Lecture 11 2013_
•Download as PPT, PDF•
0 likes•1,576 views
laser fundamentals
Report
Share
Report
Share
Recommended
To detemine the wavelength of semiconductor laser

The laser is part of almost all industrial sectors now. Laser is a coherent highly monochromatic concentrated beam of light.
Right from the computer data reading to metal welding the laser is used. The PowerPoint presentation here explains the laser experiment to determine the wavelength of a semiconductor laser, my the method of Grazing incidence (diffraction over the graduations of metal scale). The aim is to study the diffraction of patterns of laser scattered from the graduations of metal scale and hence determine the wavelength. The experiment is part of the physics curriculum in Technological universities and other science colleges.
Electromagnetic radiation

Electromagnetic radiation consists of photons, the quanta of electromagnetic fields. A freely-propagating photon in empty space (gravity-free, zero curvature vacuum) is described as a self-sustaining, helical traveling wave packet of quantized spin angular momentum moving at the speed of light. A photon is categorized as a stable, massless boson having no electric charge with spin angular momentum s = +/- hbar. The spin axis is aligned with the direction of wave vector k in either the forward or backward direction depending on helicity.
The observed EM frequency spectrum spans more than 140 octaves or ~24 orders of magnitude. The cutoff frequency of the vacuum is taken as the Planck frequency fsubP = 1.855E43 Hz.
Recommended
To detemine the wavelength of semiconductor laser

The laser is part of almost all industrial sectors now. Laser is a coherent highly monochromatic concentrated beam of light.
Right from the computer data reading to metal welding the laser is used. The PowerPoint presentation here explains the laser experiment to determine the wavelength of a semiconductor laser, my the method of Grazing incidence (diffraction over the graduations of metal scale). The aim is to study the diffraction of patterns of laser scattered from the graduations of metal scale and hence determine the wavelength. The experiment is part of the physics curriculum in Technological universities and other science colleges.
Electromagnetic radiation

Electromagnetic radiation consists of photons, the quanta of electromagnetic fields. A freely-propagating photon in empty space (gravity-free, zero curvature vacuum) is described as a self-sustaining, helical traveling wave packet of quantized spin angular momentum moving at the speed of light. A photon is categorized as a stable, massless boson having no electric charge with spin angular momentum s = +/- hbar. The spin axis is aligned with the direction of wave vector k in either the forward or backward direction depending on helicity.
The observed EM frequency spectrum spans more than 140 octaves or ~24 orders of magnitude. The cutoff frequency of the vacuum is taken as the Planck frequency fsubP = 1.855E43 Hz.
Fine structure constant

A dimensionless quantity described as a fundamental physical constant characterizing the coupling strength of the electromagnetic interaction. Introduced by Sommerfeld in 1916 to describe the spacing of splitting of spectral lines in multi-electron atoms, it is formed from four physical constants: electric charge, speed of light in vacuo, Planck's constant and electric permittivity of free space.
The inverse fine structure constant (=137.035999...) represents the spin precession whirl no. of the electron. The electron exhibits a slight precession due to an imbalance of electrostatic and magnetostatic energy levels. Electric charge is a result of this spin precession and represents a loop closure failure (torsion defect) similar to topological charge.
Rest mass results from quantum wave interference due to precession. Hence, electric charge, rest mass and the fine structure constant are interrelated and directly calculable.
ELECTRON SPIN RESONANCE SPECTROSCOPY

Describes about the principle,application and instrumentation of electron spin resonance spectroscopy
Interaction of x rays with matter

In this lecture, we will be talking only about the interaction of an ionizing electromagnetic radiation with matter, specifically about the interaction of X-Rays with the matter
Note: Gamma rays interact with the matter by the same way that X-rays interact with matter. In this lecture, we just focused on X-rays to complete our previous lecture about the production of X-rays
LASERS, CHARACTERISTICS, STIMULATED ABSORPTION, SPONTANEOUS EMISSION, STIMULA...

