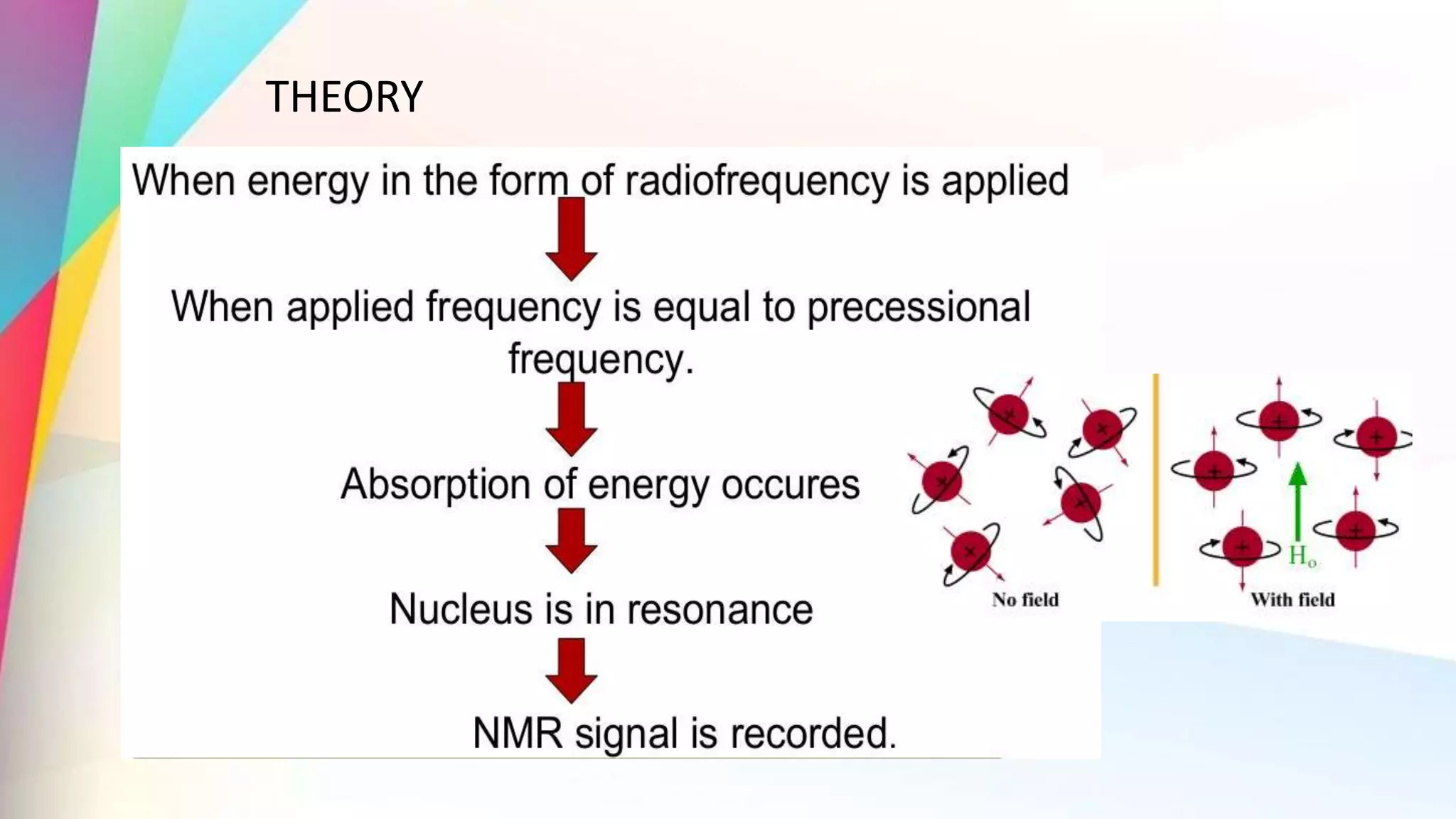
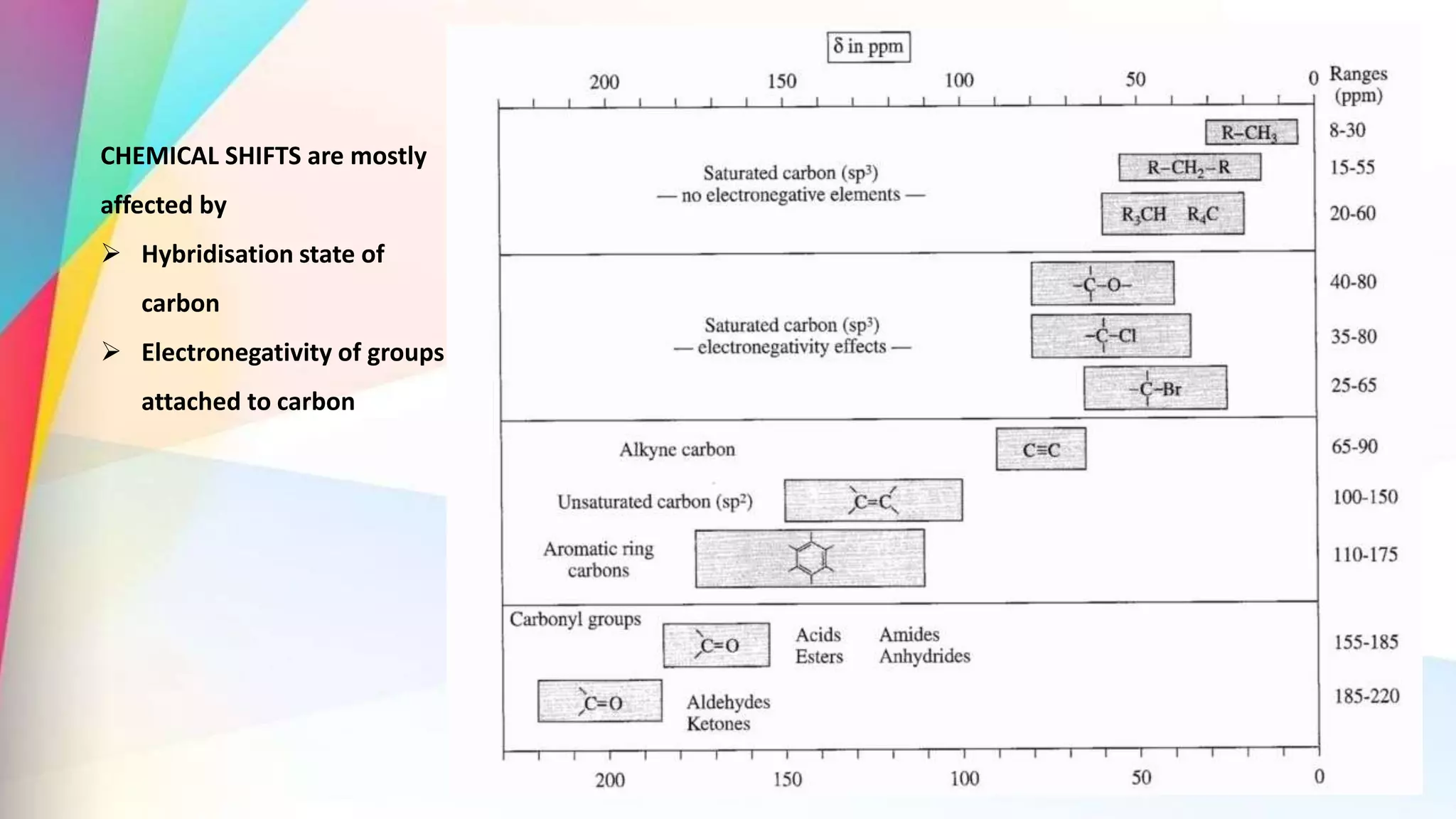
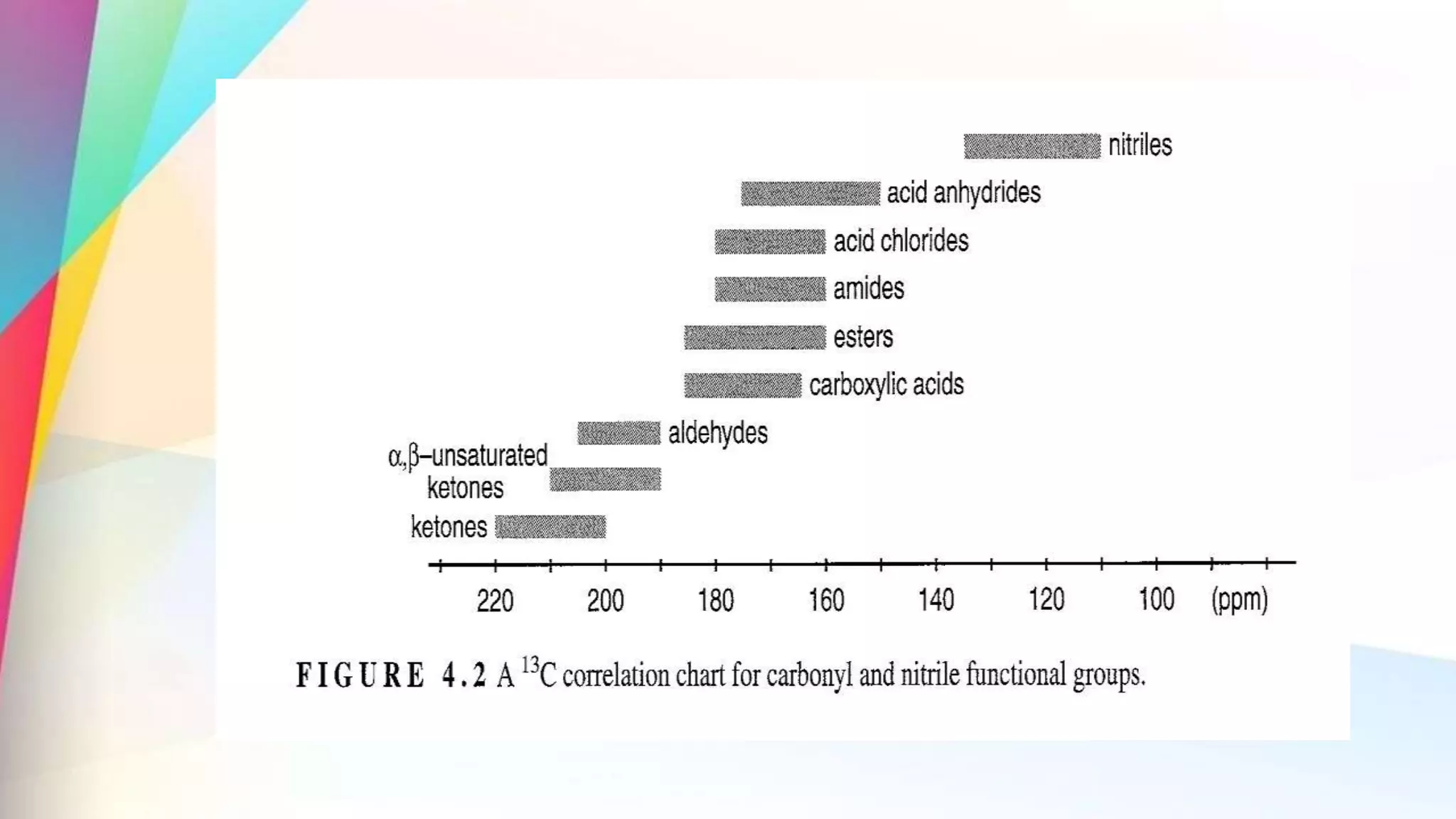
