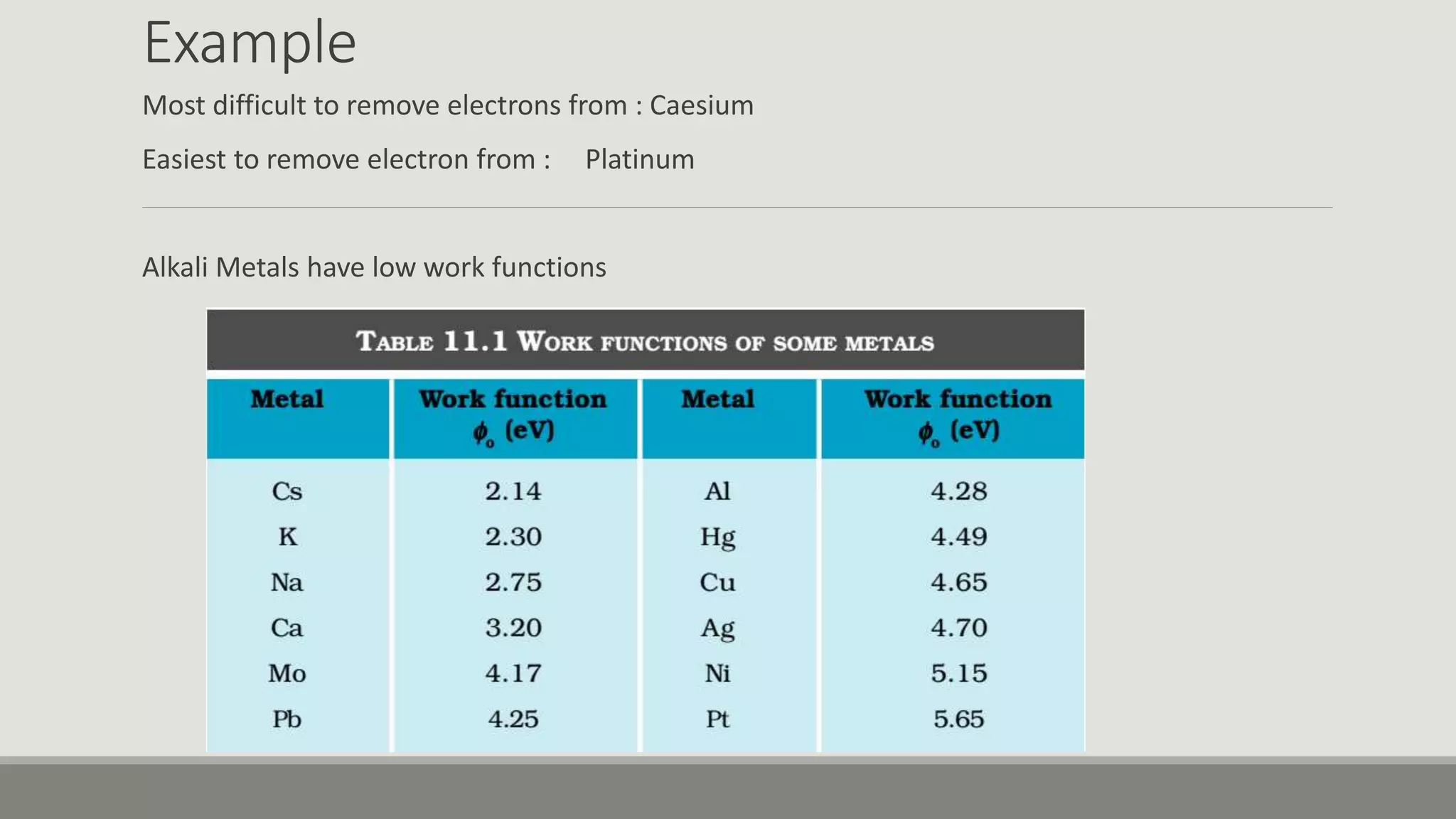
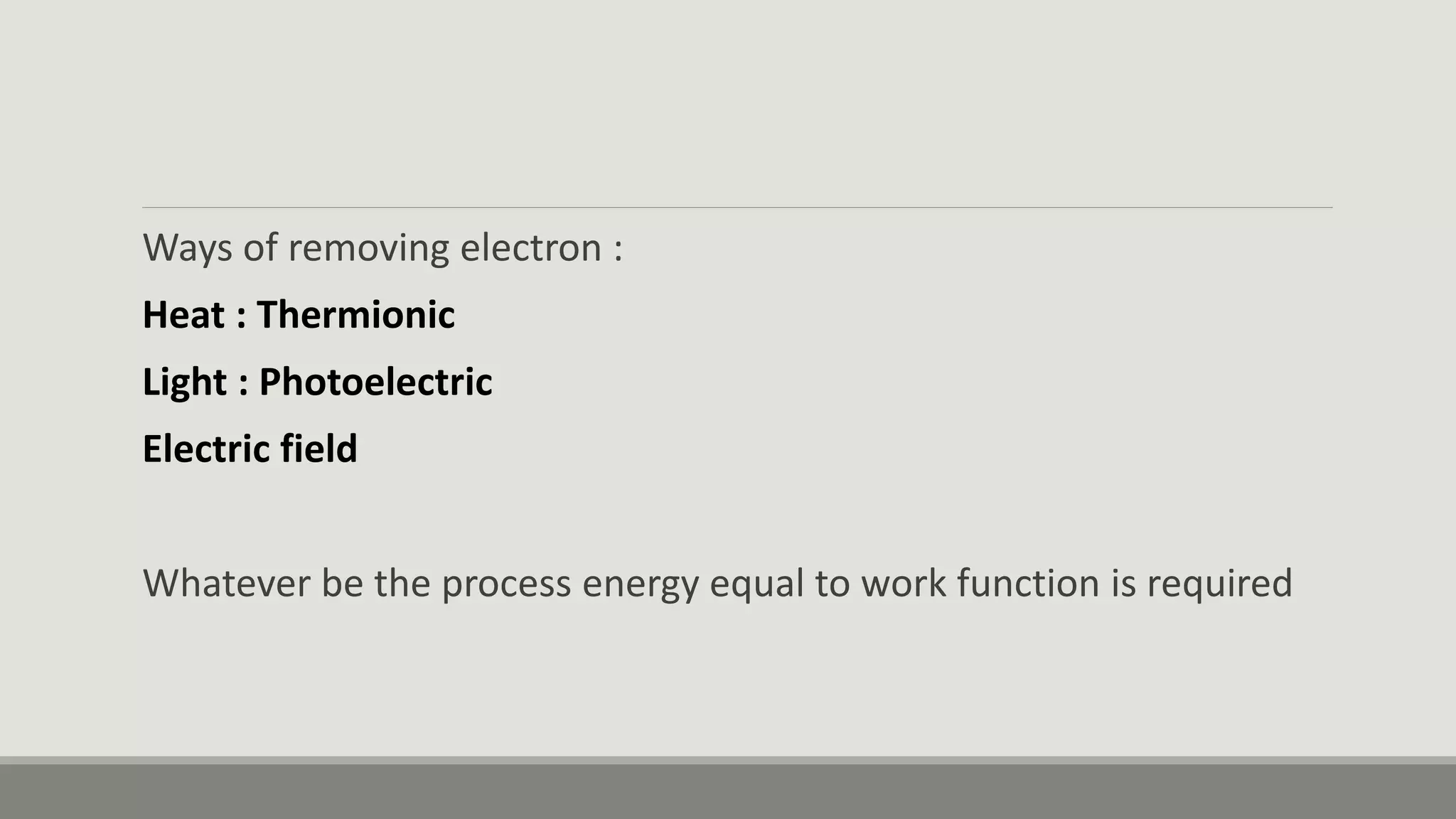
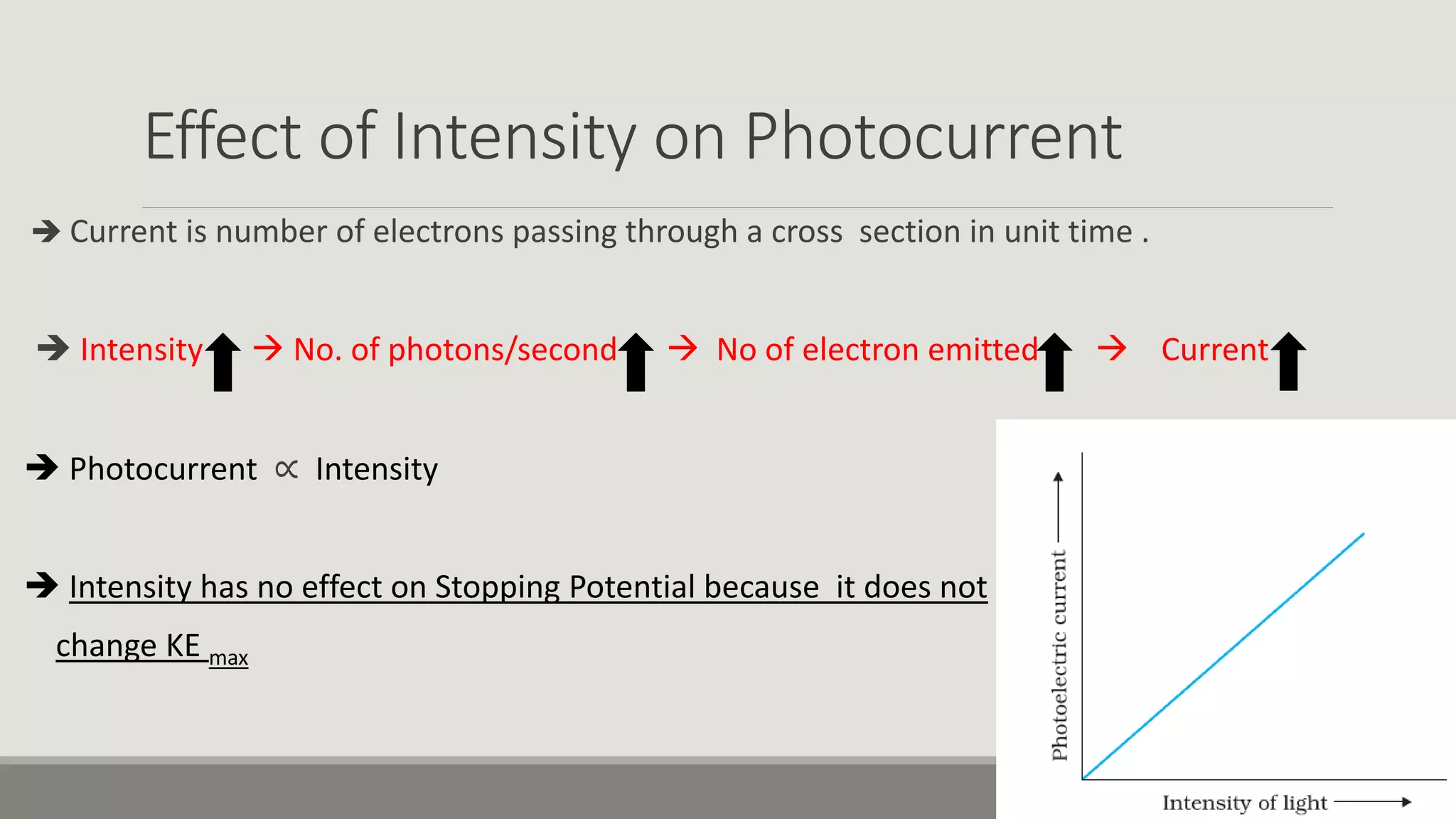
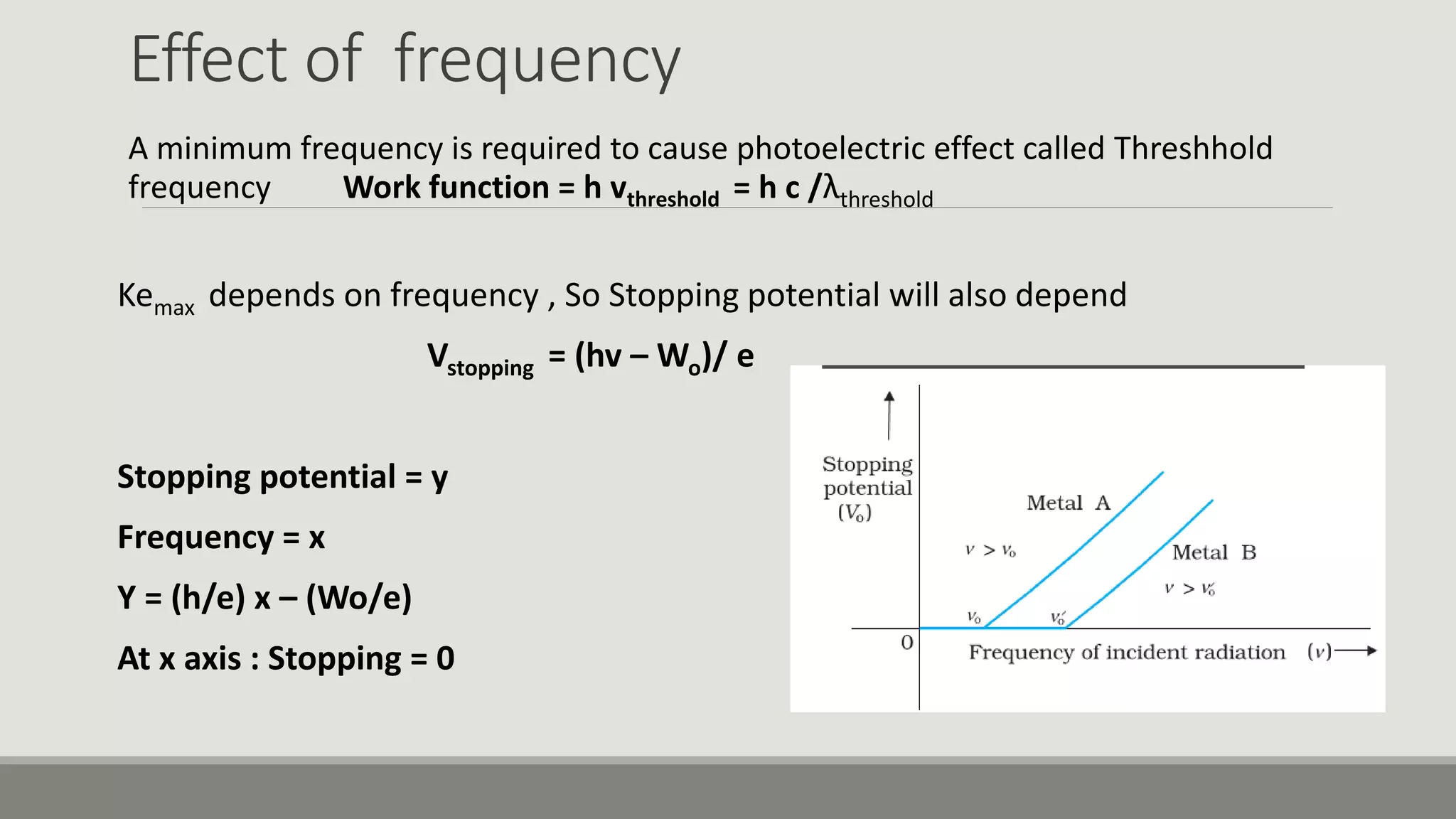