INTRODUCTION TO LASERS, CHARACTERISTICS
M.Sc.Part-II Sem- III (Unit - IV) Nuclear Magnetic Resonance Spectroscopy

Nuclear Magnetic Resonance Spectroscopy
Electron Spin Resonance Spectroscopy by arju

Detailed explanations about ESR Spectroscopy with my knowledge
Interaction of x ray with matter

This slide contains the Information regarding what changes takes place when the X-ray Interacts with matter.
Photoelectric effect ppt

what are the photoelectric effect. what are the photon ? all question answer is there.
Dual nature of radiation and matter class 12

Chapter 12th of NCERT book of class 12. Whole syllabus has been covered.
Basic Quantum Theory

Basic Quantum Theory:
Particle properties of waves
Blackbody radiation
Photoelectric effect
Compton effect
Wave properties of particles
Davisson–Germer experiment
Double slit experiment with particles
Thank you
Preeti Choudhary
Chaudharypreeti1997@gmail.com
https://www.linkedin.com/in/preeti-choudhary-266414182/
https://t.me/physiwaves
https://t.me/barcphysics
https://t.me/phyiscswaves
Class 12th Physics Photoelectric effect dual nature of matter radiations

Photoelectric effect dual nature of matter radiations important questions,concept and notes ppt
More Related Content
What's hot
Fine structure constant

A dimensionless quantity described as a fundamental physical constant characterizing the coupling strength of the electromagnetic interaction. Introduced by Sommerfeld in 1916 to describe the spacing of splitting of spectral lines in multi-electron atoms, it is formed from four physical constants: electric charge, speed of light in vacuo, Planck's constant and electric permittivity of free space.
The inverse fine structure constant (=137.035999...) represents the spin precession whirl no. of the electron. The electron exhibits a slight precession due to an imbalance of electrostatic and magnetostatic energy levels. Electric charge is a result of this spin precession and represents a loop closure failure (torsion defect) similar to topological charge.
Rest mass results from quantum wave interference due to precession. Hence, electric charge, rest mass and the fine structure constant are interrelated and directly calculable.
ELECTRON SPIN RESONANCE SPECTROSCOPY

Describes about the principle,application and instrumentation of electron spin resonance spectroscopy
Interaction of x rays with matter

In this lecture, we will be talking only about the interaction of an ionizing electromagnetic radiation with matter, specifically about the interaction of X-Rays with the matter
Note: Gamma rays interact with the matter by the same way that X-rays interact with matter. In this lecture, we just focused on X-rays to complete our previous lecture about the production of X-rays
LASERS, CHARACTERISTICS, STIMULATED ABSORPTION, SPONTANEOUS EMISSION, STIMULA...

INTRODUCTION TO LASERS, CHARACTERISTICS
M.Sc.Part-II Sem- III (Unit - IV) Nuclear Magnetic Resonance Spectroscopy

Nuclear Magnetic Resonance Spectroscopy
Electron Spin Resonance Spectroscopy by arju

Detailed explanations about ESR Spectroscopy with my knowledge
Interaction of x ray with matter

This slide contains the Information regarding what changes takes place when the X-ray Interacts with matter.
Photoelectric effect ppt

what are the photoelectric effect. what are the photon ? all question answer is there.
Dual nature of radiation and matter class 12

Chapter 12th of NCERT book of class 12. Whole syllabus has been covered.
Basic Quantum Theory

Basic Quantum Theory:
Particle properties of waves
Blackbody radiation
Photoelectric effect
Compton effect
Wave properties of particles
Davisson–Germer experiment
Double slit experiment with particles
Thank you
Preeti Choudhary
Chaudharypreeti1997@gmail.com
https://www.linkedin.com/in/preeti-choudhary-266414182/
https://t.me/physiwaves
https://t.me/barcphysics
https://t.me/phyiscswaves
Class 12th Physics Photoelectric effect dual nature of matter radiations

Photoelectric effect dual nature of matter radiations important questions,concept and notes ppt
What's hot (20)
B.Tech sem I Engineering Physics U-IV Chapter 1-ATOMIC PHYSICS

B.Tech sem I Engineering Physics U-IV Chapter 1-ATOMIC PHYSICS
LASERS, CHARACTERISTICS, STIMULATED ABSORPTION, SPONTANEOUS EMISSION, STIMULA...