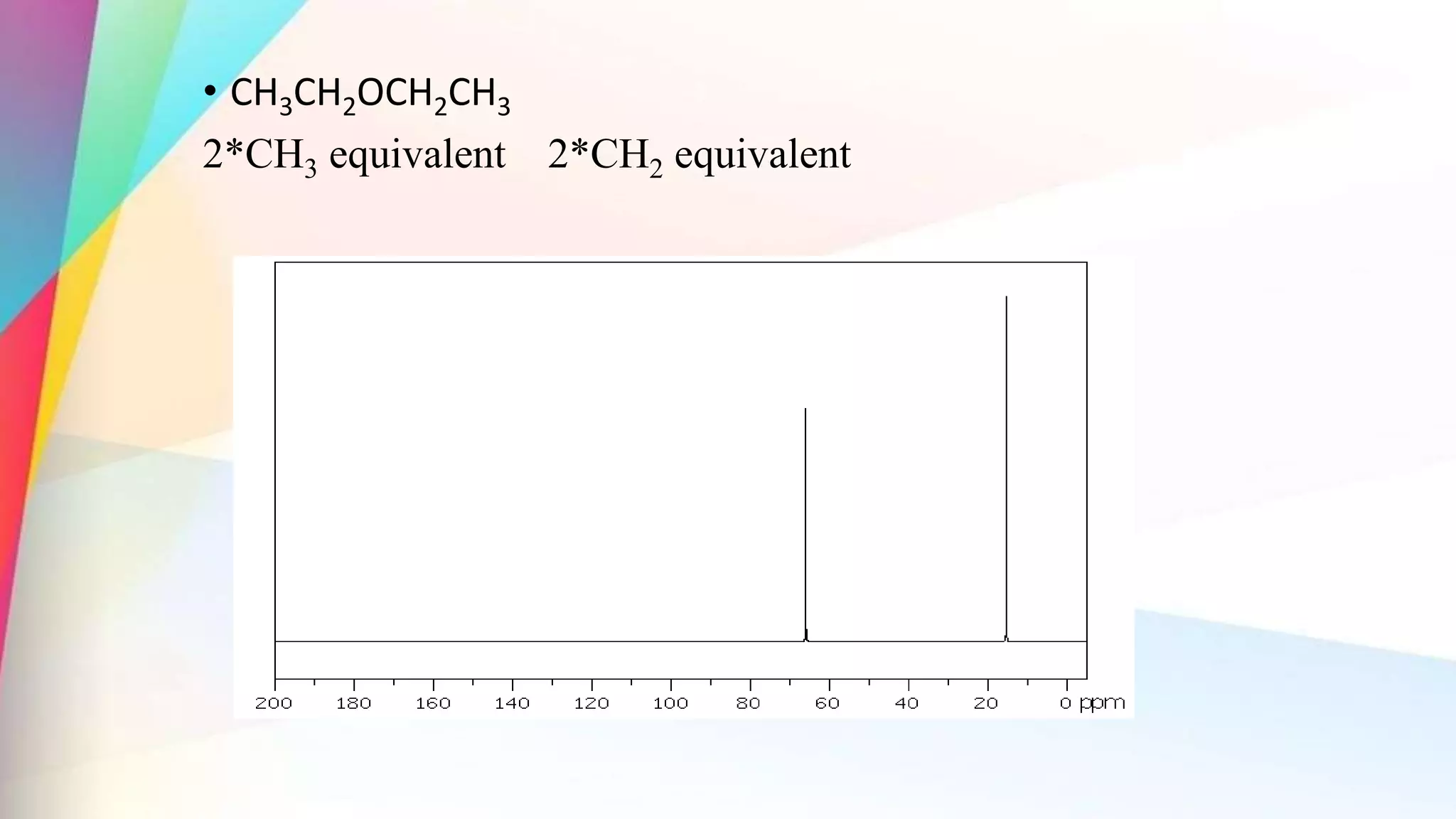
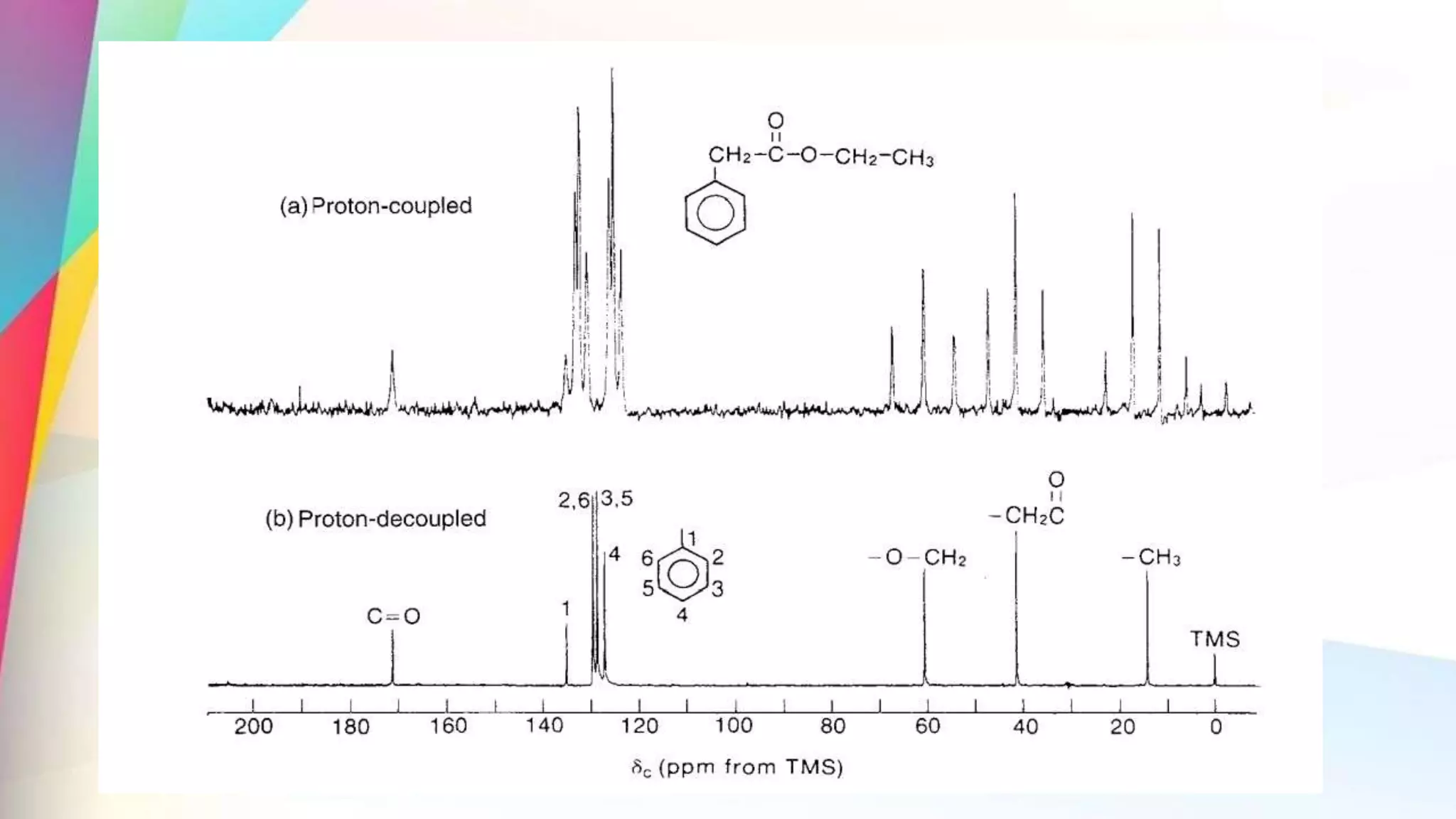
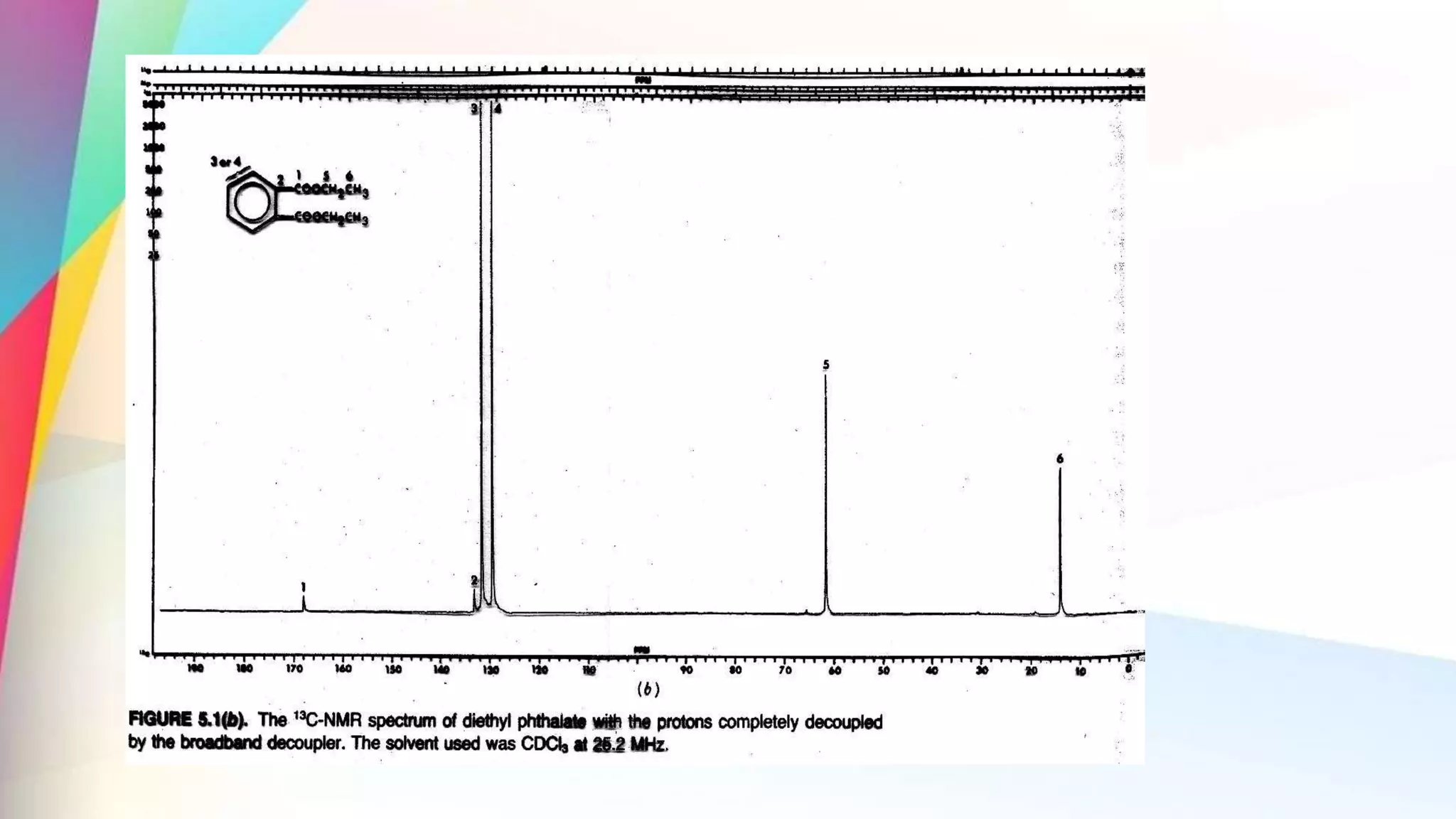
