommended
Chem 516 presentation

Transition metal derivatives of polyhedral boranes and carboranes can form in different ways. Metallocarboranes often form "sandwich" structures where the metal is bonded between two closo-carborane ligands. These structures are more stable than metallocenes due to properties of the carborane ligands. Metal derivatives of polyhedral boranes can form direct bonds to boron atoms or ionic bonds to the cluster. One example is Cu2B10H10, which has a unique diagonal bonding structure unlike the typical "sandwich". These compounds have various applications including catalysis, organic synthesis, and medicine.
Structure types of crystals

Contains information about various crystal types in solid state chemistry like Rock Salt, Wurtzite, Nickel Arsenide, Zinc Blende etc. It also gives a brief description of lattice energy and Born Haber cycle.
Dinitrogen complexes

Dinitrogen complexes are coordination compounds containing the N2 molecule as a ligand. There are several possible modes of bonding between dinitrogen and metals, including terminal ("end-on") bonding, lateral ("side-on") bonding, and bridge bonding via one or both nitrogen atoms. Only terminal, terminal bridge, and structures with two metal atoms on each nitrogen atom have been conclusively demonstrated. The first dinitrogen complex was discovered in 1965 and contained a stable [(NH3)5RuN2] structure.
Fragmentation of important functional group- Alkane and corbonyl compounds

This slide contains fragmentation of important functional groups i.e. Alkane and Carbonyl compounds in mass spectrometry.
Asymmetric Synthesis - Christeena Shaji

This document discusses asymmetric synthesis, which produces unequal amounts of stereoisomers from achiral precursors. It can be enantioselective or diastereoselective. There are two types: partial asymmetric synthesis, which forms a new chiral center from an achiral precursor using a chiral substrate, auxiliary, reagent, or catalyst; and absolute asymmetric synthesis, which uses no chiral precursors but instead relies on physical chirality like circularly polarized light. Common approaches include using a chiral pool substrate, chiral auxiliary, chiral reagent, or chiral catalyst. The mechanisms and examples of various methods are explained in detail.
Asymmetric synthesis ii

This document discusses various aspects of asymmetric synthesis, including stereochemical aspects, acyclic and cyclic stereoselection, and enantioselective synthesis. It defines terms like racemate, enantiopure, and enantiomer. It describes stereospecific and stereoselective reactions, and rules like Cram's rule and Prelog's rule that help explain stereoselection. It discusses strategies for stereoselective synthesis including additions to carbonyls and aldol reactions. It also covers topics like diastereoselective oxidations, catalytic hydrogenation, and enantioselective reductions using chiral reagents like (S)-PBMgCl and (R,R)-DIOP.
Effect of conformation on reactivity

This document discusses the effect of conformation on the reactivity of acyclic and cyclic compounds. It provides examples of how the conformation of a compound, whether the substituents are equatorial or axial, can impact the rate and outcome of various chemical reactions including E2 eliminations, SN1 and SN2 substitutions, esterification, hydrolysis, elimination reactions, molecular rearrangements, neighboring group participation, and oxidation reactions. Specifically, it notes that equatorial substituents are often more reactive than axial substituents due to differences in steric hindrance and stereoelectronic effects. Reaction rates and products can differ significantly between conformational isomers of the same compound.
Zero field splitting

A ppt compiled by Yaseen Aziz Wani pursuing M.Sc Chemistry at University of Kashmir, J&K, India and Naveed Bashir Dar, a student of electrical engg. at NIT Srinagar.
Warm regards to Munnazir Bashir also for providing us with refreshing tea while we were compiling ppt.
Recommended
Chem 516 presentation

Transition metal derivatives of polyhedral boranes and carboranes can form in different ways. Metallocarboranes often form "sandwich" structures where the metal is bonded between two closo-carborane ligands. These structures are more stable than metallocenes due to properties of the carborane ligands. Metal derivatives of polyhedral boranes can form direct bonds to boron atoms or ionic bonds to the cluster. One example is Cu2B10H10, which has a unique diagonal bonding structure unlike the typical "sandwich". These compounds have various applications including catalysis, organic synthesis, and medicine.
Structure types of crystals

Contains information about various crystal types in solid state chemistry like Rock Salt, Wurtzite, Nickel Arsenide, Zinc Blende etc. It also gives a brief description of lattice energy and Born Haber cycle.
Dinitrogen complexes

Dinitrogen complexes are coordination compounds containing the N2 molecule as a ligand. There are several possible modes of bonding between dinitrogen and metals, including terminal ("end-on") bonding, lateral ("side-on") bonding, and bridge bonding via one or both nitrogen atoms. Only terminal, terminal bridge, and structures with two metal atoms on each nitrogen atom have been conclusively demonstrated. The first dinitrogen complex was discovered in 1965 and contained a stable [(NH3)5RuN2] structure.
Fragmentation of important functional group- Alkane and corbonyl compounds

This slide contains fragmentation of important functional groups i.e. Alkane and Carbonyl compounds in mass spectrometry.
Asymmetric Synthesis - Christeena Shaji

This document discusses asymmetric synthesis, which produces unequal amounts of stereoisomers from achiral precursors. It can be enantioselective or diastereoselective. There are two types: partial asymmetric synthesis, which forms a new chiral center from an achiral precursor using a chiral substrate, auxiliary, reagent, or catalyst; and absolute asymmetric synthesis, which uses no chiral precursors but instead relies on physical chirality like circularly polarized light. Common approaches include using a chiral pool substrate, chiral auxiliary, chiral reagent, or chiral catalyst. The mechanisms and examples of various methods are explained in detail.
Asymmetric synthesis ii

This document discusses various aspects of asymmetric synthesis, including stereochemical aspects, acyclic and cyclic stereoselection, and enantioselective synthesis. It defines terms like racemate, enantiopure, and enantiomer. It describes stereospecific and stereoselective reactions, and rules like Cram's rule and Prelog's rule that help explain stereoselection. It discusses strategies for stereoselective synthesis including additions to carbonyls and aldol reactions. It also covers topics like diastereoselective oxidations, catalytic hydrogenation, and enantioselective reductions using chiral reagents like (S)-PBMgCl and (R,R)-DIOP.
Effect of conformation on reactivity

This document discusses the effect of conformation on the reactivity of acyclic and cyclic compounds. It provides examples of how the conformation of a compound, whether the substituents are equatorial or axial, can impact the rate and outcome of various chemical reactions including E2 eliminations, SN1 and SN2 substitutions, esterification, hydrolysis, elimination reactions, molecular rearrangements, neighboring group participation, and oxidation reactions. Specifically, it notes that equatorial substituents are often more reactive than axial substituents due to differences in steric hindrance and stereoelectronic effects. Reaction rates and products can differ significantly between conformational isomers of the same compound.
Zero field splitting

A ppt compiled by Yaseen Aziz Wani pursuing M.Sc Chemistry at University of Kashmir, J&K, India and Naveed Bashir Dar, a student of electrical engg. at NIT Srinagar.
Warm regards to Munnazir Bashir also for providing us with refreshing tea while we were compiling ppt.
Alpha axial haloketone rule and octant rule

This presentation discusses the use of alpha axial halo ketone rule and Octant rule for determination of absolute configuration
Metal nitrosyls and their derivatives

Metal nitrosyl compounds contain nitric oxide bonded as an NO+ ion, NO- ion, or neutral NO molecule. They can be classified into three classes based on the nitric oxide group present. Metal nitrosyls are coordination compounds where an NO molecule is attached as an NO+ ion to a metal atom or ion. Examples include metal nitrosyl carbonyls such as Co(NO+)(CO)3, metal nitrosyl halides such as Fe(NO+)2I, and metal nitrosyl thio-complexes involving Fe, Co, and Ni metals. These compounds can be prepared through the reaction of nitric oxide with metal compounds like carbonyls, halides, or ferrocyanides. Metal
Synthesis of Longifolene through retrosynthestic analysis. 