LASERS, CHARACTERISTICS, STIMULATED ABSORPTION, SPONTANEOUS EMISSION, STIMULA...
M.Sc.Part-II Sem- III (Unit - IV) Nuclear Magnetic Resonance Spectroscopy

M.Sc.Part-II Sem- III (Unit - IV) Nuclear Magnetic Resonance Spectroscopy
Photoelectric Effect And Dual Nature Of Matter And Radiation Class 12

Photoelectric Effect And Dual Nature Of Matter And Radiation Class 12
Class 12th Physics Photoelectric effect dual nature of matter radiations

Class 12th Physics Photoelectric effect dual nature of matter radiations
Viewers also liked
射頻電子 - [實驗第一章] 基頻放大器設計![射頻電子 - [實驗第一章] 基頻放大器設計](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![射頻電子 - [實驗第一章] 基頻放大器設計](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
E.E. Essential Knowledge Sereies
My Online Courses: https://www.byparams.com/courses
基頻放大器設計
Viewers also liked (20)
Similar to Lecture 11 2013_
Lecture 04; spectral lines and broadening by Dr. Salma Amir

spectral lines and broadening , lorentz, pressure, collisional broadening
Pigments and Colors:Extraction,Characterization

Pigments and Colors: Extraction, Purification and Characterization using different modern analytical tehcniques
Analytical class atomic absorption spectroscopy, P K MANI

Analytical class, theory of atomic absorption spectroscopy and its utility
Fundamentals of modern physics

The chapter contains fundamentals of Modern physics, the Quantumtheory explanation of Black body radiation photoelectric effect and Compton effect, and the beginning of the de-Broglie hypothesis, wave-like properties of matter, and its proof explained in detail. It is highly useful for first-year B.Tech and BE students.
4 radio wave propagation over the earth

Describes the Electromagnetic Wave Propagation over the Earth Surface. Please send comments to solo.hermelin@gmail.com.
For more presentations on different subjects pleade visit my website at http://www,solohermelin.com.
This presentation is in the Radar folder.
Similar to Lecture 11 2013_ (20)
Lecture 04; spectral lines and broadening by Dr. Salma Amir

Lecture 04; spectral lines and broadening by Dr. Salma Amir
Interaction of radiation with Matter - Dr. Vandana

Interaction of radiation with Matter - Dr. Vandana
Analytical class atomic absorption spectroscopy, P K MANI

Analytical class atomic absorption spectroscopy, P K MANI
More from Gabriel O'Brien
Superconducting qubits for quantum information an outlook

paper ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Quantum computation with superconductors

Quantum technology, quantum computing, quantum sensing, quantum cryptography
Ion trap quantum computation

Magazine ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Entangled states of trapped atomic ions

paper ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Cold atoms trapped by nanostructures

paper ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Semiconductor qubits in practice

paper ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
A silicon based nuclear spin quantum computer

paper ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Spin qubits for quantum information processing

book chapter ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Quantum mechanical spin

slides ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Quantum computer based on color centers in diamond

paper ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Electron spin resonance

book chapter ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Fox m quantum_optics_an_introduction_photon antibunching1

book chapter ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Fox m quantum_optics_an_introduction_optical cavities

book chapter ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Interaction of light and matter

book chapter ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Single photon sources and detectors

Paper ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Invited review article single photon sources and detectors

Paper ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
The electromagnetic field