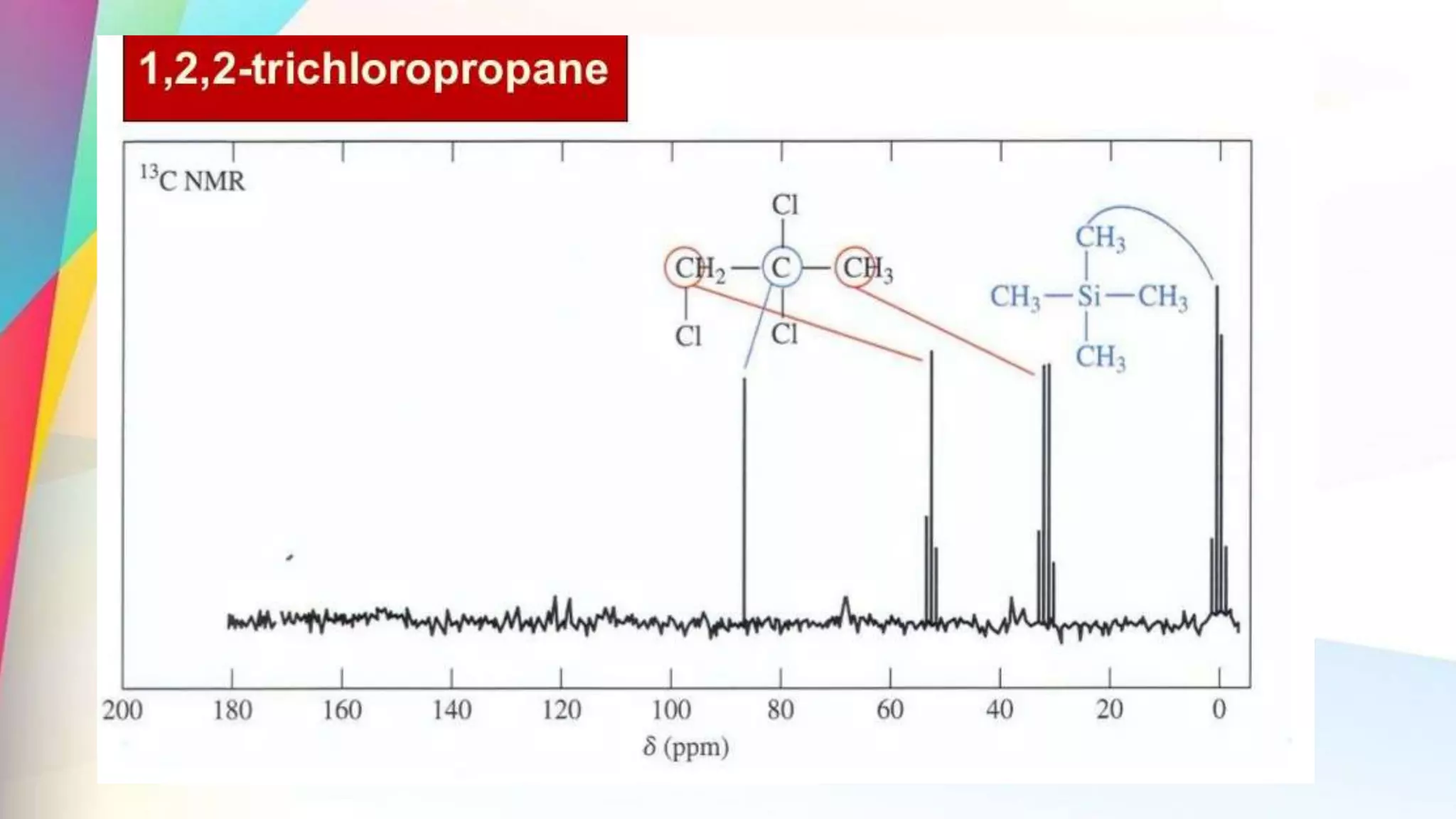
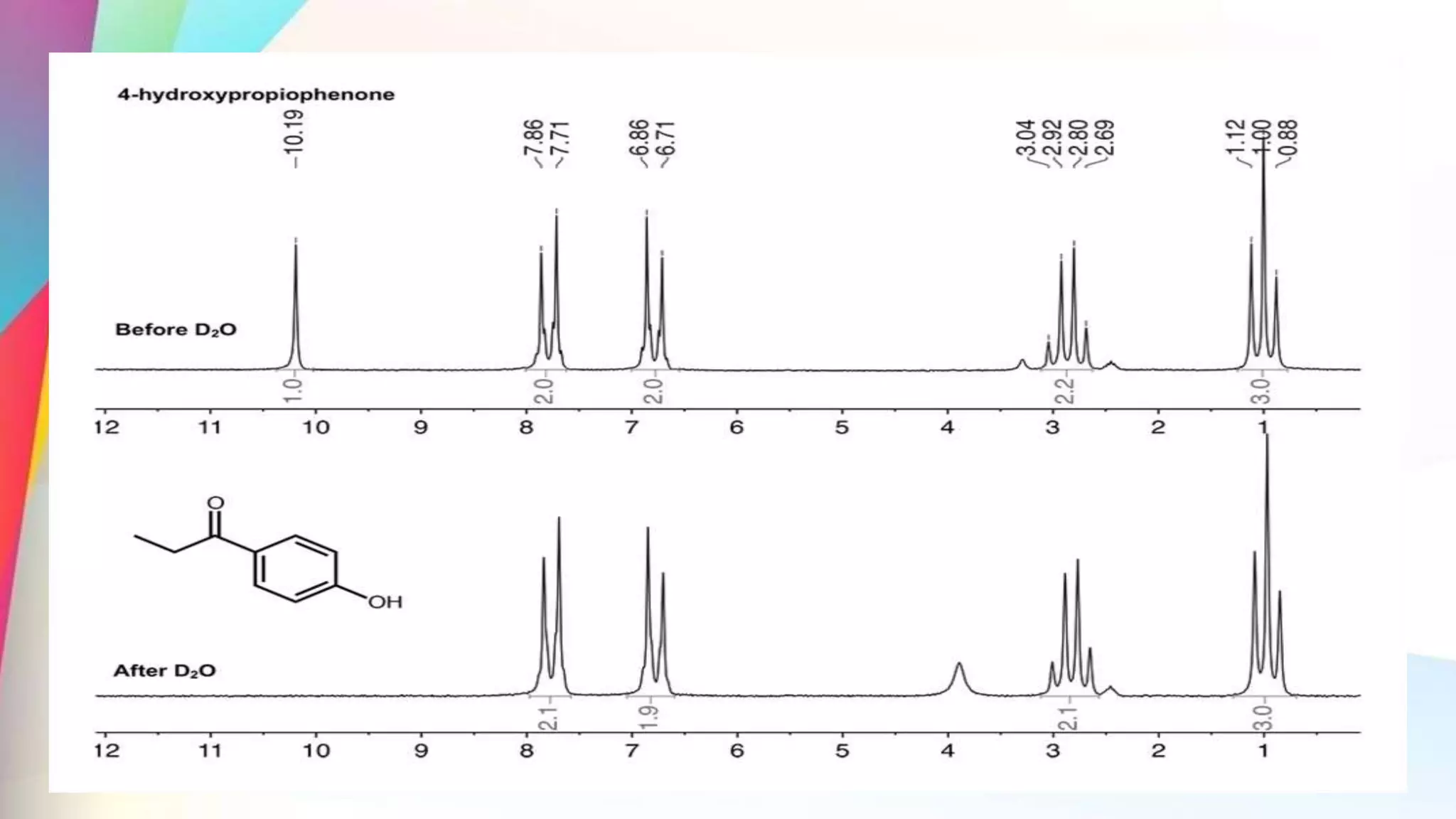
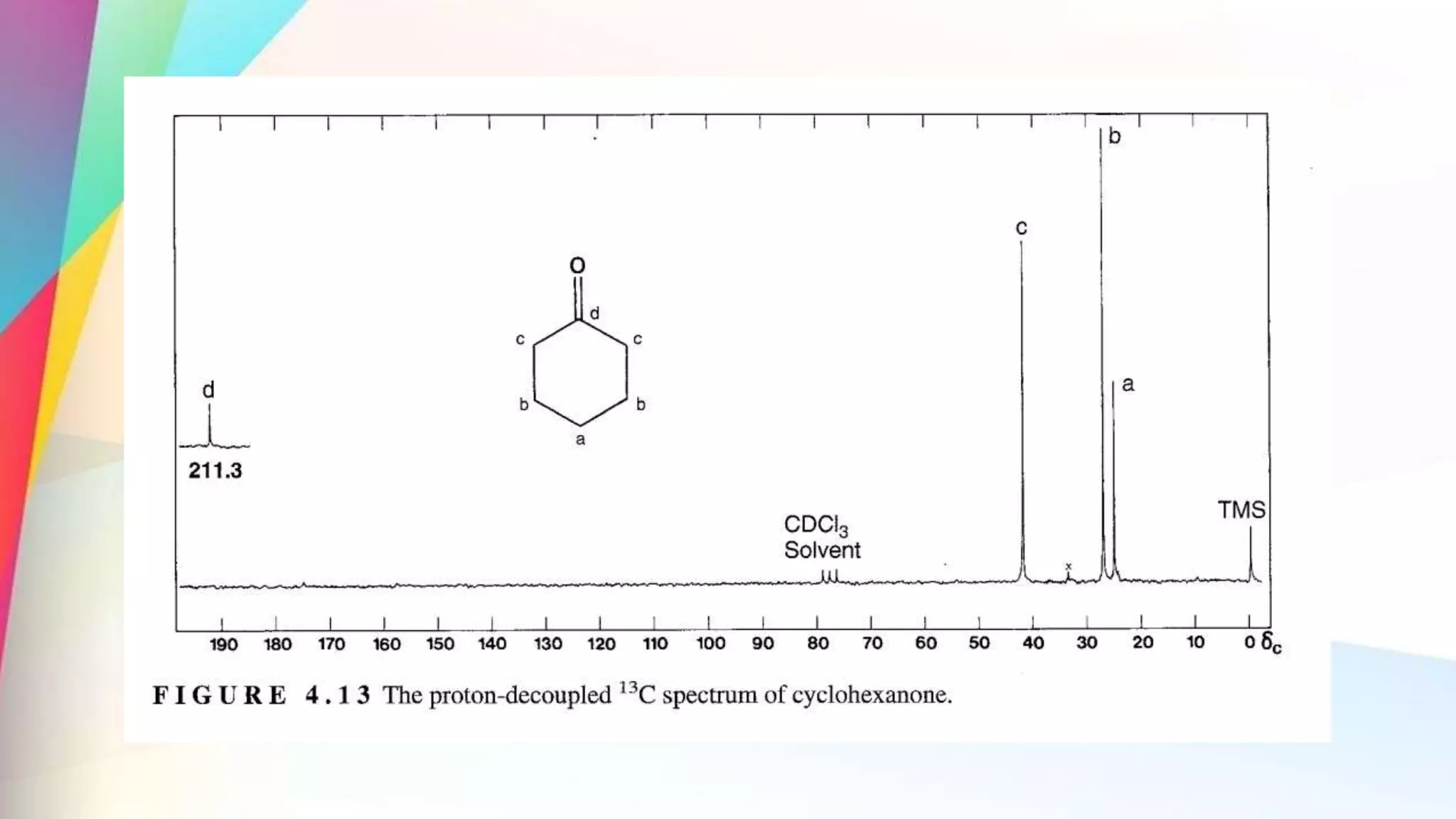
