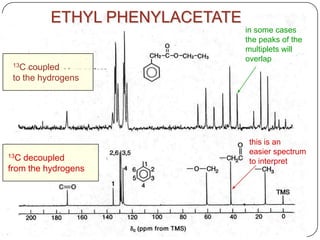
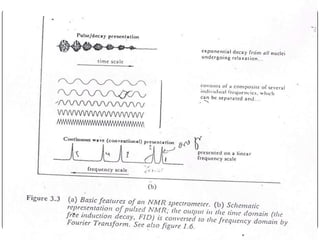
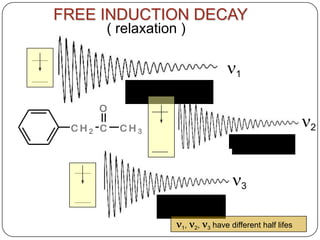
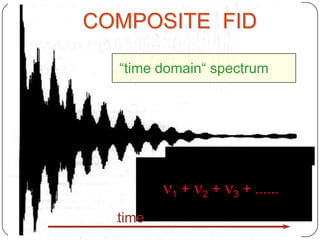
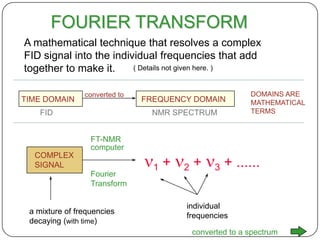
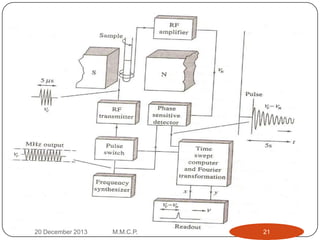
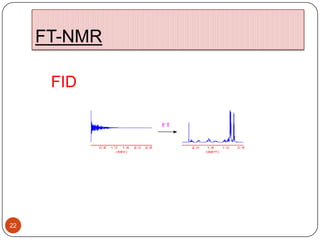
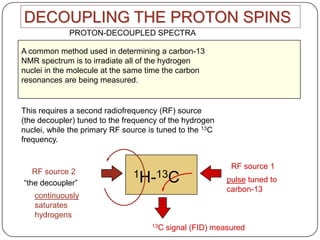
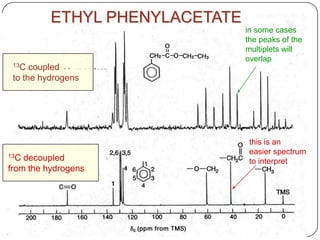
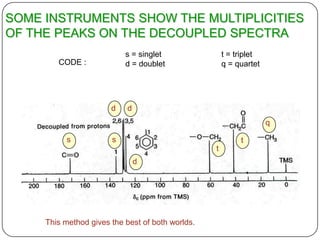
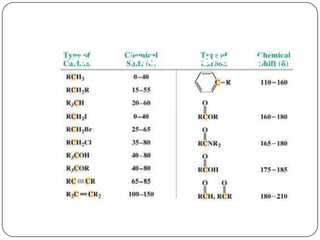
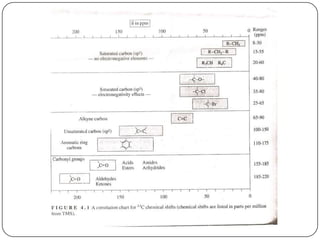
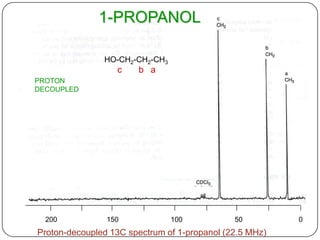
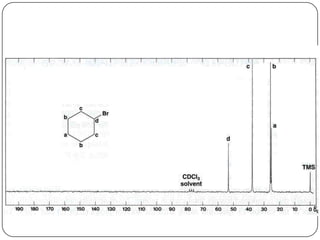
13C NMR gives distinct signals for each non-equivalent carbon atom based on its chemical environment. It has a wider chemical shift range than 1H NMR, allowing for easier separation of signals. However, 13C NMR spectra are complicated by weak signals due to the low natural abundance of 13C. Modern Fourier transform NMR techniques have helped overcome this issue. Proton-decoupled 13C NMR provides simple spectra with one peak per carbon, while proton-coupled spectra show splitting patterns indicating directly bonded protons. 13C NMR finds numerous applications in







![Types of 13c spectra
1) proton coupled 13c spectra
2) proton decoupled 13c spectra
1) PROTON COUPLED 13c SPECTRA
a) Homoannular coupling
the probablity of finding 13c adjacent carbon is very less
therefore
homonuclear [carbon- carbon ]splitting is rearaly seen
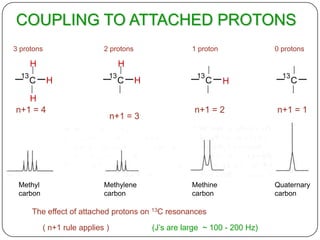
b) Hetronuclear coupling.
it involving two different atoms [carbon- hydrogen]
Here splitting arises due proton attached directly to 13c carbon
8](https://image.slidesharecdn.com/sana13nmr-131220074337-phpapp01/85/Sana-13-nmr-8-320.jpg)