Book chapter ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Quantum jumps of light recording the birth and death of a photon in a cavity

Paper ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
Quantum entanglement

Book chapter ;
Quantum technology, quantum computing, quantum sensing, quantum cryptography
More from Gabriel O'Brien (20)
Superconducting qubits for quantum information an outlook

Superconducting qubits for quantum information an outlook
Quantum computer based on color centers in diamond

Quantum computer based on color centers in diamond
Fox m quantum_optics_an_introduction_photon antibunching1

Fox m quantum_optics_an_introduction_photon antibunching1
Fox m quantum_optics_an_introduction_optical cavities

Fox m quantum_optics_an_introduction_optical cavities
Invited review article single photon sources and detectors

Invited review article single photon sources and detectors
Quantum jumps of light recording the birth and death of a photon in a cavity

Quantum jumps of light recording the birth and death of a photon in a cavity
Recently uploaded
Cancer cell metabolism: special Reference to Lactate Pathway

Normal Cell Metabolism:
Cellular respiration describes the series of steps that cells use to break down sugar and other chemicals to get the energy we need to function.
Energy is stored in the bonds of glucose and when glucose is broken down, much of that energy is released.
Cell utilize energy in the form of ATP.
The first step of respiration is called glycolysis. In a series of steps, glycolysis breaks glucose into two smaller molecules - a chemical called pyruvate. A small amount of ATP is formed during this process.
Most healthy cells continue the breakdown in a second process, called the Kreb's cycle. The Kreb's cycle allows cells to “burn” the pyruvates made in glycolysis to get more ATP.
The last step in the breakdown of glucose is called oxidative phosphorylation (Ox-Phos).
It takes place in specialized cell structures called mitochondria. This process produces a large amount of ATP. Importantly, cells need oxygen to complete oxidative phosphorylation.
If a cell completes only glycolysis, only 2 molecules of ATP are made per glucose. However, if the cell completes the entire respiration process (glycolysis - Kreb's - oxidative phosphorylation), about 36 molecules of ATP are created, giving it much more energy to use.
IN CANCER CELL:
Unlike healthy cells that "burn" the entire molecule of sugar to capture a large amount of energy as ATP, cancer cells are wasteful.
Cancer cells only partially break down sugar molecules. They overuse the first step of respiration, glycolysis. They frequently do not complete the second step, oxidative phosphorylation.
This results in only 2 molecules of ATP per each glucose molecule instead of the 36 or so ATPs healthy cells gain. As a result, cancer cells need to use a lot more sugar molecules to get enough energy to survive.
Unlike healthy cells that "burn" the entire molecule of sugar to capture a large amount of energy as ATP, cancer cells are wasteful.
Cancer cells only partially break down sugar molecules. They overuse the first step of respiration, glycolysis. They frequently do not complete the second step, oxidative phosphorylation.
This results in only 2 molecules of ATP per each glucose molecule instead of the 36 or so ATPs healthy cells gain. As a result, cancer cells need to use a lot more sugar molecules to get enough energy to survive.
introduction to WARBERG PHENOMENA:
WARBURG EFFECT Usually, cancer cells are highly glycolytic (glucose addiction) and take up more glucose than do normal cells from outside.
Otto Heinrich Warburg (; 8 October 1883 – 1 August 1970) In 1931 was awarded the Nobel Prize in Physiology for his "discovery of the nature and mode of action of the respiratory enzyme.
WARNBURG EFFECT : cancer cells under aerobic (well-oxygenated) conditions to metabolize glucose to lactate (aerobic glycolysis) is known as the Warburg effect. Warburg made the observation that tumor slices consume glucose and secrete lactate at a higher rate than normal tissues.
FAIR & AI Ready KGs for Explainable Predictions

