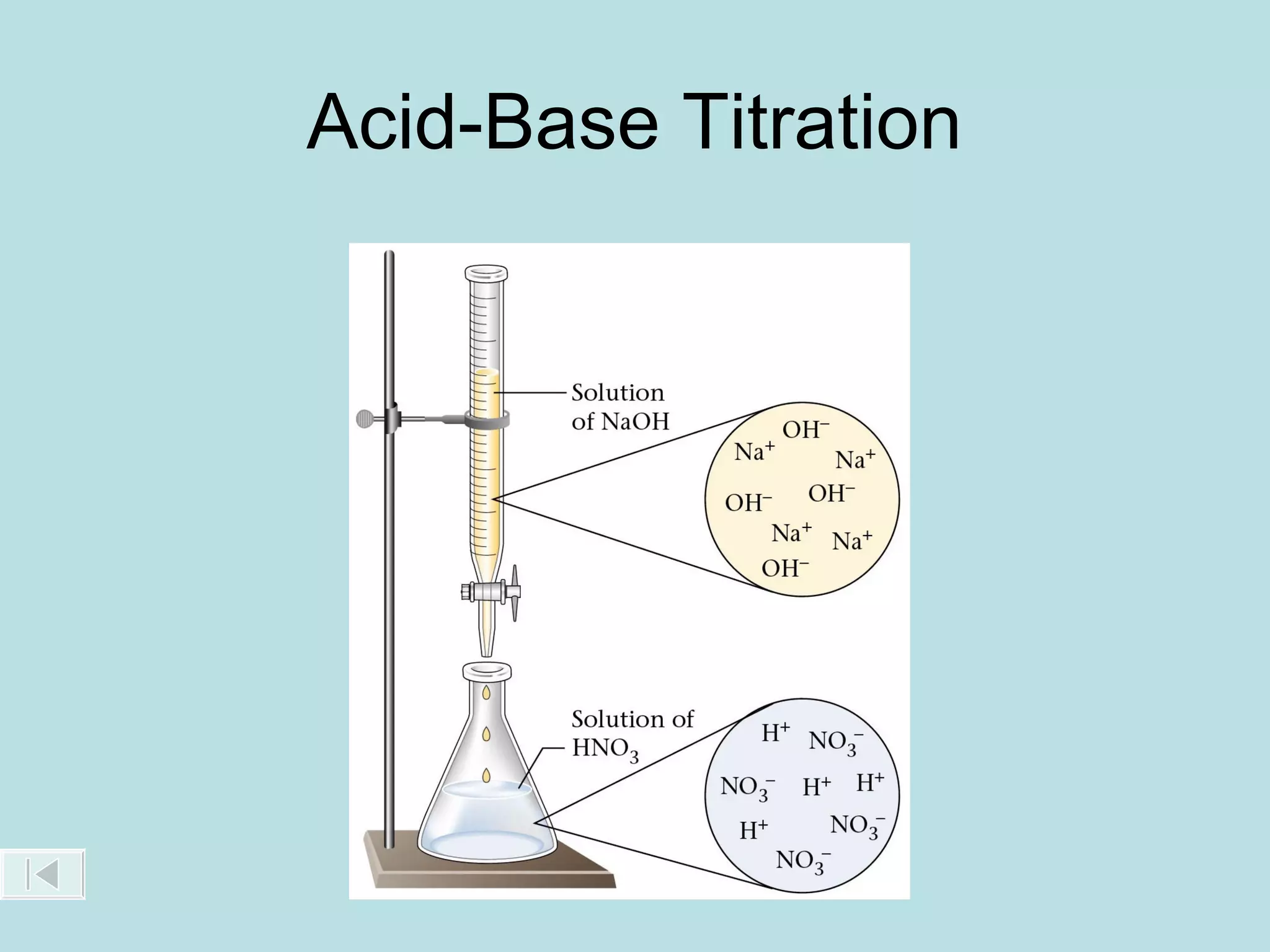
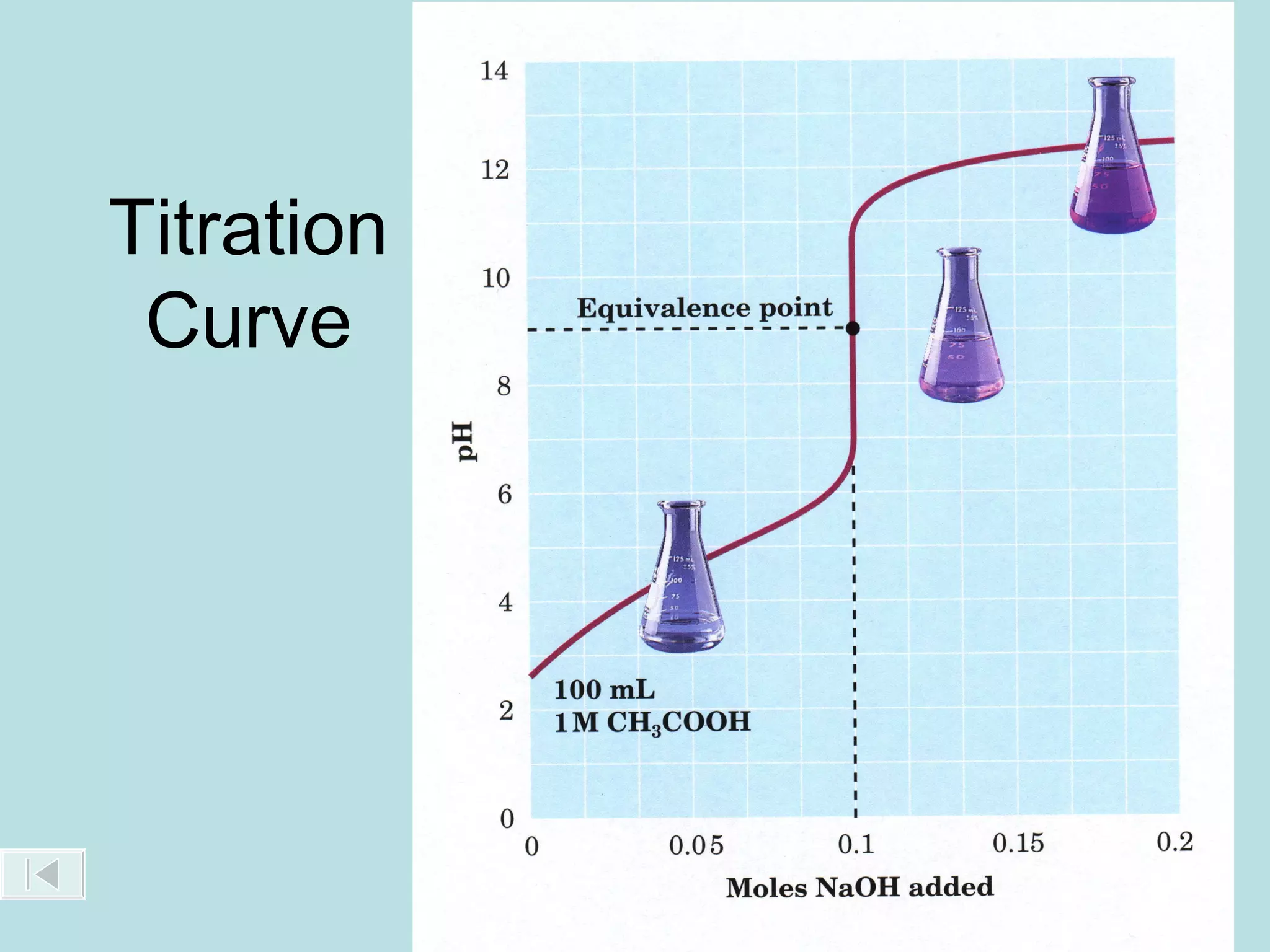
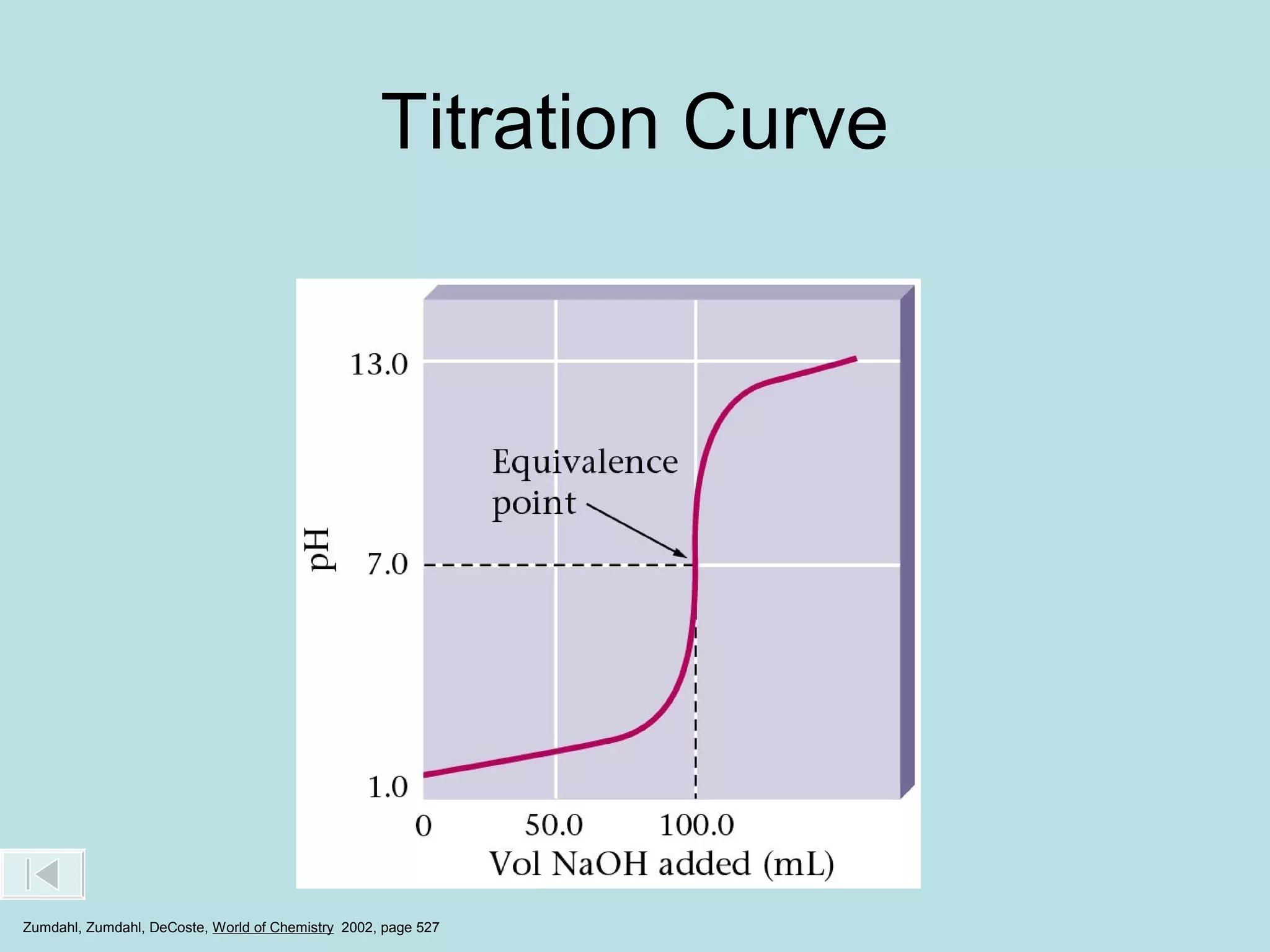
This document discusses acid-base titration and provides examples. It explains that acid-base titration is used to calculate the concentration of an acid or base of a known volume. It describes the setup and process for titrating an acid with a base, including using an indicator that changes color at the endpoint. Sample problems are worked through to calculate the concentration of an unknown acid or base by determining the moles of titrant used and applying stoichiometry to the balanced reaction.