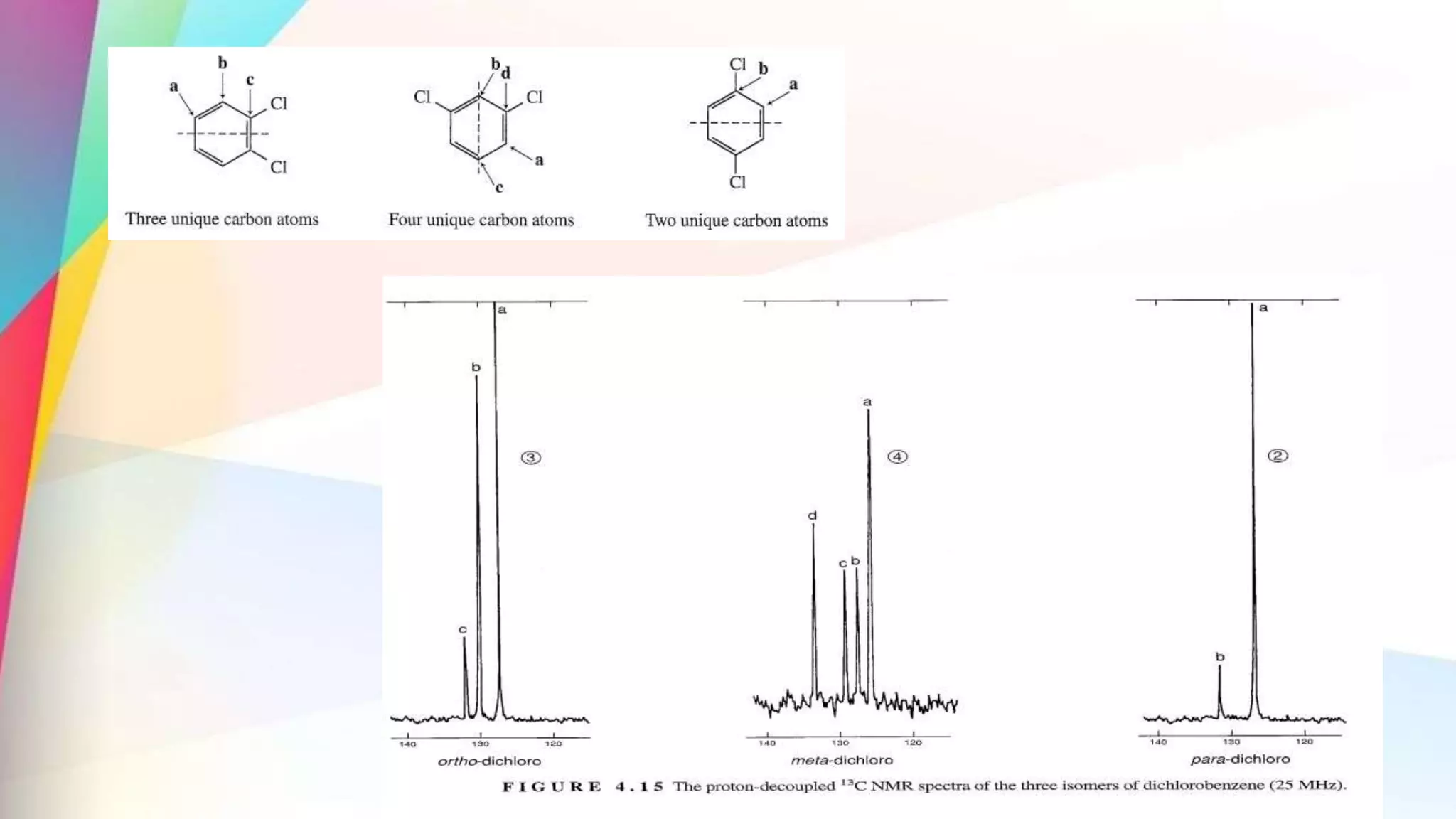
The document discusses the principles and applications of 13-carbon nuclear magnetic resonance (13C NMR), highlighting its ability to determine the structure of organic compounds through chemical shifts and the number of carbon signals. It outlines the techniques used for signal analysis, including decoupling methods and the significance of nuclear overhauser enhancement (NOE) in improving signal clarity. Additionally, it emphasizes the practical applications of 13C NMR in biological analysis and drug purity assessment.