The increased availability of biomedical data, particularly in the public domain, offers the opportunity to better understand human health and to develop effective therapeutics for a wide range of unmet medical needs. However, data scientists remain stymied by the fact that data remain hard to find and to productively reuse because data and their metadata i) are wholly inaccessible, ii) are in non-standard or incompatible representations, iii) do not conform to community standards, and iv) have unclear or highly restricted terms and conditions that preclude legitimate reuse. These limitations require a rethink on data can be made machine and AI-ready - the key motivation behind the FAIR Guiding Principles. Concurrently, while recent efforts have explored the use of deep learning to fuse disparate data into predictive models for a wide range of biomedical applications, these models often fail even when the correct answer is already known, and fail to explain individual predictions in terms that data scientists can appreciate. These limitations suggest that new methods to produce practical artificial intelligence are still needed.
In this talk, I will discuss our work in (1) building an integrative knowledge infrastructure to prepare FAIR and "AI-ready" data and services along with (2) neurosymbolic AI methods to improve the quality of predictions and to generate plausible explanations. Attention is given to standards, platforms, and methods to wrangle knowledge into simple, but effective semantic and latent representations, and to make these available into standards-compliant and discoverable interfaces that can be used in model building, validation, and explanation. Our work, and those of others in the field, creates a baseline for building trustworthy and easy to deploy AI models in biomedicine.
Bio
Dr. Michel Dumontier is the Distinguished Professor of Data Science at Maastricht University, founder and executive director of the Institute of Data Science, and co-founder of the FAIR (Findable, Accessible, Interoperable and Reusable) data principles. His research explores socio-technological approaches for responsible discovery science, which includes collaborative multi-modal knowledge graphs, privacy-preserving distributed data mining, and AI methods for drug discovery and personalized medicine. His work is supported through the Dutch National Research Agenda, the Netherlands Organisation for Scientific Research, Horizon Europe, the European Open Science Cloud, the US National Institutes of Health, and a Marie-Curie Innovative Training Network. He is the editor-in-chief for the journal Data Science and is internationally recognized for his contributions in bioinformatics, biomedical informatics, and semantic technologies including ontologies and linked data.
Earliest Galaxies in the JADES Origins Field: Luminosity Function and Cosmic ...

We characterize the earliest galaxy population in the JADES Origins Field (JOF), the deepest
imaging field observed with JWST. We make use of the ancillary Hubble optical images (5 filters
spanning 0.4−0.9µm) and novel JWST images with 14 filters spanning 0.8−5µm, including 7 mediumband filters, and reaching total exposure times of up to 46 hours per filter. We combine all our data
at > 2.3µm to construct an ultradeep image, reaching as deep as ≈ 31.4 AB mag in the stack and
30.3-31.0 AB mag (5σ, r = 0.1” circular aperture) in individual filters. We measure photometric
redshifts and use robust selection criteria to identify a sample of eight galaxy candidates at redshifts
z = 11.5 − 15. These objects show compact half-light radii of R1/2 ∼ 50 − 200pc, stellar masses of
M⋆ ∼ 107−108M⊙, and star-formation rates of SFR ∼ 0.1−1 M⊙ yr−1
. Our search finds no candidates
at 15 < z < 20, placing upper limits at these redshifts. We develop a forward modeling approach to
infer the properties of the evolving luminosity function without binning in redshift or luminosity that
marginalizes over the photometric redshift uncertainty of our candidate galaxies and incorporates the
impact of non-detections. We find a z = 12 luminosity function in good agreement with prior results,
and that the luminosity function normalization and UV luminosity density decline by a factor of ∼ 2.5
from z = 12 to z = 14. We discuss the possible implications of our results in the context of theoretical
models for evolution of the dark matter halo mass function.
The ASGCT Annual Meeting was packed with exciting progress in the field advan...

