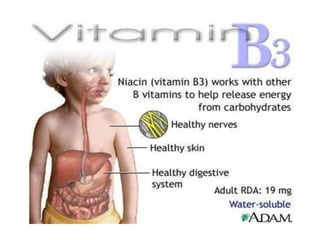
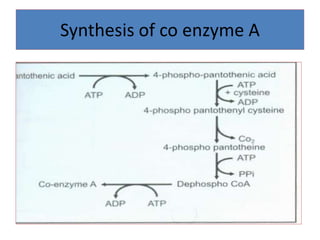
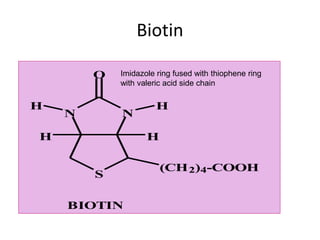
This document provides an overview of the B complex vitamins, focusing on Thiamine (B1). It discusses the chemistry, functions, deficiency symptoms, and recommended daily intake of Thiamine. Key points include: Thiamine acts as a coenzyme (Thiamine Pyrophosphate) and is important for energy metabolism. Deficiency can cause Beriberi, with symptoms of weakness, edema, and nerve damage. Good sources include whole grains, yeast, and the aleurone layer of cereals.

















































![FOLIC ACID(B9)
• The term ‘FOLIUM’ (latin ) means leaf of vegetable. Folic acid
is abundant in vegetables.
• It also obtained by yeast
• Other common name(s): folate, folacin, vitamin B9 vitamin M
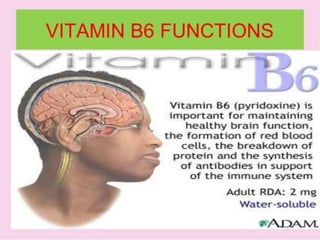
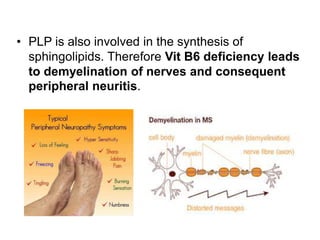
• Vitamin B9 (folic acid and folate) is essential for numerous bodily
functions. Humans cannot synthesize folate de novo; therefore,
folate has to be supplied through the diet to meet their daily
requirements.
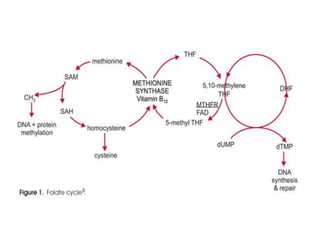
• The human body needs folate to synthesize DNA, repair DNA, and
methylate DNA as well as to act as a cofactor in certain biological
reactions.[7]It is especially important in aiding rapid cell
division and growth, such as in infancy and
pregnancy. Children and adults both require folic acid
to produce healthy red blood cellsand prevent anemia.[8]
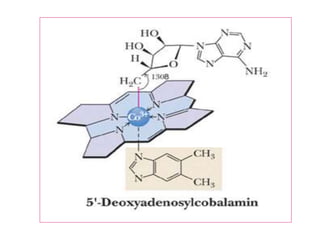
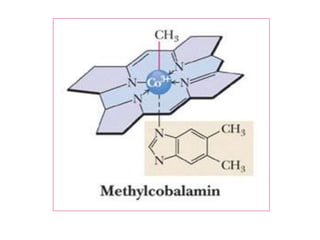
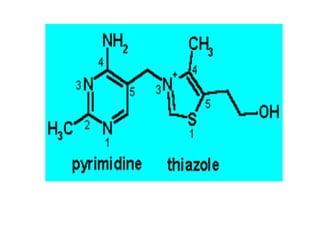
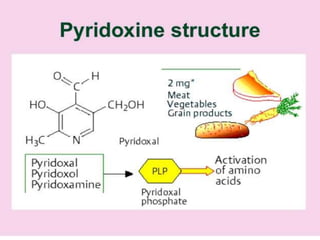
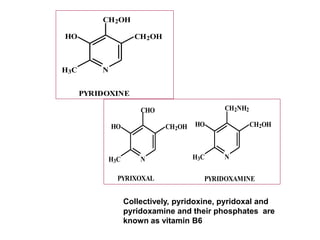
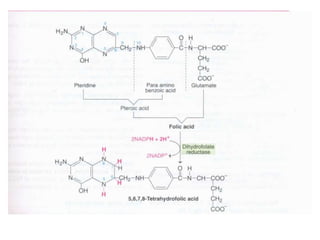
• CHEMISTRY: Folic acid is composed of three constituents.
Pteridine + PABA to form pteroic acid. This is attached to glutamic
acid to form pteroyl glutamic acid or folic acid. Initially folic acid is in
oxidised form. But reduced form is only active form
• Active co enzyme form is 5,6.7,8 tetrahydrofolic acid](https://image.slidesharecdn.com/bandc-230322151702-027d600c/85/B-and-C-ppt-68-320.jpg)




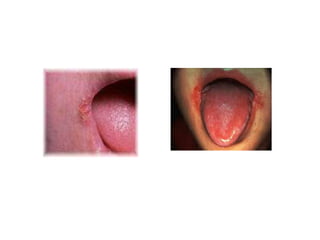
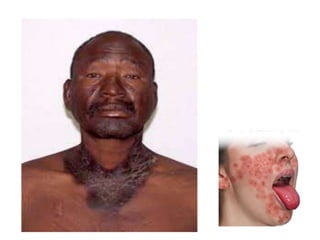
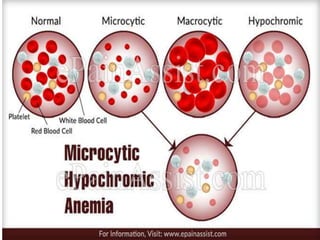
![• DEFICIENCY MANIFESTATIONS:
Common symptoms of folate deficiency include
diarrhea, macrocytic anemia with weakness or shortness of
breath, nerve damage with weakness and limb numbness
(peripheral neuropathy),[11] pregnancy complications, mental
confusion, forgetfulness or other cognitive declines, mental
depression, sore or swollen tongue, peptic or mouth ulcers,
headaches, heart palpitations, irritability, and behavioral
disorders. And also..
1. Reduced DNA synthesis and cell growth is reduced](https://image.slidesharecdn.com/bandc-230322151702-027d600c/85/B-and-C-ppt-74-320.jpg)