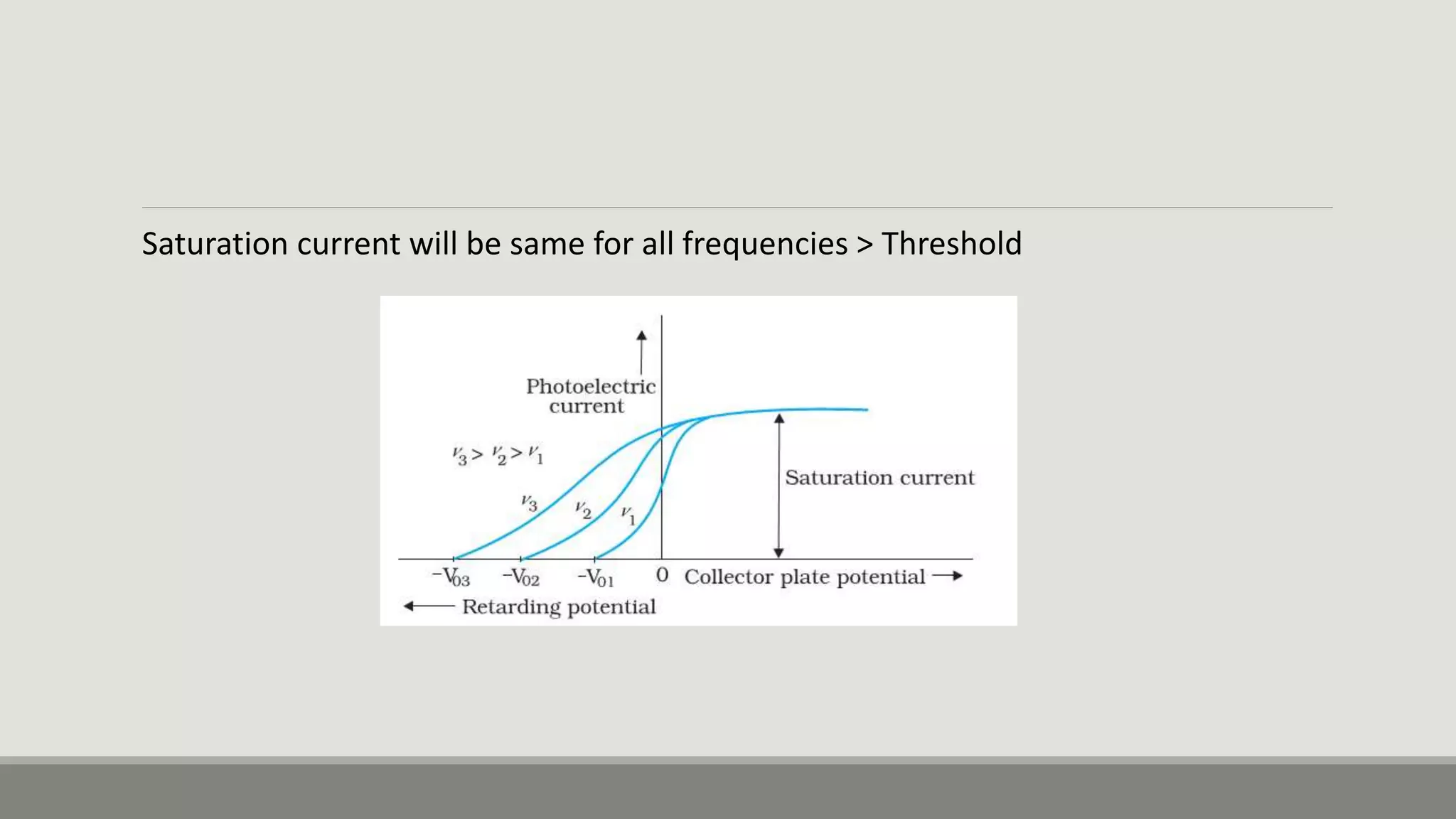
This document discusses the dual nature of radiation and matter. It explains the photoelectric effect and defines key parameters like work function, threshold frequency, stopping potential, and kinetic energy. The photoelectric effect can be explained using Einstein's equation that energy supplied must equal the work function plus maximum kinetic energy. Stopping potential and kinetic energy are shown to depend on frequency but not intensity. Plots of various variables are described and the wave-particle duality of light and matter is discussed.