These slides will introduce you readers about synthesis of Longifolene through its retrosynthestic analysis. It consist of synthetic and retrosynthetic module of Longifolene given by different scientists
Hydrogenation, catalytic hydrogenation

Hydrogenation- definition, catalytic hydrogenation, homogeneous and heterogeneous catalytic hydrogenation, mechanism of catalytic hydrogenation, advantages and disadvantages of catalytic hydrogenation, applications of catalytic hydrogenation
AUGER & ESCA Spectroscopy( Mass Spectroscopy )

Electron Microscopy :
(1) Auger electron Microscope
- Principle
-Diagram
- Theory
-Application
(2) ESCA (Electron spectroscopy for chemical analysis)/XPS
- Principle
- Diagram
-Theory
-Application
Conformational analysis of ethane butane aliphatics

The document discusses the conformational analysis of several aliphatic compounds including ethane, butane, and 1,2-dichloroethane. It defines important terms related to conformation such as staggered, eclipsed, gauche, and anti. It then analyzes the potential energy diagrams and relative stabilities of different conformations for each compound. The most stable conformations are those without torsional or steric strain, while eclipsed conformations involving atom or group overlaps are highest in energy.
Metal Cluster Higher Boranes

This document discusses metal cluster higher boranes. It begins with an introduction to boranes and their synthesis. It then describes the different types of bonds found in higher boranes, including terminal, direct, bridging, and triply bridging bonds. Specific examples of higher borane structures are examined, including diborane B2H6, tetraborane B4H10, and pentaborane B5H9. Finally, the document classifies higher boranes into closo, nido, and arachno boranes based on their skeletal structures and electron counts, according to Wade's rules. Methods for synthesizing higher boranes are also briefly mentioned.
Disconnection approach towards longifolene, juvabione and morphine. 

Disconnection approach towards longifolene, juvabione and morphine. Minal Saini , student of Chaudhary bansilal University , Bhiwani, Haryana
Longifolene is common naturally occurring, oily liquid hydrocarbon found in the high boiling fraction of certain pine resins.
Juvabione is a terpene- derived-keto-ester that has been isolated from plant sources.
Morphine is a major component of opium,it is isolated from poppy straw of the opium poppy.
Aromaticity in benzenoid and non-benzenoid compunds

This document summarizes aromaticity in benzoid and non-benzoid compounds. It defines aromaticity as the property of conjugated cycloalkenes that enhances stability through delocalization of pi electrons. The rules of aromaticity are outlined, including that aromatic compounds must be cyclic, have planar p-orbitals, and follow Hückel's rule of 4n+2 pi electrons. Benzoid aromatics include benzene and polycyclic structures like naphthalene. Non-benzoid aromatics do not contain benzene rings and examples provided are azulene, tropone, and heterocyclic compounds.
Asymmetric synthesis i

This document provides an overview of asymmetric synthesis and stereochemistry. It begins with definitions of stereoisomers including enantiomers and diastereomers. It then discusses how asymmetric synthesis creates new chiral centers, giving unequal amounts of stereoisomers. Key terms like enantiomeric excess and methods for determining ee are explained. The document outlines different types of asymmetric synthesis controlled by the substrate, auxiliary, reagent or catalyst. It also defines stereoselective and stereospecific reactions. The principles of asymmetric induction and double diastereoselection/asymmetric induction are covered. Recommended books on the topic are provided.
SYMMETRY ELEMENTS AND SYMMETRY OPERATIONS

This document discusses symmetry elements and symmetry operations in group theory and chemistry. It defines the key symmetry elements - identity (E), proper rotation axes (Cn), mirror planes (σ), inversion centers (i), and improper rotation axes (Sn). It provides examples of how these elements apply to common molecular structures like water, methane, benzene, and transition metal complexes. The document also explains the different types of mirror planes and how the symmetry operations are generated from each element.
Asymmetric Synthesis

This document provides an overview of asymmetric synthesis and strategies for achieving asymmetric induction. It defines asymmetric synthesis as a reaction that yields predominantly one chiral stereoisomer. It discusses different strategies for asymmetric induction, including using a chiral auxiliary, chiral reagent/catalyst, or starting with a chiral pool substrate. Specific examples are provided of chiral reagents like BINOL-H and Alpine borane that can be used to selectively reduce prochiral ketones. Chiral ligands like DIOP and CHIRAPHOS that are used with metal catalysts for asymmetric hydrogenation are also described.
Protecting groups and deprotection- -OH, -COOH, C=O, -NH2 groups.

This document discusses various protecting groups used in organic synthesis. It begins by defining a protecting group as a molecular framework that is introduced onto a functional group to block its reactivity under reaction conditions needed for modifications elsewhere in the molecule. The document then summarizes several common protecting groups for hydroxyl, amine, and carboxylic acid functional groups including methyl, benzyl, and silyl ethers for alcohols as well as Boc, Fmoc, Cbz, and other carbamates for amines. It provides details on the formation and cleavage of each protecting group.
Phosphine as ligand by Dr Geeta Tewari

This presentation describes about the nature of phosphine ligands, bonding and reactions of metal phosphine containing complexes. Also explains the similarity and differences of phosphine ligand with NH3 and CO ligands.
M.Sc. 2D NMR.pptx

2D NMR techniques provide additional information beyond conventional 1D NMR. COSY identifies pairs of coupled protons, while HETCOR identifies the number of protons directly bonded to a particular carbon. NOESY and ROESY spectra locate protons that are close in space. DEPT distinguishes between carbon types such as CH3, CH2, CH, and quaternary carbons. Spin decoupling simplifies spectra by removing coupling between irradiated and non-irradiated protons.
1,3 dipolar cycloadditions

The document discusses recent advances in 1,3-dipolar cycloadditions. It begins by introducing the [3+2] 1,3-dipolar cycloaddition reaction which combines a dipolarophile and 1,3-dipole to form a five-membered heterocycle. This reaction is widely used in natural product and pharmaceutical synthesis. It then describes different types of dipolarophiles and 1,3-dipoles that can be used. The reaction mechanisms, classification based on frontier molecular orbitals, effect of Lewis acids, and methods for asymmetric variants are also covered. Recent literature examples of this reaction in natural product synthesis and regioselectivity/stereoselectivity are summarized.
Neighbouring Group Participation.pptx

(i) Non-classical carbocations display delocalization of sigma bonds through 3-center-2-electron bonds in bridged systems. Neighboring group participation can assist reactions by donating electrons through lone pairs, pi bonds, aromatic rings, or sigma bonds.
(ii) The pinacol-pinacolone rearrangement involves the migration of an alkyl group from one carbon to another after the loss of a leaving group from a vicinal diol. The migration is assisted by delocalization of the carbocation intermediate onto the oxygen atom.
(iii) In asymmetrical glycols, the group with greater ability for carbocation delocalization, such as phenyl, will migrate preferentially over
Chemical shift with c13 nmr

A. 13C NMR spectroscopy provides information about carbon structures in organic compounds. It measures the small differences in magnetic field strength needed for carbon nuclei to resonate. These differences are reported in parts per million (ppm) relative to tetramethylsilane (TMS) as a standard. Factors like electronegativity, hybridization, and hydrogen bonding affect the chemical shift values. 13C NMR has applications in metabolic studies and industrial analyses of solids.
Dynamic Stereochemistry-Role of Conformation and Reactivity

Dynamic Stereochemistry-Role of Conformation and ReactivityAteos Foundation of Science Education and Research, Pune, M.S., India
Dynamic Stereochemistry and What role does conformation plays on stereochemistry is being exemplified in this presentation. Useful for the Undergraduate and Postgraduates students of Pharmacy, Pharmaceutical Chemistry and Chemical SciencesOrganosilicon compounds

This presentation has important synthesis and reactions of organosilanes like protection reaction, ipso substitution, Peterson olefination.
C13 nmr

1) 13C NMR spectroscopy provides valuable structural information when 1H NMR is insufficient or ambiguous. It directly detects carbon atoms and gives signals based on their chemical environment rather than hydrogen bonding.
2) 13C NMR spectra contain information about the number and types of carbon atoms present based on the number of signals and their chemical shifts. The chemical shifts are influenced by factors like hybridization and electronegativity.
3) Techniques like proton decoupling and DEPT allow differentiation of carbon types like CH, CH2, and CH3 based on their signal behavior under different pulse sequences.
Carbon13&2D NMR(30 Min Explanation)

This document provides an overview of 13C and 2D NMR spectroscopy. It discusses key topics such as:
1) The basics of 13C NMR including that 13C has a natural abundance of 1.1% and each nonequivalent 13C gives a different signal. Hydrogen-decoupled 13C NMR is most common.
2) Principles of 2D NMR spectroscopy including how experiments have evolution, mixing, and detection periods to produce correlation maps between nuclear spins.
3) Specific 2D experiments like COSY which identifies proton-proton couplings and HECTOR which shows carbon-proton correlations are described. Applications of 13C and 2D NMR for structure elucidation and other
More Related Content
What's hot
Alpha axial haloketone rule and octant rule

This presentation discusses the use of alpha axial halo ketone rule and Octant rule for determination of absolute configuration
Metal nitrosyls and their derivatives

Metal nitrosyl compounds contain nitric oxide bonded as an NO+ ion, NO- ion, or neutral NO molecule. They can be classified into three classes based on the nitric oxide group present. Metal nitrosyls are coordination compounds where an NO molecule is attached as an NO+ ion to a metal atom or ion. Examples include metal nitrosyl carbonyls such as Co(NO+)(CO)3, metal nitrosyl halides such as Fe(NO+)2I, and metal nitrosyl thio-complexes involving Fe, Co, and Ni metals. These compounds can be prepared through the reaction of nitric oxide with metal compounds like carbonyls, halides, or ferrocyanides. Metal
Synthesis of Longifolene through retrosynthestic analysis. 

