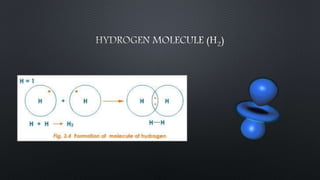
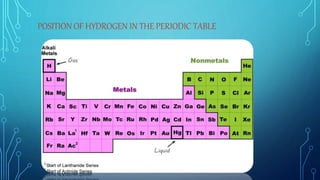
The document discusses hydrogen as the first element in the periodic table, highlighting its properties, methods of preparation, and chemical behavior. It covers hydrogen's role in forming dihydrogen, water's chemical properties, and various removal techniques for hardness in water. Additionally, the document mentions hydrogen peroxide, its preparation, uses, and the significance of heavy water in nuclear reactors.