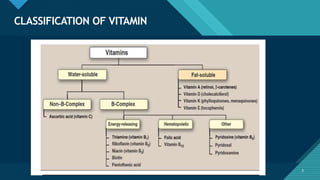
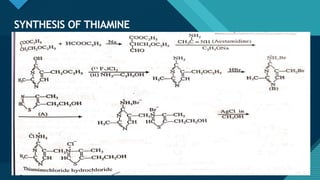
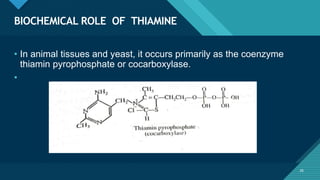
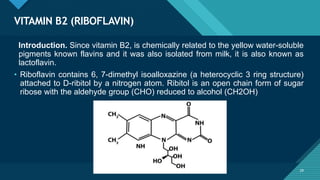
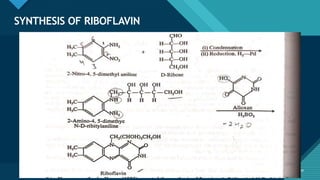
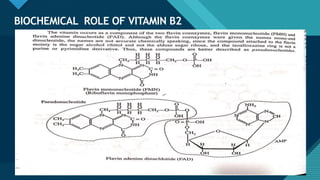
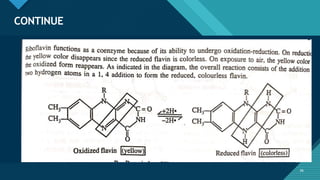
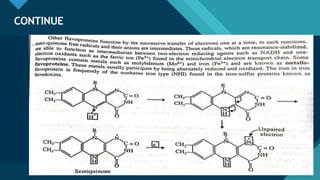
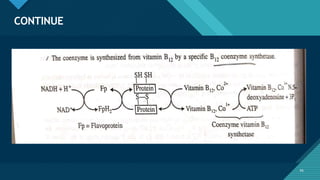
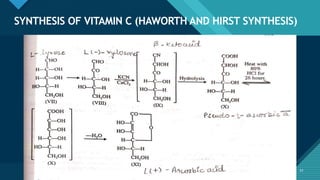
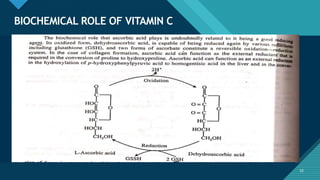
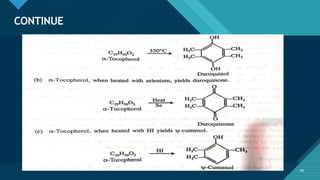
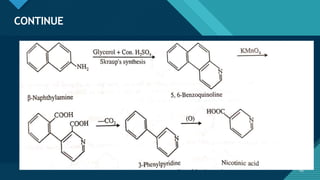
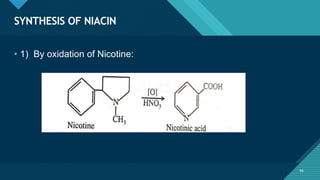
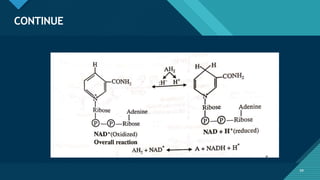
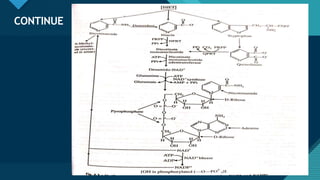
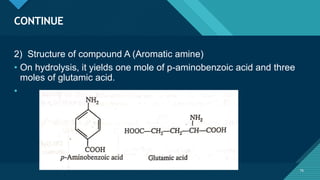
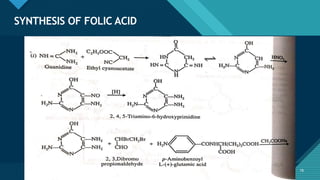
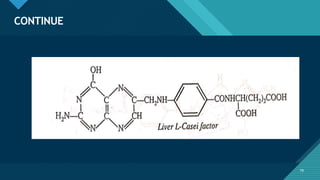
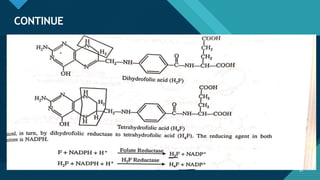
This document provides information on vitamin A, including its:
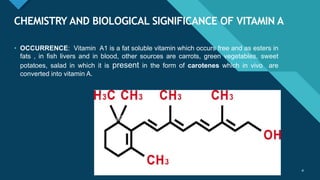
- Occurrence in foods like fish liver, carrots, and green vegetables
- Deficiency causes night blindness and xerophthalmia if severe
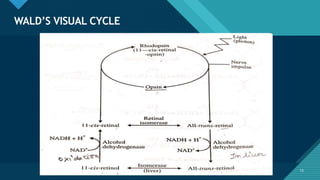
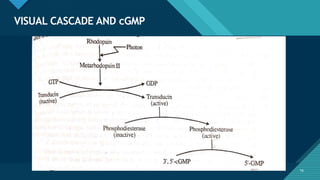
- Role in vision through the visual cycle and rhodopsin regeneration
- Other roles in growth, reproduction, and epithelial cell maintenance
It also discusses the chemistry, isolation, constitution, and biosynthesis of vitamin A.