These slides will introduce you readers about synthesis of Longifolene through its retrosynthestic analysis. It consist of synthetic and retrosynthetic module of Longifolene given by different scientists
Hydrogenation, catalytic hydrogenation

Hydrogenation- definition, catalytic hydrogenation, homogeneous and heterogeneous catalytic hydrogenation, mechanism of catalytic hydrogenation, advantages and disadvantages of catalytic hydrogenation, applications of catalytic hydrogenation
AUGER & ESCA Spectroscopy( Mass Spectroscopy )

Electron Microscopy :
(1) Auger electron Microscope
- Principle
-Diagram
- Theory
-Application
(2) ESCA (Electron spectroscopy for chemical analysis)/XPS
- Principle
- Diagram
-Theory
-Application
Conformational analysis of ethane butane aliphatics

The document discusses the conformational analysis of several aliphatic compounds including ethane, butane, and 1,2-dichloroethane. It defines important terms related to conformation such as staggered, eclipsed, gauche, and anti. It then analyzes the potential energy diagrams and relative stabilities of different conformations for each compound. The most stable conformations are those without torsional or steric strain, while eclipsed conformations involving atom or group overlaps are highest in energy.
Metal Cluster Higher Boranes

This document discusses metal cluster higher boranes. It begins with an introduction to boranes and their synthesis. It then describes the different types of bonds found in higher boranes, including terminal, direct, bridging, and triply bridging bonds. Specific examples of higher borane structures are examined, including diborane B2H6, tetraborane B4H10, and pentaborane B5H9. Finally, the document classifies higher boranes into closo, nido, and arachno boranes based on their skeletal structures and electron counts, according to Wade's rules. Methods for synthesizing higher boranes are also briefly mentioned.
Disconnection approach towards longifolene, juvabione and morphine. 

Disconnection approach towards longifolene, juvabione and morphine. Minal Saini , student of Chaudhary bansilal University , Bhiwani, Haryana
Longifolene is common naturally occurring, oily liquid hydrocarbon found in the high boiling fraction of certain pine resins.
Juvabione is a terpene- derived-keto-ester that has been isolated from plant sources.
Morphine is a major component of opium,it is isolated from poppy straw of the opium poppy.
Aromaticity in benzenoid and non-benzenoid compunds

This document summarizes aromaticity in benzoid and non-benzoid compounds. It defines aromaticity as the property of conjugated cycloalkenes that enhances stability through delocalization of pi electrons. The rules of aromaticity are outlined, including that aromatic compounds must be cyclic, have planar p-orbitals, and follow Hückel's rule of 4n+2 pi electrons. Benzoid aromatics include benzene and polycyclic structures like naphthalene. Non-benzoid aromatics do not contain benzene rings and examples provided are azulene, tropone, and heterocyclic compounds.
Asymmetric synthesis i

This document provides an overview of asymmetric synthesis and stereochemistry. It begins with definitions of stereoisomers including enantiomers and diastereomers. It then discusses how asymmetric synthesis creates new chiral centers, giving unequal amounts of stereoisomers. Key terms like enantiomeric excess and methods for determining ee are explained. The document outlines different types of asymmetric synthesis controlled by the substrate, auxiliary, reagent or catalyst. It also defines stereoselective and stereospecific reactions. The principles of asymmetric induction and double diastereoselection/asymmetric induction are covered. Recommended books on the topic are provided.
SYMMETRY ELEMENTS AND SYMMETRY OPERATIONS

This document discusses symmetry elements and symmetry operations in group theory and chemistry. It defines the key symmetry elements - identity (E), proper rotation axes (Cn), mirror planes (σ), inversion centers (i), and improper rotation axes (Sn). It provides examples of how these elements apply to common molecular structures like water, methane, benzene, and transition metal complexes. The document also explains the different types of mirror planes and how the symmetry operations are generated from each element.
Asymmetric Synthesis

This document provides an overview of asymmetric synthesis and strategies for achieving asymmetric induction. It defines asymmetric synthesis as a reaction that yields predominantly one chiral stereoisomer. It discusses different strategies for asymmetric induction, including using a chiral auxiliary, chiral reagent/catalyst, or starting with a chiral pool substrate. Specific examples are provided of chiral reagents like BINOL-H and Alpine borane that can be used to selectively reduce prochiral ketones. Chiral ligands like DIOP and CHIRAPHOS that are used with metal catalysts for asymmetric hydrogenation are also described.
Protecting groups and deprotection- -OH, -COOH, C=O, -NH2 groups.

This document discusses various protecting groups used in organic synthesis. It begins by defining a protecting group as a molecular framework that is introduced onto a functional group to block its reactivity under reaction conditions needed for modifications elsewhere in the molecule. The document then summarizes several common protecting groups for hydroxyl, amine, and carboxylic acid functional groups including methyl, benzyl, and silyl ethers for alcohols as well as Boc, Fmoc, Cbz, and other carbamates for amines. It provides details on the formation and cleavage of each protecting group.
Phosphine as ligand by Dr Geeta Tewari

This presentation describes about the nature of phosphine ligands, bonding and reactions of metal phosphine containing complexes. Also explains the similarity and differences of phosphine ligand with NH3 and CO ligands.
M.Sc. 2D NMR.pptx

2D NMR techniques provide additional information beyond conventional 1D NMR. COSY identifies pairs of coupled protons, while HETCOR identifies the number of protons directly bonded to a particular carbon. NOESY and ROESY spectra locate protons that are close in space. DEPT distinguishes between carbon types such as CH3, CH2, CH, and quaternary carbons. Spin decoupling simplifies spectra by removing coupling between irradiated and non-irradiated protons.
1,3 dipolar cycloadditions

The document discusses recent advances in 1,3-dipolar cycloadditions. It begins by introducing the [3+2] 1,3-dipolar cycloaddition reaction which combines a dipolarophile and 1,3-dipole to form a five-membered heterocycle. This reaction is widely used in natural product and pharmaceutical synthesis. It then describes different types of dipolarophiles and 1,3-dipoles that can be used. The reaction mechanisms, classification based on frontier molecular orbitals, effect of Lewis acids, and methods for asymmetric variants are also covered. Recent literature examples of this reaction in natural product synthesis and regioselectivity/stereoselectivity are summarized.
Neighbouring Group Participation.pptx

(i) Non-classical carbocations display delocalization of sigma bonds through 3-center-2-electron bonds in bridged systems. Neighboring group participation can assist reactions by donating electrons through lone pairs, pi bonds, aromatic rings, or sigma bonds.
(ii) The pinacol-pinacolone rearrangement involves the migration of an alkyl group from one carbon to another after the loss of a leaving group from a vicinal diol. The migration is assisted by delocalization of the carbocation intermediate onto the oxygen atom.
(iii) In asymmetrical glycols, the group with greater ability for carbocation delocalization, such as phenyl, will migrate preferentially over
Chemical shift with c13 nmr

A. 13C NMR spectroscopy provides information about carbon structures in organic compounds. It measures the small differences in magnetic field strength needed for carbon nuclei to resonate. These differences are reported in parts per million (ppm) relative to tetramethylsilane (TMS) as a standard. Factors like electronegativity, hybridization, and hydrogen bonding affect the chemical shift values. 13C NMR has applications in metabolic studies and industrial analyses of solids.
Dynamic Stereochemistry-Role of Conformation and Reactivity

Dynamic Stereochemistry-Role of Conformation and ReactivityAteos Foundation of Science Education and Research, Pune, M.S., India
Dynamic Stereochemistry and What role does conformation plays on stereochemistry is being exemplified in this presentation. Useful for the Undergraduate and Postgraduates students of Pharmacy, Pharmaceutical Chemistry and Chemical SciencesOrganosilicon compounds

This presentation has important synthesis and reactions of organosilanes like protection reaction, ipso substitution, Peterson olefination.
What's hot (20)
Synthesis of Longifolene through retrosynthestic analysis. 