The ASGCT Annual Meeting was packed with exciting progress in the field advancing efforts to deliver highly promising therapies to more patients.
Nutraceutical market, scope and growth: Herbal drug technology

As consumer awareness of health and wellness rises, the nutraceutical market—which includes goods like functional meals, drinks, and dietary supplements that provide health advantages beyond basic nutrition—is growing significantly. As healthcare expenses rise, the population ages, and people want natural and preventative health solutions more and more, this industry is increasing quickly. Further driving market expansion are product formulation innovations and the use of cutting-edge technology for customized nutrition. With its worldwide reach, the nutraceutical industry is expected to keep growing and provide significant chances for research and investment in a number of categories, including vitamins, minerals, probiotics, and herbal supplements.
Astronomy Update- Curiosity’s exploration of Mars _ Local Briefs _ leadertele...

Article written for leader telegram
Orion Air Quality Monitoring Systems - CWS

Professional air quality monitoring systems provide immediate, on-site data for analysis, compliance, and decision-making.
Monitor common gases, weather parameters, particulates.
Structural Classification Of Protein (SCOP)

A brief information about the SCOP protein database used in bioinformatics.
The Structural Classification of Proteins (SCOP) database is a comprehensive and authoritative resource for the structural and evolutionary relationships of proteins. It provides a detailed and curated classification of protein structures, grouping them into families, superfamilies, and folds based on their structural and sequence similarities.
Recently uploaded (20)
ESR_factors_affect-clinic significance-Pathysiology.pptx

ESR_factors_affect-clinic significance-Pathysiology.pptx
PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION

PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION
Cancer cell metabolism: special Reference to Lactate Pathway

Cancer cell metabolism: special Reference to Lactate Pathway
Earliest Galaxies in the JADES Origins Field: Luminosity Function and Cosmic ...

Earliest Galaxies in the JADES Origins Field: Luminosity Function and Cosmic ...
The ASGCT Annual Meeting was packed with exciting progress in the field advan...

The ASGCT Annual Meeting was packed with exciting progress in the field advan...
Nutraceutical market, scope and growth: Herbal drug technology

Nutraceutical market, scope and growth: Herbal drug technology
Astronomy Update- Curiosity’s exploration of Mars _ Local Briefs _ leadertele...

Astronomy Update- Curiosity’s exploration of Mars _ Local Briefs _ leadertele...
Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...

Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...
platelets- lifespan -Clot retraction-disorders.pptx

platelets- lifespan -Clot retraction-disorders.pptx
In silico drugs analogue design: novobiocin analogues.pptx

In silico drugs analogue design: novobiocin analogues.pptx
Body fluids_tonicity_dehydration_hypovolemia_hypervolemia.pptx

