Chemical Reaction And Equations
1. A chemical reaction is a process where reactants are converted into products. An equation is a symbolic representation of a reaction.
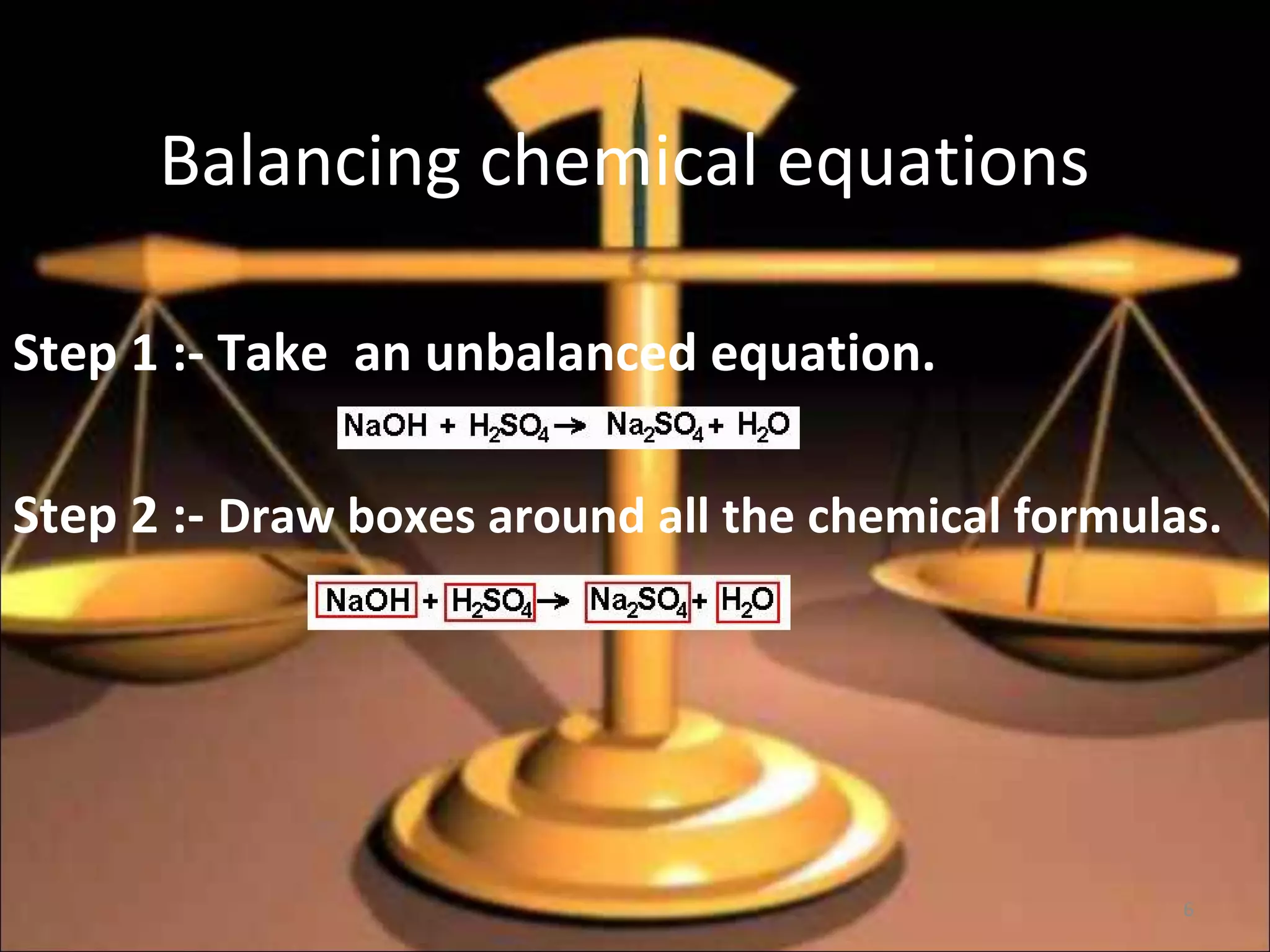
2. Key parts of a chemical equation include reactants on the left side of the arrow, products on the right side, and coefficients that indicate mole ratios.
3. There are several types of chemical reactions including combination, decomposition, displacement, and double displacement. Oxidation-reduction reactions involve the transfer of electrons between reactants.