Synthesis of Longifolene through retrosynthestic analysis.
Conformational analysis of ethane butane aliphatics

Conformational analysis of ethane butane aliphatics
Disconnection approach towards longifolene, juvabione and morphine. 

Disconnection approach towards longifolene, juvabione and morphine.
Aromaticity in benzenoid and non-benzenoid compunds

Aromaticity in benzenoid and non-benzenoid compunds
Protecting groups and deprotection- -OH, -COOH, C=O, -NH2 groups.

Protecting groups and deprotection- -OH, -COOH, C=O, -NH2 groups.
Dynamic Stereochemistry-Role of Conformation and Reactivity

Dynamic Stereochemistry-Role of Conformation and Reactivity
Similar to Cmr
C13 nmr

1) 13C NMR spectroscopy provides valuable structural information when 1H NMR is insufficient or ambiguous. It directly detects carbon atoms and gives signals based on their chemical environment rather than hydrogen bonding.
2) 13C NMR spectra contain information about the number and types of carbon atoms present based on the number of signals and their chemical shifts. The chemical shifts are influenced by factors like hybridization and electronegativity.
3) Techniques like proton decoupling and DEPT allow differentiation of carbon types like CH, CH2, and CH3 based on their signal behavior under different pulse sequences.
Carbon13&2D NMR(30 Min Explanation)

This document provides an overview of 13C and 2D NMR spectroscopy. It discusses key topics such as:
1) The basics of 13C NMR including that 13C has a natural abundance of 1.1% and each nonequivalent 13C gives a different signal. Hydrogen-decoupled 13C NMR is most common.
2) Principles of 2D NMR spectroscopy including how experiments have evolution, mixing, and detection periods to produce correlation maps between nuclear spins.
3) Specific 2D experiments like COSY which identifies proton-proton couplings and HECTOR which shows carbon-proton correlations are described. Applications of 13C and 2D NMR for structure elucidation and other
c13 nmr.pptx

This document provides an overview of C13 NMR spectroscopy. It discusses the principles and theory of NMR spectroscopy, the history of C13 NMR, and the information that can be obtained from C13 NMR spectra. Specifically, it explains that C13 NMR spectroscopy allows identification of carbon atoms in organic molecules similarly to how proton NMR identifies hydrogen atoms. It also discusses factors that influence chemical shifts in C13 NMR such as substitution effects, hybridization, and electronegativity. In summary, the document serves as an introduction to C13 NMR spectroscopy, its applications and principles.
13 C NMR Spectroscopy by Dr Anthony Melvin Crasto

13C-NMR spectroscopy provides information about organic compounds. It can determine the number of non-equivalent carbon atoms and identify carbon types like methyl, methylene, aromatic, and carbonyl groups. 13C signals are spread over a wider range than 1H NMR, making individual carbons easier to identify. Challenges include the low natural abundance of 13C and its lower gyromagnetic ratio. Techniques like signal averaging, Fourier transforms, and decoupling are used to overcome these issues and provide detailed 13C NMR spectra.
13 C NMR Spectroscopy with examples by Dr Anthony Crasto

13C-NMR spectroscopy provides information about carbon atoms in organic compounds. It works by applying a strong magnetic field to excite carbon-13 nuclei, which make up about 1% of naturally occurring carbon. The document discusses several key aspects of 13C-NMR including: principles of NMR spectroscopy; chemical shifts and peak assignments; coupling patterns; techniques to overcome low carbon abundance like signal averaging and Fourier transform; and decoupling methods to simplify spectra. Examples are provided to illustrate predicting chemical shifts and interpreting 13C-NMR spectra.
C-13 NMR Spectroscopy ppt(10 Minute explanation)

Nuclear magnetic resonance spectroscopy techniques such as 13C NMR and 2D NMR experiments like COSY and HECTOR can be used to analyze organic compounds. [13C NMR provides information about the number and types of carbon atoms in a molecule based on their chemical shifts. Two-dimensional NMR experiments reveal coupling between nuclei like 1H-13C and 1H-1H couplings to help determine molecular structure.] DEPT NMR experiments distinguish between methylene, methine and methyl carbons. 13C NMR finds applications in fields like metabolic analysis, drug purity determination and polymer characterization.
C NMR

13C NMR spectra provide information about the different types of carbon environments in a molecule. Each carbon atom appears as a single peak since 13C nuclei are not split due to the low natural abundance of 13C. The chemical shifts of 13C peaks are affected by the electronegativity of nearby atoms and range from 0-220 ppm. Hybridization also influences chemical shifts, with sp3 carbons more shielded than sp2 carbons. The number of signals in a 13C NMR spectrum equals the number of nonequivalent carbon types in the molecule.
Carbon 13

Nuclear magnetic resonance (NMR) spectroscopy can detect certain atomic nuclei that have spin, including the carbon-13 isotope. While carbon-12 does not produce a signal in NMR due to having no spin, carbon-13 accounts for about 1.1% of naturally occurring carbon and can be detected. Carbon-13 has a very weak signal that is difficult to detect due to its low natural abundance and sensitivity being 1/5700 of hydrogen-1. However, the development of Fourier-transform NMR and signal averaging techniques have allowed the detection and analysis of carbon-13 NMR spectra.
c13ppt-150515121301-lva1-app6892 (1).pdf

This document provides an overview of nuclear magnetic resonance spectroscopy using carbon-13 (13C NMR). It discusses key aspects of 13C NMR including the properties of 13C nuclei, chemical shifts, hydrogen decoupling, DEPT experiments, 2D NMR techniques like COSY and HECTOR, and applications of 13C NMR such as structure elucidation and in vivo analysis. Examples are provided to illustrate concepts like hydrogen decoupling, DEPT spectra, and 2D NMR correlations. In summary, the document serves as an introduction to 13C NMR spectroscopy, covering fundamental principles, experimental techniques, and applications of the method.
BT631-18-NMR_3

The document discusses 13C-NMR spectroscopy. It notes that while many of the theories of 1H-NMR also apply to 13C-NMR, there are several important differences. Specifically, 13C nuclei have a much weaker magnetic moment than protons, requiring more sample and signal averaging. Additionally, the range of chemical shifts is much wider for 13C than 1H, allowing each carbon to be distinguished. Modern techniques like DEPT and multidimensional NMR help overcome the challenges of analyzing 13C spectra.
C-13 NMR Spectroscopy

This document provides an overview of C-13 NMR spectroscopy. It discusses the history and principle of NMR spectroscopy, focusing on C-13. Key points include: C-13 has a nuclear spin of 1/2, allowing it to be detected by NMR, unlike C-12. The chemical shift range for C-13 is much broader than for proton NMR, from 0-220 ppm. The number of C-13 signals indicates the number of non-equivalent carbon types in a molecule. C-13 coupling is observed with directly bonded protons and other nearby nuclei. Applications of C-13 NMR include structure elucidation of organic and biochemical compounds.
Introductory discussion to 13 C NMR

Introductory discussion to 13 C NMR, THE 13C NUCLEUS, Proton-coupled and Proton decoupled 13C spectra.
Sana 13 nmr

13C NMR gives distinct signals for each non-equivalent carbon atom based on its chemical environment. It has a wider chemical shift range than 1H NMR, allowing for easier separation of signals. However, 13C NMR spectra are complicated by weak signals due to the low natural abundance of 13C. Modern Fourier transform NMR techniques have helped overcome this issue. Proton-decoupled 13C NMR provides simple spectra with one peak per carbon, while proton-coupled spectra show splitting patterns indicating directly bonded protons. 13C NMR finds numerous applications in
13C-NMR SPECTROSCOPY

This document provides an overview of nuclear magnetic resonance spectroscopy (NMR) focusing on Carbon-13 (13C) NMR. It defines NMR and explains the principles of how atomic nuclei absorb energy from radiofrequency fields in a magnetic field. The summary discusses key aspects of 13C NMR including that 13C is difficult to detect due to its low natural abundance, advantages over 1H NMR, factors affecting chemical shifts, techniques to simplify spectra like decoupling, and applications like DEPT NMR to determine functional groups.
Nmr spectroscopy by dr. umesh kumar sharma and susan jacob

