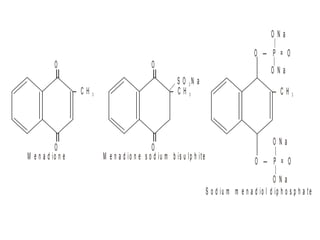
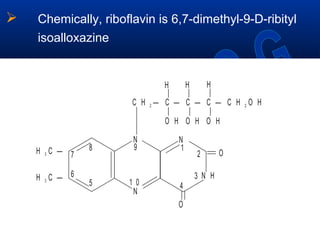
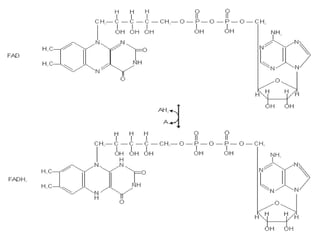
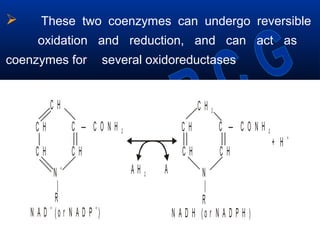
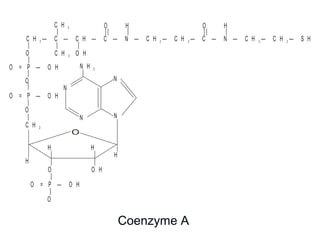
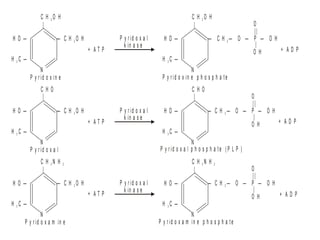
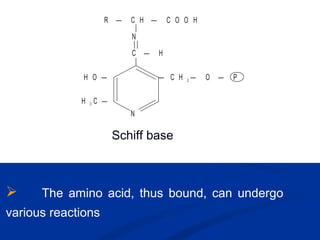
This document discusses vitamins, specifically thiamine, riboflavin, and niacin. It describes their chemical structures, functions as coenzymes, dietary sources, requirements, and deficiency diseases. Thiamine deficiency causes beriberi, while riboflavin deficiency causes angular stomatitis and glossitis. Niacin deficiency results in pellagra characterized by stomatitis, glossitis, diarrhea, and dermatitis. All three are water-soluble vitamins that must be obtained daily through diet as they are not stored in the body.























































































































































































![β - C a r o t e n e
[ O ]2
β - C a r o t e n e d io x y g e n a s e ,
b ile s a lt s
R e t in o l
N A D P H + H +
N A D P +
R e t in a ld e h y d e r e d u c t a s e
( r e t in e n e r e d u c t a s e )
C H 3
|
C
C H 3
C H 3
C
H
|
H C3
C
C
H
|
C
C
H
|
C
C
H
|
C H 3 C H 3
| | | |
H H H H H
| | | | |
H H H C3
C
C
H
|
C
C
|
C
C
H
|
C
C
|
C
C
H
|
C H 3C H 3
C H 3
|
C
C H 3
C H 3
C
H
|
H C3
C
C
H
|
C
C
H
|
C
C
H
|
C H 3 C H 3
| | | |
H H H H
| | | |
H H H C3
C H O
O H C
C
C
|
C
C
H
|
C
C
|
C
C
H
|
C H 3C H 3
+
C H 3
|
C
C H 3
C H 3
C
H
|
C
C
H
|
C
C
H
|
C
C
H
|
C H 3
C H 3
| |
H H
| |
C H O H2
R e t in o ic a c id
S p o n ta n e o u s
[ O ]
C H 3
C H 3
| | |
H H
| |
C
C
H
|
C C
C C
| |
C
C
H
|
C H 3
C H 3 C H 3
C O O H
H H
|
C H 3
|
C H 3
R e t in a l R e t in a l](https://image.slidesharecdn.com/vitamins-170219174659/85/Vitamins-191-320.jpg)