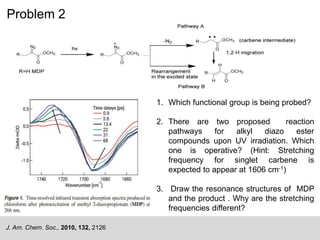
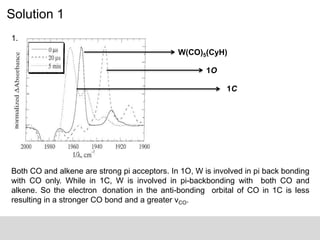
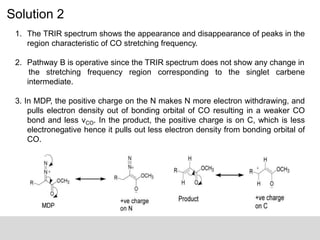
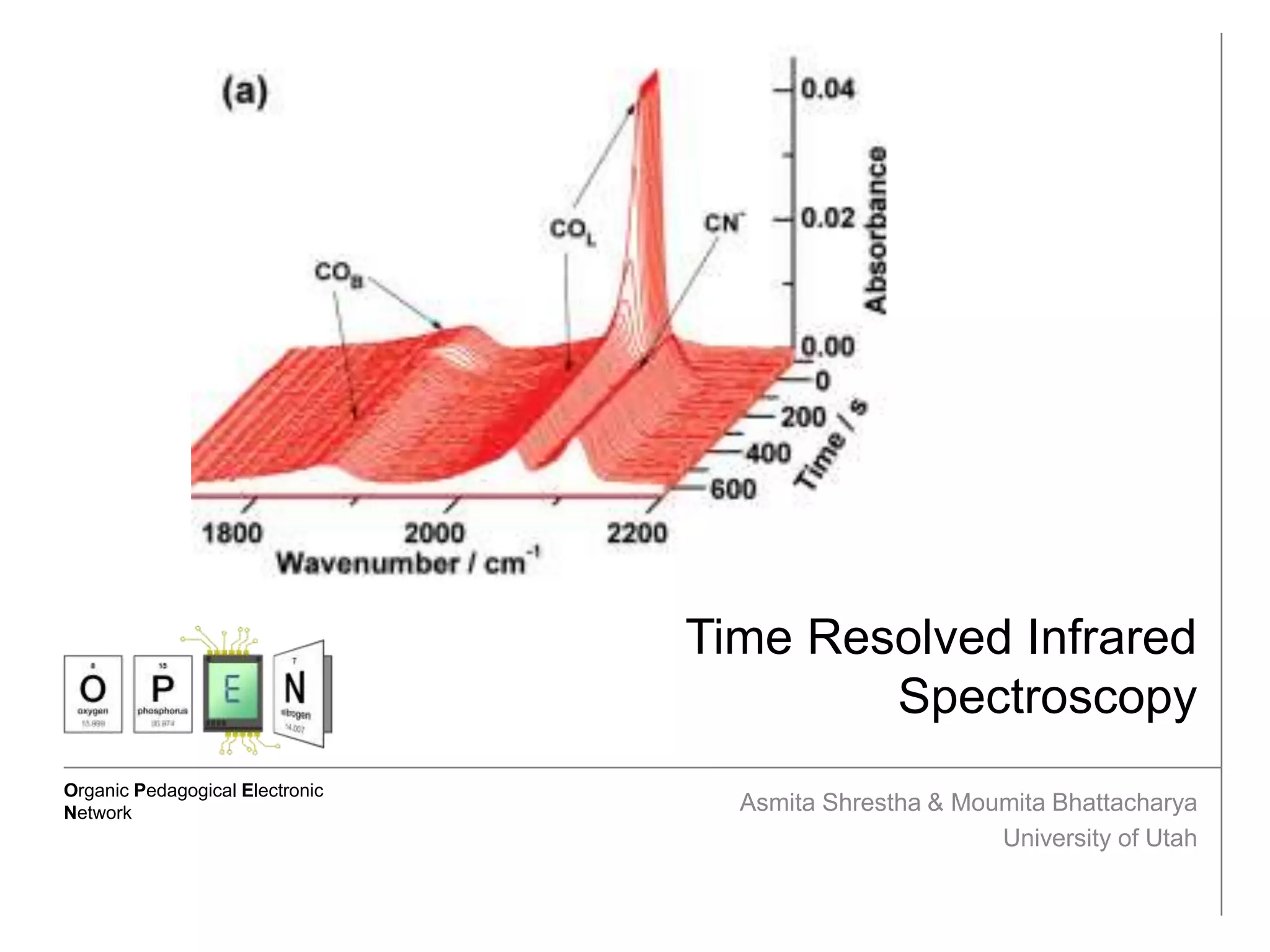
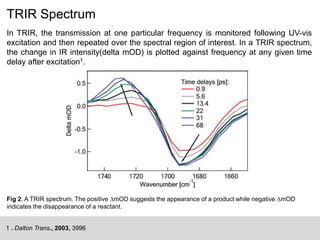
This document describes the principles and applications of time-resolved infrared spectroscopy (TRIR). TRIR combines ultraviolet flash photolysis with fast infrared detection to monitor excited states and short-lived reaction intermediates. It provides TRIR spectra of a reaction between a metal carbonyl and an organic ligand, showing the appearance and disappearance of product and reactant peaks. The document also presents two example problems demonstrating how TRIR can be used to determine reaction mechanisms and identify isomers.


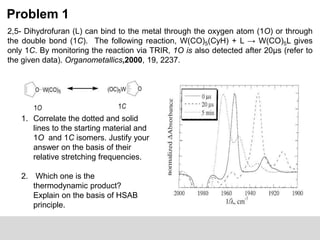
![Problem 1:
3. What is the order of the reaction? Is all 1O converted to 1C?
4. The following table suggests that kobs is independent of [W(CO)6] and
[2,5-DHF], is the isomerization inter or intramolecular?
1O
1C
[W(CO)6],
molL-1
(X103 )
[2,5-
DHF],
molL-1
T(oC) kobs (s-1)
0.2 0.022 20 1.09
59.6 46.94
0.5 0.042 29.3 2.84
59.4 40.55](https://image.slidesharecdn.com/timeresolvedinfraredspectroscopy-170125172739/85/Time-resolved-infrared-spectroscopy-6-320.jpg)