This document provides an overview of NMR spectroscopy. It discusses various NMR techniques like spin-spin decoupling and Fourier transform NMR. It explains the principles of 1H NMR, 13C NMR, and applications of NMR like structure determination and analysis of mixtures. NMR spectroscopy is a powerful analytical technique for studying molecular structure.
CMR SPPU for M Sc I

13C NMR spectroscopy provides information about carbon atoms in a molecule. It gives the number of chemically nonequivalent carbons and information about the environment of each carbon such as hybridization and attached atoms. 13C NMR spectra require longer acquisition times than 1H NMR due to the low natural abundance of 13C. Signals are spread over a wider range and 13C-13C coupling is usually not observed. Chemical shifts indicate carbon hybridization and are influenced by attached electronegative atoms. Integration is not useful in 13C NMR as signal intensity does not correlate to carbon number.
C 13 NMR

C13 NMR spectroscopy provides information about carbon atoms in molecules. It works based on the absorption of radio waves by carbon-13 nuclei in a magnetic field. There are a few key points:
1) C13 NMR is difficult to analyze due to the low natural abundance of C13 and its weaker magnetic resonance compared to protons.
2) Different types of carbon atoms (CH, CH2, CH3) can be distinguished based on their chemical shifts and coupling patterns. Proton decoupling is used to simplify spectra.
3) DEPT experiments analyze carbon types by enhancing signals from different hybridized carbons (CH, CH2, CH3) in different ways. This allows determining the number and type
13CNMRfor322.pdf

13C-NMR can be used to examine the structure of molecules. Only about 1% of carbon atoms are 13C, which can be observed by NMR. Each different type of carbon will appear at a different frequency in the 13C-NMR spectrum. The number of peaks will correspond to the number of unique carbon environments in the molecule. The intensity of peaks does not necessarily correlate to the number of carbons. Chemical shifts are reported in parts per million (ppm) relative to TMS.
CARBON 13 NMR

ITS AGAIN AN IMPORTANT TOPIC OF ANALYTICAL CHEMISTRY WHERE C13 IS AN TYPE OF NUCLEAR MAGNETIC RESONANCE ALONG WITH PROTON NMR. STUDY THIS TOPIC WELL FOR BTTER UNDERTSANDING OF NMR WHICH IS BELIEVED TO BE ONE OF THE TOUGH PART.
HOPE YOU ALL WILL USE IT WELL.
C-13 NMR.pptx

This document discusses C-13 NMR spectroscopy. It begins with an introduction to NMR spectroscopy and an overview of C-13 NMR. It then covers the history, principle, and basics of C-13 NMR including why it is required, chemical shifts, number of signals, spin-spin splitting, and factors that affect the spectroscopy. The document concludes by outlining some applications of C-13 NMR and providing references.
Similar to Cmr (20)
13 C NMR Spectroscopy with examples by Dr Anthony Crasto

13 C NMR Spectroscopy with examples by Dr Anthony Crasto
Nmr spectroscopy by dr. umesh kumar sharma and susan jacob

Nmr spectroscopy by dr. umesh kumar sharma and susan jacob
More from wadhava gurumeet
Chemoinformatic File Format.pptx

This document discusses cheminformatics and its applications. Cheminformatics combines chemistry and computer science to store and analyze chemical data for applications like drug discovery. It encompasses designing, organizing, analyzing and visualizing chemical information. Key topics covered include molecular representations, chemical databases, similarity searching, machine learning methods, and tools for molecular docking and drug discovery.
Geographical Indicators

This document discusses geographical indications (GIs), including their definition, benefits, examples, registration process, challenges, and relationship to trademarks. Some key points:
- GIs identify goods that originate from a specific geographical region and possess qualities due to that origin. Examples include Basmati rice, Darjeeling tea, and Champagne.
- Registering a GI confers legal protection and promotes the economic prosperity of producers. It can boost exports and support rural development.
- The registration process involves filing an application representing producers, publishing the application for opposition, and registering approved GIs for 10-year periods.
- Challenges include low brand value, lack of awareness, and misuse of
Gas Chromatography PPT GCW.pptx

1. Gas chromatography is a technique used to separate components of a mixture using their volatility. It involves two phases - a stationary phase and a mobile gas phase.
2. The basic components of a gas chromatograph are an injection port, column, detector, and recorder. The sample is injected and carried by the mobile gas phase through the column where separation occurs.
3. Separation is based on the difference in partitioning behavior of analytes between the stationary and mobile phases. Components with higher partition coefficients have longer retention times.
Errors PPt.pptx

Errors in analysis can be either determinate (systematic) or indeterminate (random). Determinate errors are caused by faults in the analytical procedure or instruments and result in consistently inaccurate results. Common sources of determinate error include faulty instrumentation, contaminated reagents, incorrect analytical methods, and analyst errors. Determinate errors can be identified by comparing results to a known standard or independent analytical method, and the source of the error must then be determined and corrected to improve accuracy.
Advance Green Chemistry.ppt

This document provides information about green chemistry. It discusses natural processes versus chemical processes and how green chemistry aims to make chemical processes more environmentally friendly. Some key points made include:
- Green chemistry seeks to prevent pollution by designing chemical synthesis and products to be benign.
- Natural processes are more environmentally friendly than traditional chemical processes which use toxic solvents and generate hazardous wastes.
- Green chemistry principles include using safer solvents like water or ionic liquids, performing solvent-free reactions, and using renewable feedstocks and benign catalysts.
- New techniques like microwave irradiation and ultrasound can help drive chemical reactions in a more energy efficient and atom economic manner.
analytical dose.pptx

1. The document introduces different types of dosage forms including solid, liquid, and semi-solid forms. Solid forms include tablets, capsules, powders, and granules. Liquid forms include solutions, emulsions, suspensions, syrups and elixirs. Semi-solid forms include ointments, gels, creams and pastes.
2. Dosage forms deliver drug molecules to sites of action in the body and provide benefits like accurate dosing, protecting drugs, and masking tastes. They are classified based on route of administration, physical form, and whether they are for oral, topical, inhaled or other uses.
3. Common excipients used in dosage forms are discussed
Antibiotic Drug.ppt

This document discusses various classes of antibiotics including penicillins, cephalosporins, macrolides, tetracyclines, aminoglycosides, and quinolones. It describes their mechanisms of action, common uses, and potential adverse effects. Specifically, it provides details on common drugs in each class, how they work at the cellular level to kill bacteria, infections they can treat, and side effects to monitor like ototoxicity and nephrotoxicity. The document stresses the importance of obtaining cultures before treatment and monitoring patients for both therapeutic responses and unwanted reactions.
Assay In Drug Analysis.ppt

This document discusses different types of assays used in drug analysis, including chemical, immunological, microbiological, and bioassays. It provides details on various chemical assay techniques such as photometry, colorimetry, spectrophotometry, fluorimetry, flame photometry, and different types of chromatography. It also explains the principles, types, and techniques of immunoassays like ELISA, radioimmunoassay, and fluoroimmunoassay. Microbiological assays and characteristics of good assay methods are briefly covered as well.
bioassay.pptx

Dr. Gurumeet C Wadhawa discusses biological assays. An assay is a procedure used to qualitatively or quantitatively assess the presence, amount, or functional activity of a target entity. There are three main types of assays: chemical, immuno, and bioassays. Bioassays involve estimating the concentration or potency of a pharmaceutical drug using animal or human subjects. While less precise than chemical assays, bioassays are more sensitive and can be used when chemical methods are not available or applicable. Bioassays are used to standardize and quantify various biological substances and products.
Biological Catalyst.pptx

Dr. Gurumeet C Wadhawa discusses biocatalysts such as enzymes and microbes. Enzymes are mostly proteins that catalyze biochemical reactions in living cells and have unique three-dimensional shapes that fit reactants. They are produced commercially by isolating microbial strains that naturally produce the desired enzyme and optimizing fermentation conditions. Biocatalysts are classified into six types based on the reactions they catalyze. Important enzymes in the human body include digestive enzymes and DNA polymerases. Biocatalysts have various industrial applications in fields such as pharmaceuticals, food processing, and cosmetics.
Drug Discovery.pptx

