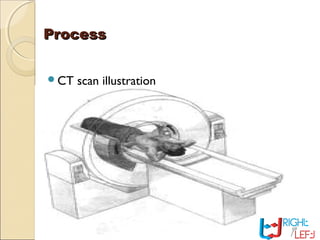
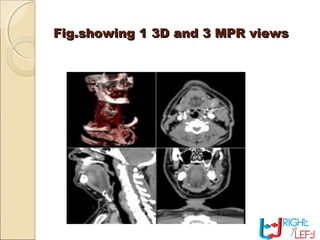
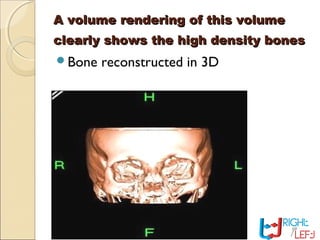
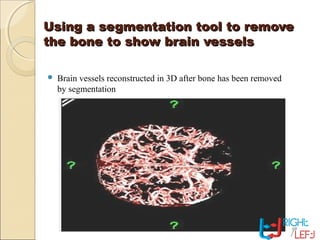
Computer tomography (CT) is a medical imaging technique that utilizes digital geometry processing to create three-dimensional images from a series of two-dimensional x-ray images. It was first developed by Godfrey Hounsfield and Allan Cormack, who shared the Nobel Prize in Medicine in 1979 for their contributions. CT scans have become a key tool in diagnosing various diseases and enhancing medical imaging through advanced techniques like multislice and helical CT, while also presenting some risks associated with contrast agents.