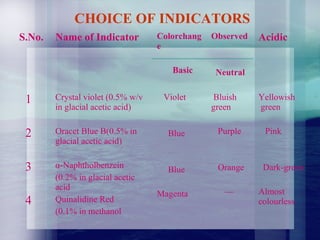
This document discusses non-aqueous titrations. It describes how non-aqueous solvents are used to titrate weakly acidic or basic substances when they are insoluble or have comparable strength to water. Examples of titrating primary amines like methyldopa using perchloric acid in acetic acid are provided. The document also lists various solvent types, reaction theory, standardization of perchloric acid and choice of indicators for non-aqueous titrations.









![EFFECT OF TEMPERATURE ON
ASSAYS
• Non-aqueous solvents possess greater coefficients of
expansion as compared to water, so there is effect of
temperature.
• The volume of titrant may be corrected by the
application of the following formula :
Vc = V [1 + 0.001 (tl + t2)]
Vc = Corrected volume of titrant,
V = Volume of titrant measured,
tl = Temperature at which titrant was standardized,
t2 = Temperature at which titration was performed.](https://image.slidesharecdn.com/nonaqueous-140623062542-phpapp01/85/Non-aqueous-titration-11-320.jpg)
![Titration of primary amines
• Ex: Methlyldopa
R.NH2 + HClO4 → [R.NH3]+
+ ClO4
-
• Specific reaction between methyldopa and perchloric
acid is expressed by the following equation](https://image.slidesharecdn.com/nonaqueous-140623062542-phpapp01/85/Non-aqueous-titration-12-320.jpg)