1. The document discusses drug discovery and development, outlining the need to address unmet medical needs like new diseases as well as the costs of existing therapies.
2. It describes the historical aspects of clinical trials and regulations dating back to the 1500s, and outlines the modern drug development process including discovery, preclinical studies, and clinical trials through the various phases.
3. The drug development pathway involves discovery, preclinical development including chemistry/pharmacology and toxicology studies on animals, and clinical development including Phase I-III trials on volunteers and patients, with the goal of regulatory approval and market introduction over approximately 10-15 years.
fda guidles.pptx

The FDA regulates food, drugs, medical devices and other products. It oversees the drug approval process which involves preclinical testing in animals, followed by Phase I-III clinical trials in humans to test safety, efficacy and side effects. If approved, the drug can be marketed and is monitored for side effects. The document outlines the drug approval process and regulations around generic drugs, biologics, manufacturing and product changes.
Green Chemistry.ppt

This document discusses environmentally friendly synthetic strategies, specifically non-conventional methods like microwave irradiation and ultrasonication. It notes that these methods have advantages over conventional methods in being cleaner with higher yields and being more eco-friendly. The document outlines a strategy to synthesize new heterocyclic compounds by combining moieties like pyrazole, benzo-γ-pyrone, and quinoline, and studying the biological activity of the resulting products. It provides details on sonochemistry and microwave-assisted organic reactions, giving examples of reactions that can be performed using these techniques. The aims are to synthesize and characterize new heterocycles and evaluate their therapeutic potential, attempting the syntheses using both conventional and non-conventional
Introduction to Pharmaceuticak Industry.pptx

The document provides an overview of the pharmaceutical sector and its key business units and functions. It discusses:
1) The pharmaceutical sector can be classified into two main groups - drug discovery and manufacturing. Manufacturing is further divided into active pharmaceutical ingredients, generics, and biologics production.
2) Drug discovery is the most important process and involves significant time and costs to develop new drugs. The manufacturing areas have departments like production, quality control, quality assurance, process development, and engineering services.
3) The roles and educational qualifications required vary across the different business units and functions, but generally include degrees in fields like chemistry, pharmacy, biotechnology, engineering, and business administration. Senior roles often require a PhD
Mevolonic Acid.pptx

The document discusses the mevalonate and methylerythritol phosphate pathways which are used by nature to synthesize terpenoids. Terpenoids are derived from isoprene units which can be joined in head-to-tail or head-to-head fashion, resulting in hemiterpenes, monoterpenes, sesquiterpenes, diterpenes, sesterterpenes, triterpenes, and tetraterpenes. The mevalonate pathway is important for synthesizing steroids while the methylerythritol phosphate pathway may be more commonly used in most organisms. A variety of natural terpenoids derived from these pathways are then discussed, including their structures
solid support.ppt

The document provides an overview of solid phase synthesis. It describes how solid phase synthesis involves coupling reagents to an inert solid support to perform multi-step organic synthesis. The key steps include attaching the starting material to a resin via a linker, performing sequential reactions on the bound intermediate, then cleaving the final product from the resin. The Merrifield method from 1963 pioneered this technique by automating the synthesis of peptides on an insoluble polystyrene resin, enabling efficient purification and the potential for parallel reactions.
Sonochemistry.pptx

This document provides an overview of ultrasound assisted organic synthesis (sonochemistry). It begins by defining ultrasound and discussing how it is used to promote and accelerate various organic chemical reactions. Key advantages of sonochemistry include increased reaction rates and product yields. It then discusses several examples of specific reaction types (e.g. condensation, substitution, addition) that have been improved through ultrasonic irradiation. The document also covers experimental parameters that can be optimized in sonochemical reactions as well as various applications in fields like pharmaceuticals, materials science, and environmental chemistry. In closing, it briefly introduces the concept of supercritical fluids.
Spectrophotometry.pptx

Spectrophotometry involves using a spectrophotometer to measure how much light is absorbed by a sample at different wavelengths. It relies on Beer's Law, which states that absorbance is directly proportional to concentration, path length, and absorptivity. A spectrophotometer directs light from a source through a sample and measures the intensity of the transmitted light, allowing the absorbance and concentration of the sample to be determined. Spectrophotometry is used in various applications including chemistry, medicine, and environmental monitoring.
stability study.pptx

The document discusses stability studies of drug formulations. It defines stability as the ability of a drug product to remain within established specifications over time under storage and usage conditions. Stability testing is conducted to determine shelf life, recommended storage conditions, and suitability of packaging. The main types of drug degradation discussed are physical degradation (changes in appearance, solubility) and chemical degradation (hydrolysis, oxidation). Specific examples of each type of degradation are provided.
SULFONAMIDES.ppt

Sulfonamides are one of the oldest classes of antibacterial drugs used to treat infections. They work by inhibiting the bacterial synthesis of folic acid, making them bacteriostatic. While their use has decreased with newer antibiotics, they remain an alternative for those allergic to penicillin and are used to treat urinary tract infections, ear infections, and other bacterial infections. Common side effects include rashes, nausea, and hematologic issues like anemia. Nursing care involves monitoring for side effects, ensuring adequate hydration when taking the drug, and patient education.
More from wadhava gurumeet (20)
Recently uploaded
Sharlene Leurig - Enabling Onsite Water Use with Net Zero Water

Sharlene Leurig - Enabling Onsite Water Use with Net Zero WaterTexas Alliance of Groundwater Districts
Presented at June 6-7 Texas Alliance of Groundwater Districts Business MeetingSAR of Medicinal Chemistry 1st by dk.pdf

In this presentation include the prototype drug SAR on thus or with their examples .
Syllabus of Second Year B. Pharmacy
2019 PATTERN.
waterlessdyeingtechnolgyusing carbon dioxide chemicalspdf

The technology uses reclaimed CO₂ as the dyeing medium in a closed loop process. When pressurized, CO₂ becomes supercritical (SC-CO₂). In this state CO₂ has a very high solvent power, allowing the dye to dissolve easily.
ESR spectroscopy in liquid food and beverages.pptx

With increasing population, people need to rely on packaged food stuffs. Packaging of food materials requires the preservation of food. There are various methods for the treatment of food to preserve them and irradiation treatment of food is one of them. It is the most common and the most harmless method for the food preservation as it does not alter the necessary micronutrients of food materials. Although irradiated food doesn’t cause any harm to the human health but still the quality assessment of food is required to provide consumers with necessary information about the food. ESR spectroscopy is the most sophisticated way to investigate the quality of the food and the free radicals induced during the processing of the food. ESR spin trapping technique is useful for the detection of highly unstable radicals in the food. The antioxidant capability of liquid food and beverages in mainly performed by spin trapping technique.
(June 12, 2024) Webinar: Development of PET theranostics targeting the molecu...

(June 12, 2024) Webinar: Development of PET theranostics targeting the molecu...Scintica Instrumentation
Targeting Hsp90 and its pathogen Orthologs with Tethered Inhibitors as a Diagnostic and Therapeutic Strategy for cancer and infectious diseases with Dr. Timothy Haystead.Katherine Romanak - Geologic CO2 Storage.pdf

Presented at June 6-7 Texas Alliance of Groundwater Districts Business Meeting
在线办理(salfor毕业证书)索尔福德大学毕业证毕业完成信一模一样

学校原件一模一样【微信:741003700 】《(salfor毕业证书)索尔福德大学毕业证》【微信:741003700 】学位证,留信认证(真实可查,永久存档)原件一模一样纸张工艺/offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原。
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
【主营项目】
一.毕业证【q微741003700】成绩单、使馆认证、教育部认证、雅思托福成绩单、学生卡等!
二.真实使馆公证(即留学回国人员证明,不成功不收费)
三.真实教育部学历学位认证(教育部存档!教育部留服网站永久可查)
四.办理各国各大学文凭(一对一专业服务,可全程监控跟踪进度)
如果您处于以下几种情况:
◇在校期间,因各种原因未能顺利毕业……拿不到官方毕业证【q/微741003700】
◇面对父母的压力,希望尽快拿到;
◇不清楚认证流程以及材料该如何准备;
◇回国时间很长,忘记办理;
◇回国马上就要找工作,办给用人单位看;
◇企事业单位必须要求办理的
◇需要报考公务员、购买免税车、落转户口
◇申请留学生创业基金
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
Immersive Learning That Works: Research Grounding and Paths Forward

We will metaverse into the essence of immersive learning, into its three dimensions and conceptual models. This approach encompasses elements from teaching methodologies to social involvement, through organizational concerns and technologies. Challenging the perception of learning as knowledge transfer, we introduce a 'Uses, Practices & Strategies' model operationalized by the 'Immersive Learning Brain' and ‘Immersion Cube’ frameworks. This approach offers a comprehensive guide through the intricacies of immersive educational experiences and spotlighting research frontiers, along the immersion dimensions of system, narrative, and agency. Our discourse extends to stakeholders beyond the academic sphere, addressing the interests of technologists, instructional designers, and policymakers. We span various contexts, from formal education to organizational transformation to the new horizon of an AI-pervasive society. This keynote aims to unite the iLRN community in a collaborative journey towards a future where immersive learning research and practice coalesce, paving the way for innovative educational research and practice landscapes.
Equivariant neural networks and representation theory

Or: Beyond linear.
Abstract: Equivariant neural networks are neural networks that incorporate symmetries. The nonlinear activation functions in these networks result in interesting nonlinear equivariant maps between simple representations, and motivate the key player of this talk: piecewise linear representation theory.
Disclaimer: No one is perfect, so please mind that there might be mistakes and typos.
dtubbenhauer@gmail.com
Corrected slides: dtubbenhauer.com/talks.html
ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...

ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...Advanced-Concepts-Team
Presentation in the Science Coffee of the Advanced Concepts Team of the European Space Agency on the 07.06.2024.
Speaker: Diego Blas (IFAE/ICREA)
Title: Gravitational wave detection with orbital motion of Moon and artificial
Abstract:
In this talk I will describe some recent ideas to find gravitational waves from supermassive black holes or of primordial origin by studying their secular effect on the orbital motion of the Moon or satellites that are laser ranged.Direct Seeded Rice - Climate Smart Agriculture

Direct Seeded Rice - Climate Smart AgricultureInternational Food Policy Research Institute- South Asia Office
PPT on Direct Seeded Rice presented at the three-day 'Training and Validation Workshop on Modules of Climate Smart Agriculture (CSA) Technologies in South Asia' workshop on April 22, 2024.
EWOCS-I: The catalog of X-ray sources in Westerlund 1 from the Extended Weste...

Context. With a mass exceeding several 104 M⊙ and a rich and dense population of massive stars, supermassive young star clusters
represent the most massive star-forming environment that is dominated by the feedback from massive stars and gravitational interactions
among stars.
Aims. In this paper we present the Extended Westerlund 1 and 2 Open Clusters Survey (EWOCS) project, which aims to investigate
the influence of the starburst environment on the formation of stars and planets, and on the evolution of both low and high mass stars.
The primary targets of this project are Westerlund 1 and 2, the closest supermassive star clusters to the Sun.
Methods. The project is based primarily on recent observations conducted with the Chandra and JWST observatories. Specifically,
the Chandra survey of Westerlund 1 consists of 36 new ACIS-I observations, nearly co-pointed, for a total exposure time of 1 Msec.
Additionally, we included 8 archival Chandra/ACIS-S observations. This paper presents the resulting catalog of X-ray sources within
and around Westerlund 1. Sources were detected by combining various existing methods, and photon extraction and source validation
were carried out using the ACIS-Extract software.
Results. The EWOCS X-ray catalog comprises 5963 validated sources out of the 9420 initially provided to ACIS-Extract, reaching a
photon flux threshold of approximately 2 × 10−8 photons cm−2
s
−1
. The X-ray sources exhibit a highly concentrated spatial distribution,
with 1075 sources located within the central 1 arcmin. We have successfully detected X-ray emissions from 126 out of the 166 known
massive stars of the cluster, and we have collected over 71 000 photons from the magnetar CXO J164710.20-455217.
THEMATIC APPERCEPTION TEST(TAT) cognitive abilities, creativity, and critic...

THEMATIC APPERCEPTION TEST(TAT) cognitive abilities, creativity, and critic...Abdul Wali Khan University Mardan,kP,Pakistan
hematic appreciation test is a psychological assessment tool used to measure an individual's appreciation and understanding of specific themes or topics. This test helps to evaluate an individual's ability to connect different ideas and concepts within a given theme, as well as their overall comprehension and interpretation skills. The results of the test can provide valuable insights into an individual's cognitive abilities, creativity, and critical thinking skillsApplied Science: Thermodynamics, Laws & Methodology.pdf

When I was asked to give a companion lecture in support of ‘The Philosophy of Science’ (https://shorturl.at/4pUXz) I decided not to walk through the detail of the many methodologies in order of use. Instead, I chose to employ a long standing, and ongoing, scientific development as an exemplar. And so, I chose the ever evolving story of Thermodynamics as a scientific investigation at its best.
Conducted over a period of >200 years, Thermodynamics R&D, and application, benefitted from the highest levels of professionalism, collaboration, and technical thoroughness. New layers of application, methodology, and practice were made possible by the progressive advance of technology. In turn, this has seen measurement and modelling accuracy continually improved at a micro and macro level.
Perhaps most importantly, Thermodynamics rapidly became a primary tool in the advance of applied science/engineering/technology, spanning micro-tech, to aerospace and cosmology. I can think of no better a story to illustrate the breadth of scientific methodologies and applications at their best.
Recently uploaded (20)
Sharlene Leurig - Enabling Onsite Water Use with Net Zero Water

Sharlene Leurig - Enabling Onsite Water Use with Net Zero Water
waterlessdyeingtechnolgyusing carbon dioxide chemicalspdf

waterlessdyeingtechnolgyusing carbon dioxide chemicalspdf
aziz sancar nobel prize winner: from mardin to nobel

aziz sancar nobel prize winner: from mardin to nobel
ESR spectroscopy in liquid food and beverages.pptx

ESR spectroscopy in liquid food and beverages.pptx
(June 12, 2024) Webinar: Development of PET theranostics targeting the molecu...

(June 12, 2024) Webinar: Development of PET theranostics targeting the molecu...
Basics of crystallography, crystal systems, classes and different forms

Basics of crystallography, crystal systems, classes and different forms
Immersive Learning That Works: Research Grounding and Paths Forward

Immersive Learning That Works: Research Grounding and Paths Forward
Equivariant neural networks and representation theory

Equivariant neural networks and representation theory
ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...

ESA/ACT Science Coffee: Diego Blas - Gravitational wave detection with orbita...
EWOCS-I: The catalog of X-ray sources in Westerlund 1 from the Extended Weste...

EWOCS-I: The catalog of X-ray sources in Westerlund 1 from the Extended Weste...
THEMATIC APPERCEPTION TEST(TAT) cognitive abilities, creativity, and critic...

THEMATIC APPERCEPTION TEST(TAT) cognitive abilities, creativity, and critic...
Applied Science: Thermodynamics, Laws & Methodology.pdf

Applied Science: Thermodynamics, Laws & Methodology.pdf
GBSN - Biochemistry (Unit 6) Chemistry of Proteins

