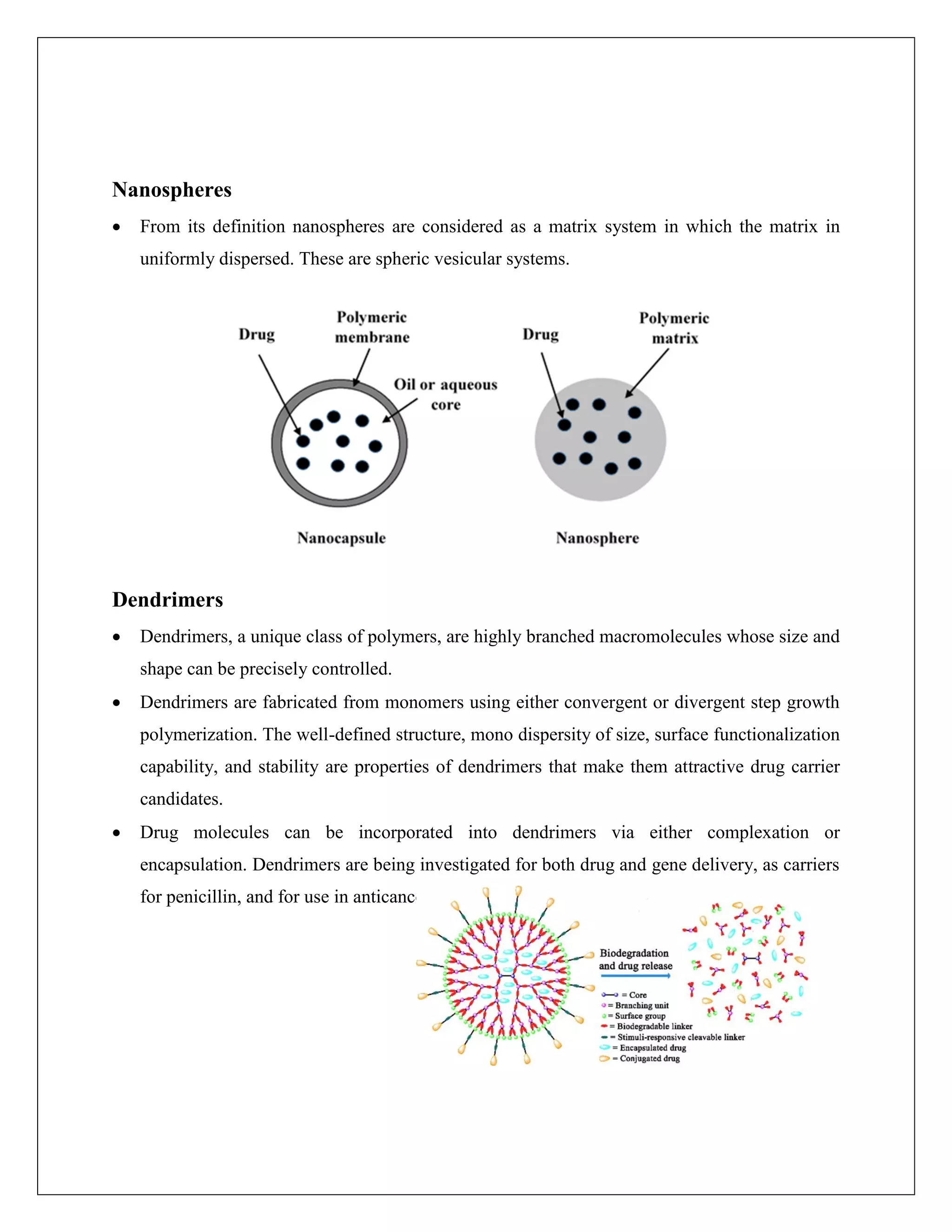
The document discusses different types of nanoparticles used in drug delivery, including liposomes, solid nanoparticles, polymeric nanoparticles, nanocapsules, nanospheres, dendrimers, nanotubes, nanowires, and nanocrystals. It also describes several methods for preparing nanoparticles, such as solvent evaporation, emulsions-diffusion, nanoprecipitation, salting out, and dialysis. Evaluation methods for prepared nanoparticles are discussed, including measuring yield, drug content, particle size, zeta potential, surface morphology, polydispersity index, in-vitro release studies, and kinetic studies.