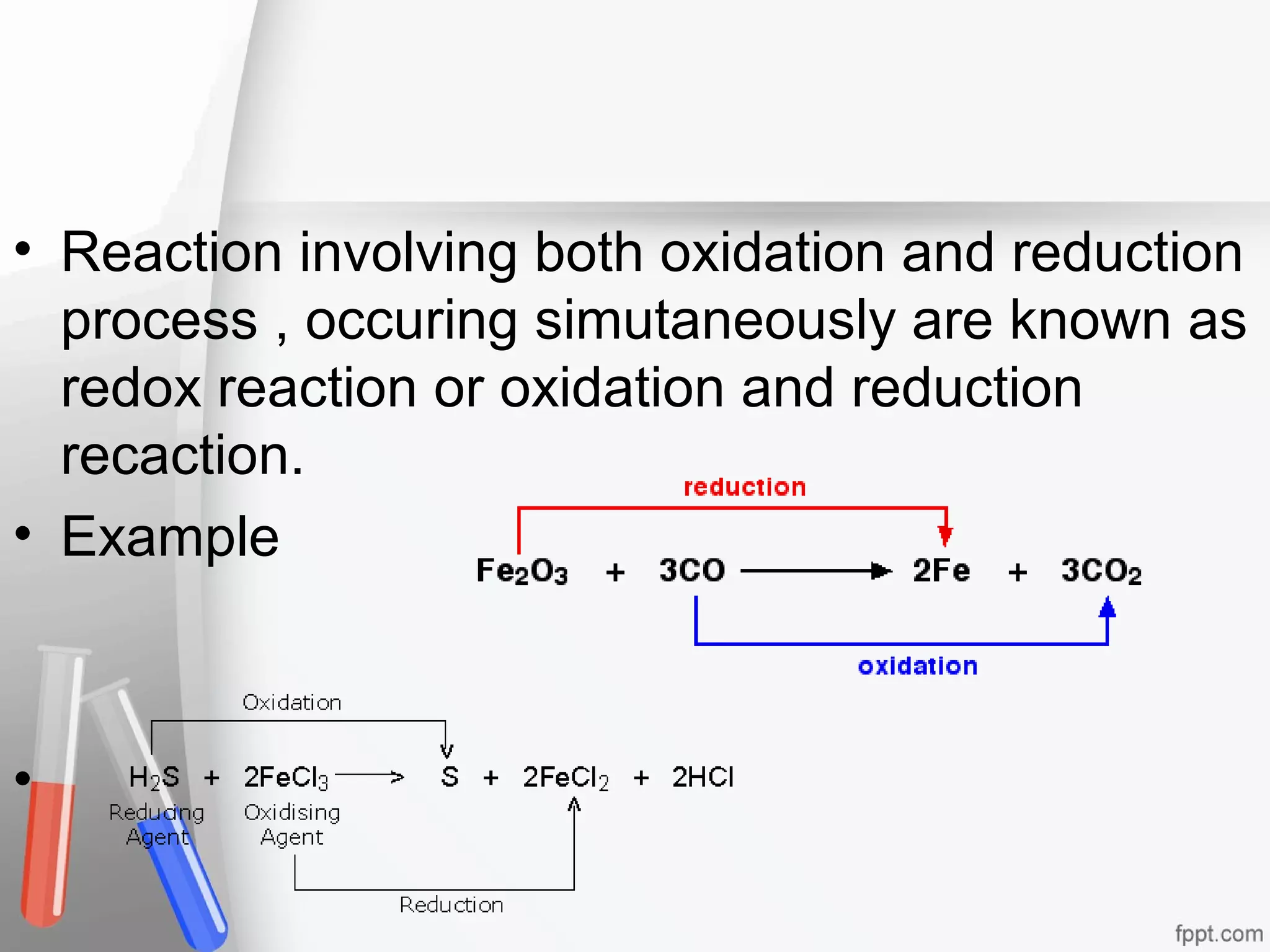
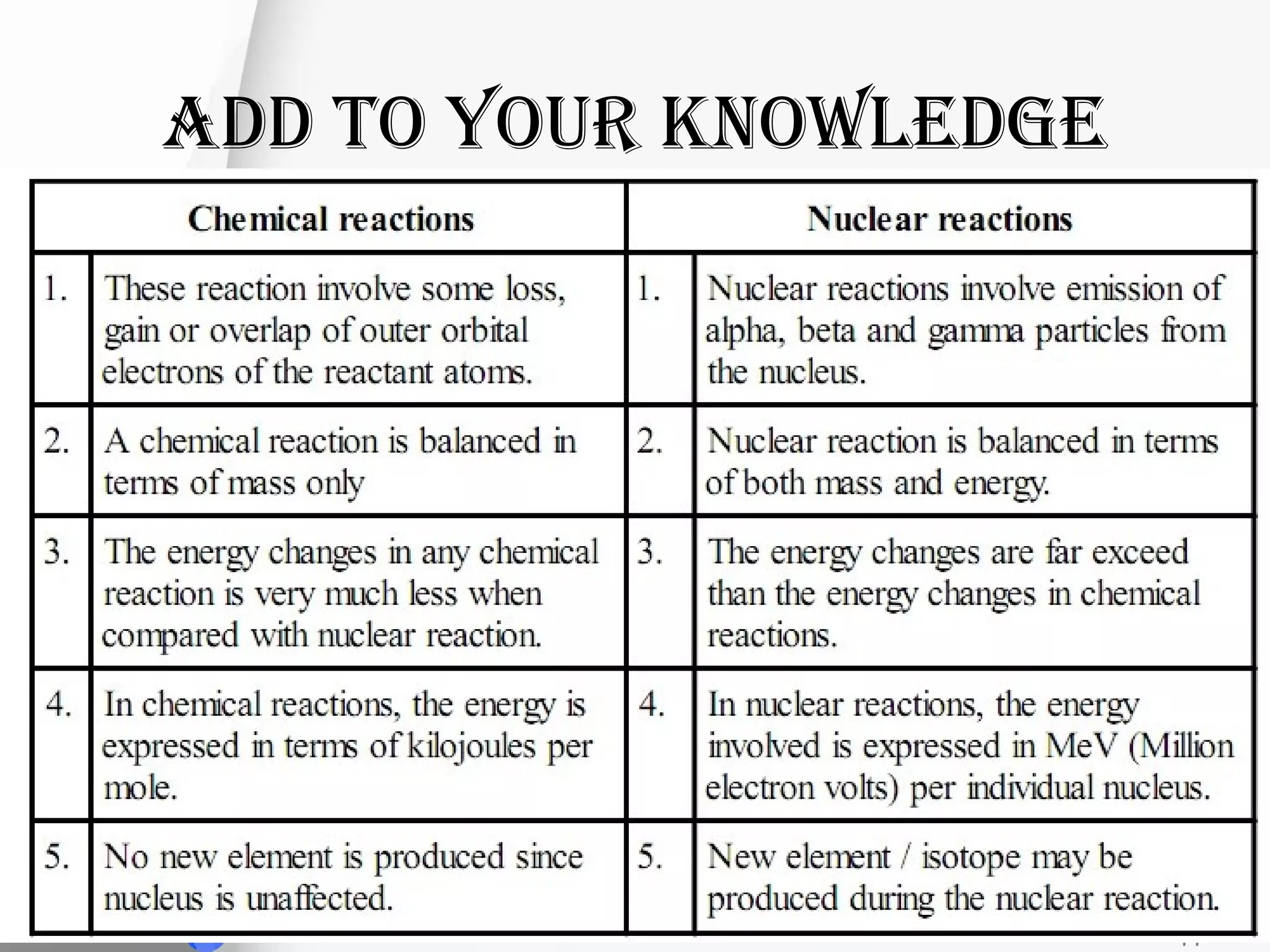
This document provides information on chemical reactions and equations. It defines a chemical reaction as a process that transforms one set of substances into another. It explains the different types of chemical equations like word equations, skeletal equations, and balanced equations. It also describes the different types of chemical reactions such as combination, decomposition, displacement, and redox reactions. It discusses oxidation and reduction processes and defines oxidizing and reducing agents. Examples of chemical equations are also provided.