GBSN - Biochemistry (Unit 6) Chemistry of Proteins
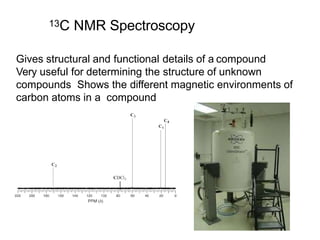
Cmr
- 1. 13C NMR Spectroscopy Gives structural and functional details of a compound Very useful for determining the structure of unknown compounds Shows the different magnetic environments of carbon atoms in a compound
- 2. ABUNDANCE 13C is difficult to record because of 1. The most abundant isotope of carbon that is 12C (99.1%) is not detected by nmr because it has an even no of protons and neutrons 12C is nmr inactive. 2. Magnetic resonance of 13C is much weaker. Moreover, gyromagnectic ratio of 13C being only one fourth that of proton, so the resonance frequency of 13C is one fourth that of proton nmr.
- 3. Advantages of 13C- NMR over 1H- NMR 1.13C- provides information about the backboneof molecules rather than the periphery. 2.The chemical shifts range for 13C- NMR for most organic compounds is 200 ppm compared to 10 –15 ppm for H, hence there is less overlap of peaks for 13C- NMR. 3. Homonuclear spin-spin coupling between carbon atoms is not observed because the natural abundance of 13C is too low for two 13C to be next to one another. Heteronuclear spin coupling between 13C and 12C does not occur because the spin quantum number of 12C is zero. 4. There are a number of excellent methods for decoupling the interaction between 13 C and 1H.
- 5. 13C NMR spectroscopy 13C NMR spectroscopy measures the difference in energy between the aligned spin state and unaligned spin state The difference in energy is measured in ppm (parts per million). The lower the energy gap the easier it is to flip from one state to another
- 6. Chemical Environments 13C NMR spectroscopy shows peaks for each of the different chemical environments of the carbon atom in a molecule. The environment of a carbon atom can be determined by looking at the sequence of bonds the carbon atom has to other atoms. If two carbon atoms have the same bond sequence they will have the same environment. 1 1 2
- 7. Chemical Environments The number of chemical environment a molecule has is the number of peaks in the 13C NMR spectrum. For example: IR has told you that you have an alcohol functional group, but you don’t know if the alcohol is propan-1-ol or propan-2-ol. The 13C NMR will allow you to tell the difference. Number of carbon chemical environments in propan- 1-ol: Number of carbon chemical environments in propan-2-ol: pg 18 and 19
- 9. Do now: How many carbon environments will the following compounds have? 3-methylbutanal 2,4-dichloropentane pentan-2-one NH2 H3C CH CH2 CH3 C C C C C H H CH3 H H H H H H H
- 10. Predicting 13C Spectra CH3 C H 3 C C C C CH3 C plane of symmetry 4 lines O CH3 CH3 O CH3 c CH3 CH3 C C O CH3 5 lines CH3 CH3 CH3 C C H 5 lines
- 11. upfield 20 40 60 220 200 180 160 140 120 100 80 0 CH3 CH2 CH C X (halogen) C N C O C C C N C C C O 13C Chemical shift ( ) TMS Aromatic C downfield 13C chemical shift (δ) 13C NMR spectroscopy The position of the peak in a 13C NMR spectrum is determinedby the shielding or deshielding effects of other atoms around it. The less electrons (ie the more electronegative atoms around the carbon) the higher magnetic field the nucleus is exposed to, hence it is harder to flip and the ppm value is higher.
- 13. 13C NMR spectroscopy Shielding and deshielding Shielding and deshielding effects explain the chemical shifts that we observe in the NMR. The closer the 13C nucleus is to an electronegative atom the more deshielded it will be. Electronegative atoms (like O, Cl, Br etc) or multiple bonds (C=C, C≡C) partially remove electrons from the 13C nucleus. This leaves it exposed to more of a magnetic field and it takes more energy to flip, resulting in a higher chemical shift. It is not possible to predict the exact position of 13C peaks as they are influenced by the environment around them but we can predict the approximate location.
- 14. 13C NMR spectroscopy Using this information could 13C NMR distinguish between the following functional groups? Acid chlorides and aldehydes Ketones and amides
- 15. 13C NMR spectroscopy This information is only a guide. It is extremely difficult to predict exactly the chemical shift of carbon atoms in a compound. Carbon atoms in the same environment will always have the same chemical shift.
- 18. SPIN –SPIN SPLITTING OF 13C SIGNALS Splitting take place acc. to 2nI+1 rule Where n= no. of nuclei I=spin quantum number CH3 = 3+1=4 quartet CH2 = 2+1=3 triplet CH = 1+1=2 doublet C = 0+1=1 singlet CDCl3 gives three peaks because its I=1 so acc. to 2nI+1 2 1 1+1=3 so it gives 1:1:1 peaks Solvents used are CDCl3, DMSO, d6acetone, d6 benzene
- 19. 13C NMR spectroscopy Comparing hex-1-ene to hex-3-ene. How many peaks would you expect to see in the 13C NMR spectra of the above compounds? Why? hex-1- ene hex-3- ene
- 20. 13C NMR spectroscopy How many different carbon environments would ethyl ethanoate have? Where would you expect the peaks to be in the spectrum? pg 28 - 34
- 21. Match the following six compounds to the following six 13C NMR spectra. propanoic acid propan-1-ol propanone propan-2-ol propanal methyl ethanoate
- 22. A
- 24. B
- 25. B propan-2-ol 2 environments C- O CH 3
- 26. C
- 28. D
- 30. E
- 32. F
- 33. F methyl ethanoate 3 environments C= O CH3 C-O
- 34. O C CD Cl3 (solve nt) O CH3CCH 3 CH3
- 36. a) The number of signals correlates with the number types of carbon in a molecule
- 37. cyclopenta ne
- 40. 13C-NMR pentane
- 41. 13C-NMR hexane
- 42. 13C-heptane
- 46. 13C-NMR toluene
- 49. pentan e
- 50. hexan e
- 51. ethyl bromide
- 53. ethanol
- 54. 2-propanol
- 55. O
- 57. Ethyl amine
- 58. Acetaldehyde
- 61. 2-pentanone O
- 62. acetic acid
- 63. Propionic acid
- 65. Acetamide
- 67. 1-pentene
- 68. 2-butyne
- 69. Benzene
- 70. Benzaldehyde
- 71. c) DEPT data DEPT = distortionless enhancement by polarization Distinguishes: CH3 - methyl groups -CH2- methylene groups I -CH- methine groups I -C- I 4o carbons ( not detected by DEPT)
- 72. DEPT 13C spectrum of citronella C10H16O
- 75. HC CCH2CH2CH2CH3
- 78. H2C=C=CHCH2CH2OH
- 79. CO2H
- 81. N H3C H3C CCH3 O
- 87. CF3COCH3 O
- 89. Predicte d Chemica l Shifts of Ca and Cb a b Ca = (-2.5) + 4(9.1) + 9.4 + 2(-2.5) + 3(-1.5) + (-8.4) = 25.4 ppm Cb = (-2.5) + 2(9.1) + 5(9.4) +(-7.2) + (-2.5) = 53.0 ppm base 4o(1o) 4 o(2o) base 2o(4o) 2 o(3o)
- 90. 2,2,4- Trimet hylpent ane 53.28, 31.10, 30.16, 25.51, 24.73
- 91. 0 1.0 2.0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0 Chemical shift (, ppm) ClCH2 Figure 13.20(a) (page 511) CH3 ClCH2CH2CH2CH2CH3 1H
- 92. Chemical shift (, ppm) Figure 13.20(b) (page 511) ClCH2CH2CH2CH2CH3 0 20 40 60 80 100 120 140 160 180 200 13C CDCl3 a separate, distinct peak appears for each of the 5 carbons
- 93. Examples (chemical shifts in ppm from TMS) 23 138 sp3 hybridized carbon is more shielded than sp2
- 94. Examples (chemical shifts in ppm from TMS) OH O sp3 hybridized carbon is more shielded than sp2 61 202
- 95. Examples (chemical shifts in ppm from TMS) OH 23 an electronegative atom deshields the carbon to which it is attached 61
- 96. Table 13.3 (p 513) Type of carbon Chemical shift (), ppm Type of carbon Chemical shift (), ppm RCH3 0-35 CR2 R2C 65-90 CR RC R2CH2 15-40 R3CH 25-50 R4C 30-40 100-150 110-175
- 97. Table 13.3 (p 513) Type of carbon Chemical shift (), ppm RCH2Br 20-40 RCH2Cl 25-50 35-50 RCH2NH2 50-65 RCH2OH RCH2OR 50-65
- 98. Table 13.3 (p 513) Type of carbon Chemical shift (), ppm Type of carbon Chemical shift (), ppm RCH2Br 20-40 RCH2Cl 25-50 35-50 RCH2NH2 50-65 RCH2OH RCH2OR 50-65 RCOR O 160-185 RCR O 190-220
- 99. CH3 OH Figure 13.21 (page 514) Chemical shift (, ppm) 0 20 40 60 80 100 120 140 160 180 200 7 carbons give 7 signals, but intensities are not equal
- 100. Chemical shift (, ppm) 0 20 40 60 80 100 120 140 160 180 200 Figure 13.23 (a) (page 516) O C C CH CH CH CH2 CH2 CH2 CH3 CCH2CH2CH2CH3 O
- 101. Chemical shift (, ppm) 0 20 40 60 80 100 120 140 160 180 200 Figure 13.23 (a) (page 516) CH CH CH CH2 CH2 CH2 CH3 CCH2CH2CH2CH3 O
- 102. Chemical shift (, ppm) 0 20 40 60 80 100 120 140 160 180 200 Figure 13.23 (b) (page 516) CH CH CH CH2 CH2 CH2 CH3 CCH2CH2CH2CH3 O CH and CH3 unaffected C and C=O nulled CH2 inverted