Body fluids_tonicity_dehydration_hypovolemia_hypervolemia.pptx
Lecture 11 2013_
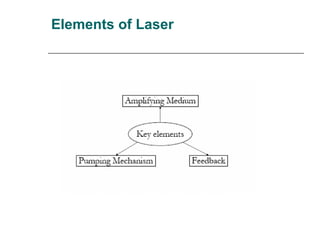
- 2. Working Principle of Laser
- 3. Working Principle of Laser Einstein’s assumptions & implications Thermodynamic equilibrium at arbitrary temperature T exists between the radiation field and the atoms The spectral density u(ν) of the radiation energy has the distribution characteristics of a blackbody at temperature T The atom population densities Nl and Nu at energy levels El and Eu, respectively, are distributed according to the Boltzman distribution at that temperature Population densities Nl and Nu are constant in time
- 4. Working Principle of Laser The radiative process and assumptions above, it follows that the rate of change of atoms in level Eu is given by ( ) ( )υυ uBNuBNAN dt dN lululuulu u +−−== 0 The spectral energy density can be written as ( ) 1 18 /3 33 − = kTh ec hn u υ υπ υ The Boltzman distribution ( ) kTh l ukTEE l u l u e g g e g g N N lu // υ−−− == …..(1) …..(2) …..(3)
- 5. Working Principle of Laser Solving eq.(1) in terms of u(υ) and substituting Nu/Nl from eq. (3), we obtain ( ) ( ) ul kTh lu u l ul ulullu ul BeB g g A BNNB A u − = − = // υ υ Compare it to eq. (2), gives 3 33 8 c hn B A ul ul υπ = lululu BgBg = ……(5) ……(6) ……(5)
- 6. Working Principle of Laser The importance of eqs. (5) and (6) cannot be underestimated. They tell us that: (i) The fundamental Einstein’s coefficients Aul, Bul and Blu are all inter-related. (ii) guBul = glBlu , i.e. stimulated emission and absorption are inverse processes. However note that the rate dNu/dt and dNl/dt differ depending on the population densities Nu and Nl. If Nu > Nl it leads to an increase in u(υ), an amplification. And if Nl > Nu it leads to a decrease in u(υ), an attenuation. For laser to operate, it is necessary that Nu be greater than Nl – a condition called population inversion. (iii) Since Bul/Aul is proportional to the reciprocal of the cube of the frequency, the higher the frequency the smaller Bul becomes in comparison with Aul. Since Bul is related to stimulated emission and Aul is related to spontaneous emission, it would seem that lasers of short wavelength radiation would be more difficult to build and operate.Two important ideas for the successful operation of a laser emerge from a review of Einstein’s study of the interaction of electromagnetic radiation with matter are, stimulated emission and population inversion.
- 7. Lasing condition Population inversion Necessary condition for amplification. The case of the upper level being more populated than the lower level. If stimulated emission rate exceeds absorption rate, net optical gain. The relationship for the intensity at a specific distance z into medium at a frequency ν and width Δν can be expressed as,
- 9. Population inversion If the value of the exponent is positive, the beam will increase in intensity and so amplification will occur. If it is negative, the beam will decrease in intensity and absorption will occur. The values of σul and z are always positive, thus amplification will occur only if This condition is not normal under thermal equilibrium l l u N g g N =u
- 11. Emission Broadening Homogeneous Broadening Due to the isotropic collisions with other atoms, which also causes non-radiative decay The processes lead to a Lorentzian distribution of emitting frequencies All of the atoms in level u have an equal probability of participating in the emission at any frequency ν of that emission shape – all atoms behave the same way
- 12. Emission Broadening Homogeneous Broadening The process can decrease either the decay time τu of the atoms residing in the excited level u OR affect the linewidth – depending on collision intervals Dephasing collisions interrupts the phase of radiating atoms without increasing their population decay rate
- 13. Emission Broadening Inhomogeneous Do not affect the lifetime, but do affect the linewidth The processes include Amorphous Crystal broadening, Doppler broadening and Isotope broadening Emission processes that lead to a Gaussian distribution of emitting frequencies Specific portions of the population density Nu contribute to different portions of the emission linewidth
- 14. Emission Broadening Amorphous Crystal Broadening Glass materials have various small regions oriented in slightly different directions Thus, each of the glass molecules can have slightly different energy levels This leads to different radiating frequencies for different regions Since the emission line is composed of the sum of all of the individual lines, this leads to a much broader emission spectrum
- 15. Emission Broadening Doppler Broadening Due to random movements of radiating atoms in all directions with a range of velocities This causes frequency shifts depending on the directions of the movements The faster the atoms move on the average, the broader the bandwidth A single photon might be able to stimulate one atom to emit because that atom happened to be Doppler shifted to the photon’s frequency, but it might not be able to stimulate another atom because it had a different Doppler shift than the first I.e. different atoms contribute to the gain at different frequencies within the laser bandwidth
- 16. Emission Broadening Isotope Broadening Due to the presence of more than one isotropic form of the species These different isotopes consist of atoms having the same number of protons and electrons, but with different numbers of neutrons Atoms with slightly different numbers of neutrons within their nuclei exhibit small differences in energy level values The slightly different energy level values for different isotopes provide slightly different frequencies for the transitions
